Abstract
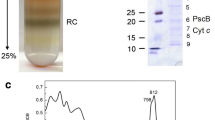
Chlorosome-depleted membranes and a reaction center complex with well-defined subunit composition were prepared from the green sulfur bacterium Chlorobium vibrioforme under anaerobic conditions. The reaction center complex contains a 15-kDa polypeptide with the N-terminal amino acid sequence MEPQLSRPETASNQVR/. This sequence is nearly identical to the N-terminus of the pscD gene product from Chlorobium limicola (Hager-Braun et al. (1995) Biochemistry 34: 9617–9624). In the presence of ferredoxin and ferredoxin:NADP+ oxidoreductase, the membranes and the isolated reaction center complex photoreduced NADP+ at rates of 333 and 110 μmol (mg bacteriochlorophyll a)−1 h−1, respectively. This shows that the isolated reaction center complex contains all the components essential for steady state electron transport. Midpoint potentials at pH 7.0 of 160 mV for cytochrome c 551 and of 245 mV for P840 were determined by redox titration. Antibodies against cytochrome c 551 inhibit NADP+ reduction while antibodies against the bacteriochlorophyll a-binding Fenna-Matthews-Olson protein do not.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- FMO protein:
-
Fenna-Matthews-Olson protein
- TMBZ:
-
3,3′,5,5′-tetramethylbenzidine
References
Amesz J and Knaff DB (1988) Molecular mechanisms of bacterial photosynthesis. In: Zehnder AJB (ed) Biology of Anaerobic Microorganisms, pp 113–178. Wiley, New York
Andersen B, Scheller HV and Møller BL (1992) The PSI-E subunit of Photosystem I binds ferredoxin:NADP+ oxidoreductase. FEBS Lett 311: 169–173
Bengis C and Nelson N (1977) Subunit structure of chloroplast Photosystem I reaction center. J Biol Chem 252: 4564–4569
Buchanan BB and Evans MCW (1969) Photoreduction of ferredoxin and its use in NAD(P)+ reduction by a subcellular preparation from the photosynthetic bacterium, Chlorobium thiosulfatophilum. Biochim Biophys Acta 180: 123–129
Büttner M, Xie D-L, Nelson H, Pinther W, Hauska G and Nelson N (1992) Photosynthetic reaction center genes in green sulfur bacteria and Photosystem I are related. Proc Natl Acad Sci USA 89: 8135–8139
Chitnis PR, Reiley PA, Miedel MC and Nelson N (1989) Structure and targeted mutagenesis of the gene encoding 8-kDa subunit of Photosystem I from the cyanobacterium Synechocystis sp. PCC 6803. J Biol Chem. 264: 18374–18380
Dutton PL (1978) Redox potentiometry: Determination of midpoint potentials of oxidation-reduction components of biological electron-transfer systems. Methods Enzymol 54: 411–435
Evans MCW and Buchanan BB (1965) Photoreduction af ferredoxin and its use in carbon dioxide fixation by a subcellular system from a photsynthetic bacterium. Proc Natl Acad Sci USA 53: 1420–1425
Feiler U, Nitschke W and Michel H (1992) Characterization of an improved reaction center preparation from the photosynthetic green sulfur bacterium Chlorobium containing the FeS centers FA and FB and a bound cytochrome subunit. Biochemistry 31: 2608–2614
Fling SP and Gregerson DS (1986) Peptide and molecular weight determinations by electrophoresis using a high-molarity Tris buffer system without urea. Anal Biochem 155: 83–88
Fowler CF, Nugent NA and Fuller RC (1971) The isolation and characterization of a photochemically active complex from Chloropseudomonas ethylica. Proc Natl Acad Sci USA 68: 2278–2282
Hager-Braun C, Xie D-L, Jarosch U, Herold E, Büttner M, Zimmermann R, Deutzmann R, Hauska G and Nelson N (1995) Stable photobleaching of P840 in Chlorobium reaction center preparations: Presence of the 42-kDa bacteriochlorophyll a protein and a 17-kDa polypeptide. Biochemistry 34: 9617–9624
Hurt EC and Hauska G (1984) Purification of membrane-bound cytochromes and a photoactive P840 protein complex of the green sulfur bacterium Chlorobium limicola f. thiosulfatophilum. FEBS Lett 168: 149–154
Illinger N, Xie D-L, Hauska G and Nelson N (1993) Identification of the subunit carrying FeS-centers A and B in the P840-reaction center preparation of Chlorobium limicola. Photosynth Res 38: 111–114
Kjær B, Jung Y-S, Yu L, Golbeck JH and Scheller HV (1994) Iron-sulfur centers in the photosynthetic reaction center complex from Chlorobium vibrioforme. Differences from and similarities to the iron-sulfur centers in Photosystem I. Photosynthe Res 41: 105–114
