Abstract
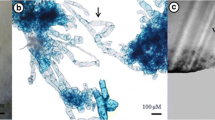
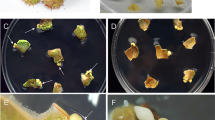
Adventive embryogenesis was successfully induced in cultures of zygotic and somatic embryos on MS medium supplemented with BA and NAA. A procedure has been proved successful for the in vitro multiplication of somatic embryos regenerated at low frequencies from filament and callus cultures. The occurrence and rate of adventive embryogenesis did not depend on the origin of the primary embryos (zygotic and somatic), but did depend on the developmental stage. Primary embryos are capable of embryogenesis in each of the different phases of embryogenesis, though the rate is different. BA concentrations of 22–44 μM increased the rate of adventive embryogenesis and accelerated the development of embryos. The highest proliferation rate (22–25x/5 weeks) was achieved at hormone concentrations of 44 μM BA and 5.4 μM NAA.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- BA:
-
benzyladenine
- CH:
-
casein hydrolysate
- CM:
-
coconut milk
- 2,4-D:
-
dichlorophenoxyacetic acid
- MS:
-
Murashige & Skoog medium
- WPM:
-
woody plant medium
- NAA:
-
1-naphtaleneacetic acid
References
Chalupa V (1990) Somatic embryogenesis and plant regeneration in Quercus petraea (Matt.) Liebl., Tilia platyphyllos Scop., and Aesculus hippocastanum L. Lestnictvi. Forestry 36: 599–604
Dameri R M, Caffaro L, Gastaldo P & Profumo P (1986) Callus formation and embryogenesis with leaf explants of Aesculus hippocastanum L. J. Plant Physiol. 126: 93–97
Durzan D J & Gupta P K (1987) Somatic embryogenesis and polyembryogenesis in Douglas-fir cell suspension culture. Plant Sci. 52: 229–235
Gamborg O L, Miller R A & Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cells. Exp. Cell Res. 50: 151–158
Gamborg O L, Davies B P & Stahlhut R W (1983) Cell division and differentiation in protoplasts from cell cultures of Glycine species and leaf tissues of soybean. Plant Cell Rep. 2: 213–215
Gingas V M & Lineberger R D (1989) Asexual embryogenesis and plant regeneration in Quercus. Plant Cell Tiss. Org. Cult. 17: 191–203
James J D, Passey A J & Deeming D C (1984) Adventitious embryogenesis and the in vitro culture of apple seed parts. J. Plant Physiol. 115: 217–229
Jörgensen J (1988) Embryogenesis in Quercus petraea and Fagus sylvatica. J. Plant Physiol. 132: 638–640
Jörgensen J (1989) Somatic embryogenesis in Aesculus hippocastanum L. by culture of filament callus. J. Plant Physiol. 135: 240–241
Merkle S A & Wiecko A T (1989) Regeneration of Robinia pseudoacacia via somatic embryogenesis. Can. J. For. Res. 19: 285–288
Muralidharan E M & Mascarenhas F M (1987) In vitro plantlet formation by organogenesis in E. camaldulensis and by somatic embryogenesis in Eucalyptus citriodora. Plant Cell Rep. 6: 256–259
Murashige T & Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant. 15: 473–497
Novak F J, Donini B & Owusa G (1986) Somatic embryogenesis and in vitro plant development of Cocoa (Theobroma cacao). Nuclear Techniques and In Vitro Culture For Plant Improvement (Proceedings), 443–449
Perez C, Fernandez B & Rodriquez R (1983) In vitro plantlet regeneration through asexual embryogenesis in cotyledonary segments of Corylus avellana L. Plant Cell Rep. 2: 226–228
Radojevic L, Vujieic R & Neskovic M (1975) Embryogenesis in tissue culture of Corylus avellana L. Z. Pflanzenphysiol. 77: 33–41
Radojevic L (1978) In vitro induction of androgenic plantlets in Aesculus hippocastanum L. Protoplasma 96: 369–374
Radojevic L (1988) Plant regeneration of Aesculus hippocastanum L. (Horse chestnut) through somatic embryogenesis. J. Plant Physiol. 132: 322–326
Radojevic L, Djordjevic N & Tucic B (1989) In vitro induction of pollen embryos and plantlets in Aesculus carnea Hayne through anther culture. Plant Cell Tiss. Org. Cult. 17: 21–26
Saito A (1980) In vitro differentiation of embryoid from somatic callus tissues in Aesculus. J. Jpn. For. Soc. 62: 308–310
Sellars R M, Southward G M & Phillips G C (1990) Adventitious somatic embryogenesis from cultured immature zygotic embryos of peanut and soybean. Crop. Sci. 30: 408–414
Tulecke W, Mcgranahan & Ahmadi H (1988) Regeneration by somatic embryogenesis of triploid plants from endosperm of walnut, (Juglans regia L. cv. Manregian). Plant Cell Rep. 7: 301–304
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kiss, J., Heszky, L.E., Kiss, E. et al. High efficiency adventive embryogenesis on somatic embryos of anther, filament and immature proembryo origin in horse-chestnut (Aesculus hippocastanum L.) tissue culture. Plant Cell Tiss Organ Cult 30, 59–64 (1992). https://doi.org/10.1007/BF00040001
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00040001