Summary
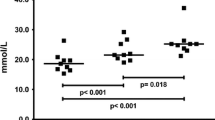
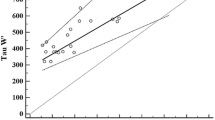
Physical performance deteriorates during strenuous exercise as manifested by a decrease in maximal aerobic power and increased activity of serum muscle enzymes. The relationship between these parameters was investigated in 41 trained subjects during 24 h marches and the following recovery period. Peak O2 uptake and serum activity of creatine phosphokinase (CPK) and glutamic oxalacetic transaminase (GOT) were measured. During the marches there was a simultaneous, significant elevation in serum CPK and GOT activity and a significant reduction in peak O2 uptake. During the early recovery period (24 h) no significant changes occurred in muscle enzyme activity and peak O2 uptake; thereafter (up to 72 h after the end of the march), a gradual decline in enzyme activity levels with a concomitant increase in peak O2 uptake was observed, reaching pre-march values. A “mirror image” relationship between muscle enzyme activity and peak O2 uptake was found during three clearly distinguished phases: a) 24 h march, b) early recovery stage and c) late recovery stage. These findings suggest, that muscle enzyme leakage from muscle cells is closely related to the decline in muscular function and aerobic power. Thus, muscle enzyme activity might be a practical measure of physical performance capacity during the early and late stages of recovery from prolonged endurance exercise.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Apple FS, Rogers MA, Sherman WA, Ivy JL (1984) Comparison of serum creatine kinase and creatine kinase MB activities post marathon race versus post myocardial infarction. Clin Chim Acta 138:111–118
Apple FS, Rogers MA, Casal DC, Sherman WM, Ivy JL (1985) Creatine Kinase — MB isoenzyme adaptations in stressed human skeletal muscle of marathon runners. J Appl Physiol 59:149–153
Astrand PO, Rodhal K (1977) Textbook of Work Physiology. McGraw-Hill, New York
Berg A, Haralambie G (1978) Changes in serum creatine kinase and hexose phosphate isomerase activity with exercise duration. Eur J Appl Physiol 39:191–201
Convertino VA, Greenleaf JE, Bernauer EM (1980) Role of thermal and exercise factors in the mechanism of hypervolemia. J Appl Physiol 48:657–664
Edwards RHT (1983) Biochemical basis of fatigue in exercise performance: catastrophe theory of muscle fatigue. In: Biochemistry of exercise. Proceeding of the 5th International Symposium. Human Kinetics Publishers Inc, Champaign, IL, pp 3–28
Ekblom B (1969) Effect of physical training on oxygen transport system in man. Acta Physiol Scand [Suppl] 328:9–45
Forssell G, Nordlander R, Nyquist O, Orinius E, Styrelius I (1975) Creatine phosphokinase after submaximal physical exercise in untrained individuals. Acta Med Scand 197:503–505
Galun E, Epstein Y (1984) Serum creatine kinase activity following a 120 km march. Clin Chem Acta 143:281–283
Gleser MA, Vogel JA (1973) Endurance capacity for prolonged exercise on the bicycle ergometer. J Appl Physiol 34:438–442
Gollnick PD, King DW (1969) The immediate and chronic effect of exercise on the number and structure of skeletal muscle mitochondria. In: Biochemistry of exercise medicine and sport, vol. 3. Karger, Basel, pp 239–244
Hunter JB, Critz JB (1971) Effect of training on plasma enzyme levels in man. J Appl Physiol 31:20–23
Karlsson J, Saltin B (1970) Lactate, ATP, and CP in the working muscles during exhaustive exercise in man. J Appl Physiol 29:598–602
Karlsson J, Nordasjo LO, Jorfeldt L, Saltin B (1972) Muscle lactate, ATP, and CP levels during exercise after physical training in man. J Appl Physiol 33:199–203
Kesseler G, Morgenstern S, Snyder L, Varady R (1975) Improved 3-point assay for ALT and AST in serm using the Technicon SMAC high speed computer-controlled biochemical analyzer to eliminate the common errors found in enzyme analysis. 9th Int Conf Clin Chem, Toronto
Knochel JP (1972) Exertional rabdomyolysis. N Engl J Med 287:927–929
Komi PV, Rusko H, Vos J, Vihko V (1977) Anaerobic performance capacity in athletes. Acta Physiol Scan 100:107–114
Magazanik A, Shapiro Y, Meytes D, Meytes I (1974) Enzyme blood levels and water balance during a marathon race. J Appl Physiol 36:214–217
Olerud JE, Homer LD, Carroll HW (1974) Incidence of acute exertional rabdomyolysis. Arch Int Med 136:692–697
Pandolf KB, Givoni B, Goldman RF (1977) Predicting energy expenditure with loads while standing or walking very slowly. J Appl Physiol 43:577–581
Reinhart WH, Staubli M, Kochli HP, Straub PW (1982) Creatine Kinase and MB-fraction after a long distance race. Clin Chim Acta 125:307–310
Ross JH, Attwood EC, Atkin GE, Villar RN (1983) A study on the effects of severe repetitive exercise on serum myoglobin, creatine kinase, transaminase and lactate dehydrogenase. Quart J Med 206:268–279
Salminen A, Vihko V (1983) Exercise myopathy: selectively enhanced proteolytic capacity in rat skeletal muscle after prolonged running. Exp Mol Pathol 38:61–68
Salminen A, Vihko V (1984) Autophagic response to strenuous exercise in mouse skeletal muscles fibers. Virchows Arch [Cell Pathol] 45:97–106
Saltin B, Rowell LB (1980) Functional adaptation to physical activity and inactivity. Fed Proc 39:1506–1513
Sanders TM, Bloor CM (1975) Effect of repeated endurance exercise on serum enzyme activities in well conditioned males. Med Sci Sports 7:44–47
Shapiro Y, Magazanik A, Sohar E, Reich CB (1973) Serum enzyme changes in untrained subjects following a prolonged march. Can J Physiol Pharmacol 51:271–276
Thomson WHS, Sweetin JC, Hamilton IJ (1975) ATP and muscle enzyme efflux after physical exertion. Clin Chem Acta 59:241–245
Wilkinson JH, Robinson JM (1974) Effect of energy-rich compounds on release of intracellular enzymes from human leukocytes and rat lymphocytes. Clin Chem 20:1331–1336
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Galun, E., Burstein, R., Tur-Kaspa, I. et al. Prediction of physical performance through muscle enzymes activity. Europ. J. Appl. Physiol. 57, 597–600 (1988). https://doi.org/10.1007/BF00418468
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00418468