Kusumoto N, Inoue K, Nasu H and Sakurai H (1994) Preparation of a photoactive reaction center complex containing photoreducible Fe-S centers and photooxidizable cytochrome c from the green sulfur bacterium Chlorobium tepidum. Plant Cell Physiol 35: 17–25
Li N, Zhao J, Warren PV, Warden J, Bryant DA and Golbeck JH (1991) PsaD is required for the stable binding of PsaC to the Photosystem I core of Synechococcus sp PCC 6301. Biochemistry 30: 7863–7872
Miller M, Liu X, Snyder SW, Thurnauer MC and Biggins J (1992) Photosynthetic electron transfer reactions in the green sulfur bacterium Chlorobium vibrioforme: Evidence for the functional involvement of iron-sulfur redox centers on the acceptor side of the reaction center. Biochemistry 31: 4354–4363
Meyer TE (1994) Purification and properties of cytochrome c-555 from phototrophic green sulfur bacteria. Meth Enzymol 243: 426–435
Nitschke W, Feiler U and Rutherford AW (1990) Photosynthetic reaction center of green sulfur bacteria studied by EPR. Biochemistry 29: 3834–3842
Oh-oka H, Kakutani S, Matsubara H, Malkin R and Itoh S (1993) Isolation of the photoactive reaction center complex that contains three types of Fe-S centers and a cytochrome c subunit from the green sulfur bacterium Chlorobium limicola f. thiosulfatophilum, strain Larsen. Plant Cell Physiol 34: 93–101
Okkels JS, Kjær B, Hansson Ö, Svendsen I, Møller BL and Scheller HV (1992) A membrane bound monoheme cytochrome c 551 of a novel type is the immediate electron donor to P840 of the Chlorobium vibrioforme photosynthetic reaction center complex. J Biol Chem 267: 21139–21145
Okumura N, Shimada K and Matsuura K (1994) Photo-oxidation of membrane-bound and soluble cytochrome c in the green sulfur bacterium Chlorobium tepidum. Photosynthe Res 41: 125–134
Olson JM, Giddings TH and Shaw EK (1976a) An enriched reaction center preparation from green photosynthetic bacteria. Biochim Biophys Acta 449: 197–208
Olson JM, Prince RC and Brune DC (1976b) Reaction-center complexes from green bacteria. In: Olson JM and Hind G (eds) Chlorophyll-Proteins, Reaction Centers and Photosynthetic Membranes, pp 238–246. Brookhaven National Laboratory, Upton, NY
Prince RC and Olson JM (1976) Some thermodynamic and kinetic properties of the primary photochemical reactants in a complex from a green photosynthetic bacterium. Biochim Biophys Acta 423: 357–362
Rousseau F, Sétif P and Lagoutte B (1993) Evidence for the involvement of PS I-E subunit in the reduction of ferredoxin by Photosystem I. EMBO J 12: 1755–1765
Serrano A and Rivas J (1982) Purification of ferredoxin-NADP+ oxidoreductase from cyanobacteria by affinity chromatography on 2′-5′-ADP-Sepharose 4B. Anal Biochem 126: 109–115
Swarthoff T and Amesz J (1979) Photochemically active pigment-protein complexes from the green photosynthetic bacterium Prosthecochloris aestuarii. Biochim Biophys Acta 548: 427–432
Swarthoff T, Gast H, Hoff AJ and Amesz J (1981a) An optical and ESR investigation on the acceptor side of the reaction center of the green photosynthetic bacterium Prosthecochloris aestuarii. FEBS Lett 130: 93–98
Swarthoff T, van derVeek-Horsley KM and Amesz J (1981b) The primary charge separation, cytochrome oxidation and triplet formation in preparations from the green photosynthetic bacterium Prosthecochloris aestuarii. Biochim Biophys Acta 635: 1–12
Thomas PE, Ryan D and Levin W (1976) An improved staining procedure for the detection of the peroxidase activity of cytochrome P-450 on sodium dodecyl sulfate polyacrylamide gels. Anal Biochem 75: 168–176
Vasmel H, Swarthoff T, Kramer HJM and Amesz J (1983) Isolation and properties of a pigment-protein complex associated with the reaction center of the green photosynthetic sulfur bacterium Prosthecochloris aestuarii. Biochim Biophys Acta 725: 361–367.
Zannoni D and Daldal F (1993) The role of c-type cytochromes in catalyzing oxidative and photosynthetic electron transport in the dual functional plasmamembrane of facultative phototrophs. Arch Microbiol 160: 413–423
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kjær, B., Scheller, H.V. An isolated reaction center complex from the green sulfur bacterium Chlorobium vibrioforme can photoreduce ferredoxin at high rates. Photosynth Res 47, 33–39 (1996). https://doi.org/10.1007/BF00017751
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00017751