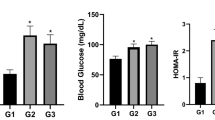
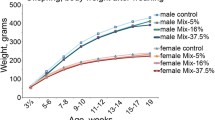
Abstract
Pesticide exposure may induce biochemical alterations including oxidative stress and lipid peroxidation. However, in the context of developmental origin of health and disease, putative trans-generational effect of exposure to pesticides are insufficiently studied. We therefore aimed to evaluate the biochemical effect of gestational exposure to four pesticides on female Wistar rats and their offspring at adult age. We studied 30 female nulliparous Wistar rats divided into 5 equal groups. Group 1 served as the control group and received distilled water while group 2, 3, 4 and 5 received orally pesticide 1 (imidacloprid), pesticide 2 (chlorpyrifos), pesticide 3 (imidacloprid + lambda cyhalothrin) and pesticide 4 (oxamyl) respectively once daily throughout gestation at a dose equivalent to 1/10 lethal dose 50. The mothers were followed up until one month post gestation. The offspring were followed up from birth until adult age (12 weeks). In all animals at each time point we evaluated malondialdehyde (MDA), oxidative stress and liver function enzymes. There was similar variation of total body weight in all the groups during and after gestation. However, Female Wistar rats of the exposed groups had significant alterations in liver SOD (−30.8% to +64.1%), catalase (−38.8% to −85.7%) and GSH (−29.2% to −86.5%) and; kidney catalase (> 100%), GSH (> 100%). Moreover, MDA, alanine transaminase (ALT) and aspartate transaminase (AST) levels were significantly higher in pesticide exposed rats compared to the control group. Similar alterations in antioxidant enzymes, MDA and liver function enzymes were observed in offspring of treated rats evidenced at weaning and persisting until adult age. Exposure to pesticides causes oxidative stress and lipid peroxidation in exposed female Wistar rats and their offspring. The persistence in offspring at adult age suggests transgenerational adverse effects.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- ALT:
-
Alanine Transaminase
- AST:
-
Aspartate Transaminase
- BSA:
-
Bovine Serum Albumin
- EC:
-
Emulsifiable concentrate
- EDTA:
-
Ethylenediaminetetraacetic acid
- GPx:
-
Glutathione peroxidase
- GR:
-
Glutathione reductase
- MDA:
-
Malondialdehyde
- NADP:
-
Nicotinamide adenine dinucleotide phosphate
- nAChR:
-
Nicotinic acetylcholine receptors
- OD:
-
Optical Density
- GSSG:
-
Oxidized Glutathione
- GSH:
-
Reduced glutathione
- SL:
-
Soluble liquid
- SEM:
-
Standard error of mean
- SOD:
-
Superoxide dismutase
References
Ming, Y., Jeremy, B., Jonathan, W.M. and Ambikaipakan, S. (2013) Occupational pesticide exposures and respiratory health. Int. J. Environ. Res. Public Health, 10, 6442–6471.
Shin, H.J., Cho, H.G., Park, C.K., Park, K.H. and Lim, H.B. (2017) Comparative in vitro biological toxicity of four kinds of air pollution particles. Toxicol. Res., 33, 305–313.
Food and Agriculture Organization of the United Nations (FAO). Database on Pesticides Consumption [accessed 2013 Nov 15]. Available from: http://www.fao.org/waicent/FAO-INFO/economic/pesticid.htm/.
Clark, J.M. and Symington S.B. (2007) Pyrethroid action on calcium channels. Neurotoxicological implications. Invert. Neurosci., 7, 3–16.
Nagata, K., Song, J.H., Shono, T. and Narahashi T. (1998) Modulation of the neuronal nicotinic acetylcholine receptorchannel by the nitromethylene heterocycle imidacloprid. J. Pharmacol. Exp. Ther., 285, 731–738.
Abdallah, F., Fetoui, H., Fakhfakh, F. and Keskes, L. (2012) Caffeic acid and quercetin protect erythrocytes against the oxidative stress and the genotoxic effects of lambda-cyhalothrin in vitro. Hum. Exp. Toxicol., 31, 92–100.
