Abstract
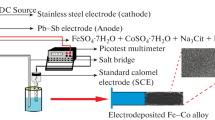
Nanocrystalline iron coatings were electrodeposited at six different current densities, from 1 to 25 A dm–2 from a bath containing iron(II) sulfate at 60 and 80°C. The impact of the current density on the bath deterioration was investigated. The bath color changed from emerald green (485 nm maximum absorption wavelength, λmax) to turbid yellow (λmax = 470 nm) with some orange precipitates after electrodeposition up to 10 A dm–2. Further electrodeposition up to 25 A dm–2 changed the bath colour to dark brown (λmax = 435 nm). Ultraviolet-Visible spectra were recorded to verify the bath deterioration, which, in turn, prevented the coating growth. The color change and also the shift of the maximum absorption wavelength were discussed in terms of the crystal field theory and also of the anodic oxidation of the hexaaquairon(II) ion to the hexaaquairon(III) ion on the anode surface. The Pourbaix diagram for iron was used to interpret these results and the instability of the bath which was utilized here for electrodeposition of nanocrystalline iron coatings. Addition of saccharin also led to the bath instability. The influence of the bath instability on the average grain size, appearance, surface morphology and thickness of the coatings was discussed. The results proved that the iron(II) sulfate solution is an unstable bath for electrodeposition of nanocrystalline iron coatings.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Harty, S.F., McGeough, J.A., and Tulloch, R.M., Surf. Technol., 1981, vol. 12, pp. 39–55.
Watanabe, T. Nano Plating—Microstructure Formation Theory of Plated Films and a Database of Plated Films, Amsterdam: Elsevier, 2004.
Kasper, C.H., J. Res. Natl. Bur. Stand. (U.S.), 1937, vol. 18, pp. 535–541.
Zakirov, S.Z., Met. Sci. Heat Treat., 1975, vol. 17, pp. 709–711.
Moravej, M., Prima, F., Fiset, M., and Mantovani, D., Acta Biomater., 2010, vol. 6, pp. 1726–1735.
Moravej, M., Purnama, A., Fiset, M., Couet, J., and Mantovani, D., Acta Biomater., 2010, vol. 6, pp. 1843–1851.
Gow, K.V., Iyer, S.P., Wu, H.H., Castelliz, K.M., et al., Surf. Technol., 1979, vol. 8, pp. 333–346.
Jartych, E., Zurawicz, J.K., Maczka, E., and Borc, J., Mater. Chem. Phys., 2001, vol. 72, pp. 356–359.
Koza, J., Uhlemann, M., Gebert, A., and Schultz, L., J. Solid State Electrochem., 2008, vol. 12, pp. 181–192.
Schlesinger, M. and Paunovic, M., Modern Electroplating, New York: Wiley, 2011.
Inoue, K., Nakata, T., and Watanabe, T., Mater. Trans., 2002, vol. 43, pp. 1318–1324.
Lai, S.H., McGeough, J.A., and Lau, P., J. Mech. Work. Technol., 1978, vol. 1, pp. 231–243.
Diaz, S.L., Calderón, J.A., Barcia, O.E., and Mattos, O.R., Electrochim. Acta, 2008, vol. 53, pp. 7426–7435.
Afshari, V. and Dehghanian, C., J. Appl. Electrochem., 2010, vol. 40, pp. 1949–1956.
Afshari, V. and Dehghanian, C., Anti-Corros. Methods Mater., 2010, vol. 57, pp. 142–147. https://doi.org/10.1108/00035591011040100.
Afshari, V. and Dehghanian, C., Int. J. Mater. Res., 2010, vol. 101, pp. 366–371.
Cotton, F.A., Wilkinson, G., Murillo, C.A., and Bochmann, M., Advanced Inorganic Chemistry, New York: Wiley, 1999.
Ballhausen, C.J. and Weiner, M.A., J. Electrochem. Soc., 1963, vol. 110, art. ID 97C.
Green, T.A., Gold Bull., 2007, vol. 40, no. 2, pp. 105–114.
Green, T.A. and Roy, S., J. Electrochem. Soc., 2006, vol. 153, pp. 157–163.
Lowinsohn, D., Alipázaga, M.V., Coichev, N., and Bertotti, M., Electrochim. Acta, 2004, vol. 49, pp. 1761–1766.
Tjandrawan, V. and Nicol, M.J., Hydrometallurgy, 2013, vol. 131, pp. 81–88.
Su, C.W., Yang, W., Guo, J.M., and Zhang, Y.J., Vacuum, 2012, vol. 86, pp. 2095–2101.
Zarpellon, J., Jurca, H.F., Klein, J.J., Schreiner, W.H., et al., Electrochim. Acta, 2007, vol. 53, pp. 2002–2008.
Evreinova, N.V., Shoshina, I.A., Naraev, V.N., and Tikhonov, K.I., Russ. J. Appl. Chem., 2008, vol. 81, pp. 1180–1183.
Najafi Sayar, P. and Bahrololoom, M.E., Trans. IMF, 2009, vol. 87, pp. 246–253.
Najafi Sayar, P. and Bahrololoom, M.E., J. Appl. Electrochem., 2009, vol. 39, pp. 2489–2496.
Rashidi, A.M. and Amadeh, A., Surf. Coat. Technol., 2009, vol. 204, pp. 353–358.
Shirazi, S.H., Bahrololoom, M.E., and Shariat, M.H., Surf. Eng. Appl. Electrochem., 2016, vol. 52, pp. 434–442.
Williamson, G.K. and Hall, W.H., Acta Metall., 1953, vol. 1, pp. 22–31.
Delahay, P., Pourbaix, M., and van Rysselberghe, P., J. Chem. Educ., 1950, vol. 27, p. 683.
Al Tanvir, T., Hossain, M.E., Al Mamun, M., and Ehsan, M.Q., J. Bangladesh Acad. Sci., 2014, vol. 37, pp. 195–203.
Haider, S.Z., Malik, K.J., and Ahmed, K.J., J. Bangladesh Acad. Sci., 1981, vol. 5, pp. 81–90.
Jovanovski, G., Croat. Chem. Acta, 2000, vol. 73, pp. 843–868.
Baran, E.J. and Yilmaz, V.T., Coord. Chem. Rev., 2006, vol. 250, pp. 1980–1999.
Earnshaw, A. and Harrington, T.J., Chemistry of the Transition Elements (Oxford Chemistry), Oxford: Oxford Univ. Press, 1973.
Tabakovic, I., Riemer, S., Tabakovic, K., Sun, M., et al., J. Electrochem. Soc., 2006, vol. 153, pp. 586–593.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
About this article
Cite this article
Bahrololoomi, A., Bahrololoom, M.E. Instability of Ferrous Sulfate Bath for Electrodeposition of Nanocrystalline Iron Coating. Surf. Engin. Appl.Electrochem. 54, 562–576 (2018). https://doi.org/10.3103/S1068375518060029
Received:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S1068375518060029