Abstract
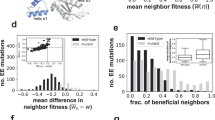
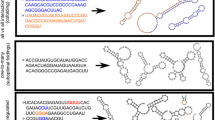
Evolution of RNA secondary structure is studied using simulation techniques and statistical analysis of fitness landscapes. The transition from RNA sequence to RNA secondary structure leads to fitness landscapes that have local variations in their “ruggedness.” Evolution exploits these variations. In stable environments it moves the quasispecies toward relatively “flat” peaks, where not only the master sequence but also its mutants have a high fitness. In a rapidly changing environment, the situation is reversed; evolution moves the quasispecies to a region where the correlation between secondary structures of “neighboring” RNA sequences is relatively low. In selection for simple secondary structures the movement toward flat peaks leads to pattern generation in the RNA sequences. Patterns are generated at the level of polynucleotide frequencies and the distribution of purines and pyrimidines. The patterns increase the modularity of the sequence. They thereby prevent the formation of alternative secondary structures after mutations. The movement of the quasispecies toward relatively rugged parts of the landscape results in pattern generation at the level of the RNA secondary structure. The base-pairing frequency of the sequences increases. The patterns that are generated in the RNA sequences and the RNA secondary structures are not directly selected for and can be regarded as a side effect of the evolutionary dynamics of the system.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Agur Z (1987) Resilience and variability in pathogens and hosts. IMA J Math Appl Med Biol 4:295–307
Agur Z, Kersberg M (1987) The emergence of phenotypic novelties through progressive change. Am Natur 129:862–875
Bonhoeffer S, McCaskill JS, Stadler PF, Schuster P (1993) RNA multi-structure landscapes. Eur Biophys J 22:13–24
Eigen M (1985) Macromolecular evolution: dynamical ordering in sequence space. In: Pines D (ed) Emerging synthesis in science. Santa Fe Institute Studies in the Sciences of Complexity. Addison Wesley, Reading, MA, vol I, pp 25–69
Eigen M, McCaskill JS, Schuster P (1989) The molecular quasispecies. Adv Chem Phys 75:149–263
Eigen M, Schuster P (1979) The hypercycle: a principle of natural self-organization. Springer, Berlin
Fontana W, Konings DAM, Stadler PF, Schuster P (1993) Statistics of RNA secondary structure. Biopolymers 33:1389–1404
Fontana W, Schuster P (1987) A computer model for evolutionary optimization. Biophys Chem 26:123–147
French D, Laskov R, Scharff M (1989) The role of somatic hypermutation in generation of antibody diversity. Science 244:1152–1157
Hamilton WD, Axelrod R, Tanese R (1990) Sexual reproduction as ad adaptation to resist parasites (a review). Proc Natl Acad Sci USA 87:3566–3573
Hogeweg P, Hesper B (1984) Energy directed folding of RNA sequences. Nucleic Acids Res 12:67–74
Hogeweg P, Hesper B (1992) Evolutionary dynamics and the coding structure of sequences; multiple coding as a consequence of crossover and high mutation rates. Comput Chem 16:171–182
Holland JH (1975) Adaptation in natural and artificial systems. University of Michigan Press, Ann Arbor
Huynen MA (1993) Evolutionary dynamics and pattern generation in the sequence and secondary structure of RNA: a bioinformatic approach. PhD thesis, University of Utrecht
Huynen MA, Hogeweg P (1989) Genetic algorithms and information accumulation during the evolution of gene regulation. In: Schaffer JD (ed) Proceedings of the Third International Conference on Genetic Algorithms. Morgan Kaufmann, San Mateo, pp 225–230
Huynen MA, Konings DAM, Hogeweg P (1993) Multiple coding and the evolutionary properties of RNA secondary structure. J Theor Biol 165:251–267
Ikegami T, Kaneko K (1992) Evolution of host-parasitoid network through homeochaotic dynamics. CHAOS 2:397–407
Jaeger JA, Turner DH, Zuker M (1989) Improved predictions of secondary structures for RNA. Proc Natl Acad Sci USA 86:7706–7710
Kaneko K, Ikegami T (1992) Homeochaos: dynamic stability of a symbiotic network with population dynamics and evolving mutation rates. Physica D 56:406–429
Kauffman SA (1989) Adaptation on rugged fitness Landscapes. In: Stein DL (ed) Lectures in the sciences of complexity, vol 1. Addison Wesley, New York, pp 619–712
Kauffman SA, Johnsen S (1991) Co-evolution to the edge of chaos: coupled fitness landscape, poised states, and co-evolutionary avalanches. In: Langton CG, Taylor C, Farmer JD, Rasmussen S (eds) Artificial life II. Addison Wesley, New York, pp 325–369
Konings DAM (1989) Pattern analysis of RNA secondary structure: a bioinformatic approach. PhD thesis, Utrecht University
Konings DAM, Hogeweg P (1989) Pattern analysis of RNA secondary structure. J Mol Biol 207:597–614
Maynard-Smith J (1970) Natural selection and the concept of a protein space. Nature 255:563–566
Nowak MA, May RM, Anderson RM (1990) The evolutionary dynamics of HIV-1 quasispecies and the development of immunodeficiency disease. AIDS 4:1095–1103
Schuster P (1989) Optimization of RNA structure and properties. In: Perelson AS, Kauffman SA (eds) Molecular evolution on rugged landscapes: proteins, RNA and the immune system. Addison Wesley, Redwood City, pp 47–71
Weinberger E (1990) Correlated and uncorrelated fitness landscapes and how to tell the difference. Biol Cybern 63:325–336
Wright S (1932) The roles of mutation, inbreeding, crossbreeding and selection in evolution. Proceedings of the Sixth International Congress on Genetics 1, pp 356–366
Wright S (1967) Surfaces of selective value. Proc Natl Acad Sci U S A 58:165–172
Author information
Authors and Affiliations
Additional information
Correspondence to: M.A. Huynen
Rights and permissions
About this article
Cite this article
Huynen, M.A., Hogeweg, P. Pattern generation in molecular evolution: Exploitation of the variation in RNA landscapes. J Mol Evol 39, 71–79 (1994). https://doi.org/10.1007/BF00178251
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00178251