El-Demerdash, F.M. (2007) Lambda-cyhalothrin-induced changes in oxidative stress biomarkers in rabbit erythrocytes and alleviation effect of some antioxidants. Toxicol. In Vitro, 21, 392–397.
Fetoui, H., Garoui, E.M., Makni-ayadi, F. and Zeghal, N. (2008) Oxidative stress induced by lambda-cyhalothrin in rat erythrocytes and brain: attenuation by vitamin C. Environ. Toxicol. Pharmacol., 26, 225–231.
Fetoui, H., Garoui, E.M. and Zeghal, E. (2009) Lambdacyhalothrin-induced biochemical and histopathological changes in the liver of rats: ameliorative effect of ascorbic acid. Exp. Toxicol. Pathol., 61, 189–196.
Fetoui, H., Makni, M., Garouiel, M. and Zeghal, N. (2010) Toxic effects of lambda-cyhalothrin, a synthetic pyrethroid pesticide, on the rat kidney: involvement of oxidative stress and protective role of ascorbic acid. Exp. Toxicol. Pathol., 62, 593–599.
Mohany, M., Badr, G., Refaat, I. and El-Feki, M. (2011) Immunological and histological effects of exposure to imidacloprid insecticide in male albino rats. Afr. J. Pharm. Pharmacol., 5, 2106–2114.
Tuzmen, N., Candan, N., Kays, E. and Demiryas, N. (2008) Biochemical effects of chlorpyrifos and deltamethrin on altered antioxidativedefense mechanisms and lipid peroxidation in rat liver. Cell Biochem. Funct., 26, 119–124.
El-Demerdash, F.M. (2011) Oxidative stress and hepatotoxicity induced by synthetic pyrethroids-organophosphate insecticides mixture in rat. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev., 29, 145–158.
El-Demerdash, F.M. (2012) Cytotoxic effect of fenitrothion and lambda-cyhalothrin mixture on lipid peroxidation and antioxidant defense system in rat kidney. J. Environ. Sci. Health B, 47, 262–268.
Tukhtaev, K., Tulemetov, S., Zokirova, N. and Tukhtaev, N. (2012) Effect of long term exposure of low doses of lambdacyhalothrin on the level of lipid peroxidation and antioxidant enzymes of the pregnant rats and their offspring. Med. Health Sci. J., 13, 93–98.
Marklund, S. and Marklund, G. (1974) Involvement of the superoxide anion radical in the autooxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem., 47, 469–474.
Aebi, H. (1984) Catalase in vitro. Enzymology, 105, 121–126.
Wendel, A. (1980) Enzymatic Bases of Detoxification. Academic press. 1, 333.
Moron, M.S., Depierre, J.W. and Mennervik, B. (1979) Levels of glutathione, glutathione reductase and glutathione Stransferase activities in rat lung and liver. Biochim. Biophys. Acta, 582, 67–78.
Mavis, R.D. and Stellwagen, E. (1968) Purification and subunit structure of glutathione reductase from Bakers’ yeast. J. Biol. Chem., 243, 809–814.
Ohkawa, H., Ohishi, N. and Yagi, K. (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem., 95, 351–358.
Lowry, O.H., Rosebrough, N.J., Farr, A.L. and Randal, R.J. (1951) Protein measurement with the folin phenol reagent. J. Biol. Chem., 193, 265–275.
Lobin, K.K., Jaunky, V.C. and Ramesh, V. (2017) A review of pesticide use in EU and African countries and associated polices in Proceedings of 120th The IIER International Conference (2017 Sep 15–16). Port Louis, Mauritius.
Kimengsi, J.N. and Muluh, N.G. (2013) A comparative assessment of the effect of climate variations on the crops of the Cameroon development corporation (CDC): adaptation options. Env. Nat. Res. Research, 3, 144–156.
Brice, K.N., Patricia, A.F., Norbert, N.T. and Mpoame, M. (2017) Environmental and human health assessment in relation to pesticide use by local farmers and the Cameroon development corporation (cdc), Fako division, South-west cameroon. Eur. Sci. J., 13, 454–473.
