Abstract
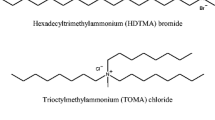
In this study surfactant-modified natural bentonite was examined for the adsorption of bisphenol A [2,2-bis(4-hydroxyphenyl)propane; BPA] from aqueous solutions. Batch experiments were performed to investigate the adsorption kinetics, equilibrium and thermodynamics between the adsorbent surfaces and BPA. At acidic pH conditions, removal of BPA was increased due to a neutral molecule form of BPA and hydrophobic surface created by the loaded surfactant molecules. The adsorption of BPA on modified bentonite was enhanced when the ionic strength was increased. The adsorption behavior of BPA onto surfactant-modified bentonite followed the pseudo-second order kinetic model. Langmuir isotherm provided the best fit for adsorption. The adsorption of BPA on modified bentonite is more favorable at lower temperature. According to the experimental results, a physical adsorption is responsible mechanisms for adsorption of BPA on modified bentonite.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Campbell, C.G., Borglin, S.E., Green, F.B., et al., Chemosphere, 2006, vol. 65 (8), pp. 1265–1280.
Park, Y., Sun, Z., Ayoko, G.D., and Frost, R.L., Chemosphere, 2014, vol. 107, pp. 249–256.
Liu, G., Ma, J., Li, X., and Qin, Q., J. Hazard. Mater., 2009, vol. 164, pp. 1275–1280.
Ipek, I., Yüksel, S., Kabay, N., and Yüksel, M., J. Chem. Technol. and Biotechnol., 2014, vol. 89 (6), pp. 835–840.
Zheng, S., Sun, Z., Yuri, Park, et al., Chem. Eng. J., 2013, vol. 234, pp. 416–422.
Laatikainen, K., Laatikainen, M., Bryjak, M., et. al., Separ. Sci. and Technol., 2014, vol. 49 (5), pp. 763–772.
Gök, Ö., Özcan, S., and Özcan, A., Appl. Surface Sci., 2010, vol. 256 (17), pp. 5439–5443.
Zhao, J., Zhu, C., Lu, J., et al., Electrochim. Acta, 2014, vol. 118 (1), pp. 169–175.
Kim, Y.H., Lee, B., Choo, K.H., and Choi, S.J., Micropor. and Mesopor. Mater., 2011, vol. 138, pp. 184–190.
Dong, L., Wu, D., Chen, X., and Lin, Y., J. Colloid and Interface Sci., 2010, vol. 348 (2), pp. 585–590.
Tsai, W.T., Lai, C.W., and Su, T.Y., J. Hazard. Mater., 2006, vol. 134, pp. 169–175.
Namasivayam, C. and Sumithra, S., Clean Technol. and Environ. Policy, 2007, vol. 9 (3), pp. 215–223.
Plazinski, W., Rudzinski, W., and Plazinska, A., Adv. Colloid and Interface Sci., 2009, vol. 152 152, pp. 2–13.
Qiu, F., Feng, M., Wei, Z., et al., J. Appl. Polymer Sci., 2015, vol. 133 (9), pp. 43066.
Li, H., Zhang, D., Han, X., and Xing, B., Chemosphere, 2014, vol. 95, pp. 150–155.
Wang, H.L., Fei, Z.H., Chen, J.L., et al., J. Environ. Sci., 2007, vol. 19, pp. 1298–1304.
Yang, Q., Gao, M., Luo, Z., and Yang, S., Chem. Eng. J., 2016, vol. 285, pp. 27–38.
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the authors in English.
About this article
Cite this article
Genc, N., Durna, E. & Kilicoglu, O. Removal of Bisphenol from Aqueous Solution by Surfactant-Modified Bentonite. J. Water Chem. Technol. 41, 236–241 (2019). https://doi.org/10.3103/S1063455X19040052
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S1063455X19040052