Abstract
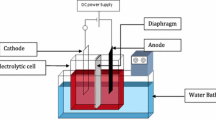
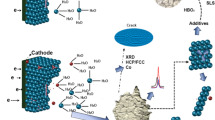
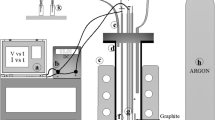
Cobalt is an essential engineering metal among the less abundant metals found in the earth crust with wide range of applications. However due to the scarcity of resources, it is required to produce the metal in a cost effective manner to meet the supply demands. Cobalt is produced by electrodeposition from chloride or sulphate media, the latter being a widely used method. The use of additives in the electrolytic bath during electrodeposition from sulphate solutions can not only solve issues related to current efficiencies, but also result in production of uniform, smooth and bright deposits of the metal. The present work investigates the effect of Tween 80 a non-ionic additive, on the electrodeposition of cobalt. The concentration of the additive Tween 80 was varied over a range of 1–50 mg/L to evaluate the changes in current efficiency, specific energy consumption and quality of electrodeposited cobalt metal. The results indicated that, apart from increasing the current efficiency, the additive produced bright cobalt deposits. A maximum current efficiency of 98.1% was achieved with 20 mg/L Tween 80 in the electrolytic bath. X-ray diffraction studies have revealed that <100> direction is the most preferred orientation of crystal growth during cobalt electrodeposition. However it is shifted to <110> at higher concentrations (50 mg/L) of the additive. Scanning electron micrographs indicate that smooth, compact and uniform deposits of cobalt are obtained at 20 mg/L beyond which there is deterioration in the quality of deposits. Cyclic voltammetric studies indicated polarization of the cathode in the presence of Tween 80. This behaviour was also reflected in the decrease of the rate of electron transfer as evident from exchange current density (i 0) values.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Davis, J.R., Uses of Cobalt in: ASM Specialty Handbook: Nickel, Cobalt, and Their Alloys, 2000.
Verney, L.R., Overview of Cobalt Supply and Production. Extractive Metallurgy of Nickel and Cobalt, The Metallurgical Society, 1988.
Kongstein, O.E., Haarberg, G.M., and Thonstad, J., J. Appl. Electrochem., 2007, vol. 37, p. 669.
Mulaudzi, N. and Kotze, M., The Southern African Institute of Mining and Metallurgy Base Metals Conference, 2013. p. 1.
Franklin, T.C., Surf. Coatings Technol., 1987, vol. 30, no. 4, p. 415.
Oniciu, L. and Muresan, L., J. Appl. Electrochem., 1991, vol. 21, no. 7, p. 565.
Tripathy, B.C., Singh, P., and Muir, D.M., Metallurg. Mater. Trans. B, 2001, vol. 32, no. 3, p. 395.
Das, S.C. and Subbaiah, T., J. Appl. Electrochem., 1987, vol. 17, no. 4, p. 675.
Jeffery, M.I., Choo, W.L., and Breuer, P.L., Miner. Eng., 2000, vol. 13, no. 12, p. 1231.
Renner, R.F. and Liddell, K.C., J. Appl. Electrochem., 2002, vol. 32, no. 6, p. 621.
Tripathy, B.C. and Kotze, M., AU Patent 2012101517 A4, Australia Patent Office, 2012.
Sekar, R., Inst. Mater. Finish., 2015, vol. 93, no. 1, p. 44.
Span and Tween, 2015, Croda Europe, Limited. Retrieved from chemagentru/component/flexicontent/download/849/968/19
Wen, Ten-chin, Wei, Ming-Gwo, and Lin, Kwang-Lung, J. Electrochem. Soc., 1990, vol. 137, no. 9, p. 2700.
Shvets, T.M., Melnichenko, Z.M., Vasilenko, V.P., Ivanova, L.Yu., and Natanson, E.M., Sov. Powder Metallurgy Metal Ceram., 1972, vol. 11, pp. 179–183.
Robinson, D.J. and O’Keefe, T.J., J. Appl. Electrochem., 1976, vol. 6, no. 1, p. 1.
Tripathy, B.C., Das, S.C., Singh, P., and Hefter, G.T., J. Appl. Electrochem., 1997, vol. 27, no. 6, p. 673.
Reddy, A.K.N., J. Electroanal. Chem., 1963, vol. 6, no. 2, p. 141.
Peek, Edgar, Akre, Torjus, and Asselin, Edouard, J. Met., 2009, vol. 61, no. 10, p. 43.
Abe, K. and Shimizu, F., J. Met., 1997, vol. 49, no. 12, p. 42.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
About this article
Cite this article
Patnaik, P., Tripathy, B.C., Bhattacharya, I.N. et al. Effect of Tween 80 on electrochemical deposition of cobalt from sulphate solutions. Russ. J. Non-ferrous Metals 57, 331–337 (2016). https://doi.org/10.3103/S1067821216040076
Received:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S1067821216040076