Gesesew, H.A., Woldemichael, K., Massa, D. and Mwanri, L. (2016) farmers knowledge, attitudes, practices and health problems associated with pesticide use in rural irrigation villages, Southwest Ethiopia. PLoS ONE, 11, e0162527.
Damalas, C.A. and Koutroubas S.D. (2016) Farmers’ exposure to pesticides: toxicity types and ways of prevention. Toxics, 4, 1.
Balani, T., Agrawal, S. and Thaker, A.M. (2011) Hematological and biochemical changes due to short-term oral administration of imidacloprid. Toxicol. Int., 18, 2–4.
El-Gendy, K.S., Aly, N.M., Mahmoud, F.H., Kenawy, A. and El-Sebae, A.K. (2010) The role of vitamin C as antioxidant in protection of oxidative stress induced by imidacloprid. Food Chem. Toxicol., 48, 215–221.
Kapoor, U., Srivastava, M.K., Bhardwaj, S. and Srivastava, L.P. (2010) Effect of imidacloprid on antioxidant enzymes and lipid peroxidation in female rats to derive its No Observed Effect Level (NOEL). J. Toxicol. Sci., 35, 577–581.
Ahmed, M.M. and Zaki, N.I. (2009) Assessment the ameliorative effect of pomegranate and rutin on chlorpyrifos-ethylinduced oxidative stress in rats. Nature and Science, 7, 49–61.
Baş, H. and Kalender, Y. (2011) Chlorpyrifos induced cardiotoxicity in rats and the protective role of quercetin and catechin. GUJS, 24, 387–395.
Gultekin, F., Delibas, N., Yasar, S. and Kiline, I. (2001) In vivo changes in antioxidant systems and protective role of melatonin and a combination of vitamin C and vitamin E on oxidative damage in erythrocytes induced by chlorpyrifosethyl in rats. Arch. Toxicol., 75, 88–96.
Oncu, M., Gultekin, F., Karaöz, E., Altuntas, I. and Delibas, N. (2002) Nephrotoxicity in rats induced by chlorpyrifosethyl and ameliorating effects of antioxidants. Hum. Exp. Toxicol., 21, 223–230.
Verma, R.S. and Srivastava, N. (2001) Chlorpyrifos induced alterations in levels of thiobarbituric acid reactive substances and glutathione in rat brain. Indian J. Exp. Biol., 39, 174–177.
Gopinath, G., Uvarajan, S. and Tamizselvi, A. (2014) Chlorpyrifos-induced oxidative stress and tissue damage in the liver of Swiss albino mice: the protective antioxidative role of root extract of withania somnifera. IJIRST, 1, 100–104.
Soujanya, S., Lakshman, M., Anand, K.A. and Gopala, R.A. (2013) Evaluation of the protective role of vitamin C in imidacloprid-induced hepatotoxicity in male albino rats. J. Nat. Sci. Biol. Med., 4, 63–67.
Verma, R.S., Mehta, A. and Srivastava, N. (2007) In vivo chlorpyrifos induced oxidative stress: attenuation by antioxidant vitamins. Pestic. Biochem. Physiol., 88, 191–196.
Trevisan, M., Browne, R., Ram, M., Muti, P., Freudenheim, J., Carosella, A.M. and Armstrong, D. (2001) Correlates of markers of oxidative status in the general population. Am. J. Epidemiol., 154, 348–356.
Author information
Authors and Affiliations
Corresponding author
Additional information
This is an Open-Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Rights and permissions
This is an Open-Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Ndonwi, E.N., Atogho-Tiedeu, B., Lontchi-Yimagou, E. et al. Gestational Exposure to Pesticides Induces Oxidative Stress and Lipid Peroxidation in Offspring that Persist at Adult Age in an Animal Model. Toxicol Res. 35, 241–248 (2019). https://doi.org/10.5487/TR.2019.35.3.241
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.5487/TR.2019.35.3.241