Abstract
Objectives and Design
Knowledge of which and how many species are part of an ecosystem is fundamental to understanding the diversity of organisms, including parasitic organisms that vary widely among host populations. This study describes the composition, infection patterns, and similarity in composition of helminths associated with P. albifrons, P. cicada, and P. cuvieri.
Materials and Methods
Four samplings were carried out in two caatinga areas in the southern region of Ceará State, Brazil. The specimens were collected by active search, necropsied, and surveyed for helminths. The infection patterns for all helminth species found were estimated through prevalence, mean abundance, and mean intensity of infection.
Results
A total of 242 helminth specimens were collected from 264 hosts—100 Physalaemus albifrons with overall prevalence of 20%, 93 Physalaemus cicada with overall prevalence of 27%, and 71 Physalaemus cuvieri with overall prevalence of 15%. Ten parasite taxa with a mean of one–three parasite species per host were identified. The nematode Raillietnema spectans was present in the three host species and had the highest prevalence.
Conclusions
The results of this study represent 38.4% of known parasites for the species of the subfamily Leiuperinae, and 52.6% for the species of Physalaemus. Four parasite taxa (Oswaldocruzia cf. mazzai, Raillietnema spectans, Schrankiana schranki, and Cylindrotaenia americana) are new records for the host species studied, contributing to the knowledge of the parasitic interactions of amphibians of the genus Physalaemus.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Species inventories are the base for scientific studies since the knowledge of which and how many species are part of an ecosystem is fundamental to understanding the diversity and function of the organisms. The parasitism is part of this diversity, being the most common life strategy in the planet [69]. Parasites are diverse organisms that are an integral part of nature, but were neglected for a long time in biodiversity inventories [32, 69].
The helminth fauna associated with amphibians is rich and diverse. The latest checklist of helminths of amphibians in South America state that only 8% of all amphibian species have had their helminth fauna surveyed, in which representatives of the Hylidae and Leptodactylidae are the most studied hosts [25].
Despite the efforts to survey the helminth fauna of the Neotropical region, many areas and hosts remain neglected regarding the study of helminths. The composition of parasite communities can vary significantly among host populations of the same species [11, 70]. This way, data on occurrence and distribution of parasite species broaden the database used to understand species richness patterns of helminth associated with amphibians [26].
According to Campião et al. [25], anurans have patterns of helminth parasite communities with wide distribution, high diversity, and low host specificity. These communities are influenced by several factors, like host size, sex, diet, infection site, species, and behavior [13, 26, 42]. Besides, the characteristics of host habitats are a key factor for the probability of parasite colonization [41].
Leptodactylid frogs show rich parasite communities, with wide taxonomic diversity, composed mainly of nematodes [27, 74, 77, 84, 85]. Most of the records are adult nematodes parasitizing the gastrointestinal tract of the hosts, with members of the Cosmocercidae, Kathlaniidae, Molineidae, Physalopteridae, and pulmonary Rhabdiasidae among the most recorded helminths [25, 26].
The genus Physalaemus [37] is characterized as a heterogeneous taxon [66]. From the 42 species recorded in Brazil [79], only ten had their helminth fauna surveyed: Physalaemus albonotatus [24, 46, 52], Physalaemus biligonigerus [49, 88], Physalaemus cuvieri [2, 31, 60, 75, 85], Physalaemus lopesi [8], Physalaemus marmoratus [89], Physalaemus nattereri [31], Physalaemus olfersii [85], Physalaemus santafecinus [45, 52], Physalaemus signifer [36, 88], and Physalaemus soaresi [35, 88].
Species of Physalaemus have wide geographical distribution in the Neotropical region, from Mexico to South America [66]. In Brazil, Physalaemus albifrons (Spix 1824) can be found in open areas from Maranhão State to the south of Minas Gerais State [5]; Physalaemus cicada [12] inhabit open areas of Caatinga, Cerrado, and domains of Atlantic forest [21, 62, 81, 82]; and P. cuvieri (Fitzinger 1826) is widely distributed in the South, Southeast, Northeast, and Central West, besides some areas in Uruguay, Paraguay, and Argentina [39, 50].
The aim of this study is to describe the composition, infection patterns, and similarity in composition of helminths associated with P. albifrons, P. cicada, and P. cuvieri from a Caatinga area in the south of Ceará State, Brazil.
Materials and Methods
This study was carried out in two sampling sites, ‘Sítio Pedra Preta’ (06°46′57.7″S, 39°33′11.2″W) and ‘Sítio Várzea’ (06°51′55.7″S, 39°32′08.0″W), both located at the Municipality of Farias Brito, Ceará State, Brazil. The sites have vegetation typical of Caatinga, with rainy season from January to April and mean precipitation of 896.5 mm3 [55]. The samplings were performed in temporary ponds on January 20th and 27th, and March 4th and 28th, 2017, with a total of 48 sampling hours.
The specimens were collected by visual search [10], individualized in plastic containers, and euthanized with lethal injection of Sodium thiopental [29]. Afterwards, all host specimens were weighed and had their snout-vent length (SVL) taken with a digital caliper Mitutoyo® (0.01 mm of precision). Host samples were fixed in 10% formalin according to Franco and Salomão [38], preserved in 70% ethanol and deposited at the Herpetological Collection of the Universidade Regional do Cariri (URCA-H), Crato, Ceará (URCA-H 12749-13003).
For parasitological examination, the hosts were necropsied while fresh, and the organs like gastrointestinal tract, lungs, liver, and kidneys were separated and checked under stereomicroscope. The helminths found were collected following the methodology of Amato et al. [3]. For identification of the helminths, the nematodes were clarified in lactic acid, and cestodes, trematodes, and acanthocephalans were stained with hydrochloric carmine. Thereafter, the helminths were mounted in temporary slides and examined under the light microscope ZEISS Axio Imager M2 [4].
The infection patterns for all helminth species found were estimated through prevalence (number of parasitized host individuals divided by the total number of individuals, expressed in percentage), mean abundance (number of helminths divided by total number of individuals in the sample), and mean intensity of infection (number of helminths divided by the number of parasitized individuals), followed by their respective standard errors [20]. Simpson’s dominance index was used to estimate which parasite community showed the greatest diversity among the host species P. albifrons, P. cicada, and P. cuvieri. A correlation test using Pearson’s coefficient was carried out to verify whether there was correlation between SVL of the hosts and abundance of endoparasites. Jaccard’s index for qualitative similarity, based on presence and absence of helminths, was used to evaluate the similarity of the parasitic infra-communities among the host species P. albifrons, P. cicada, and P. cuvieri.
The analyses were all carried out using the R software, with the package “R commander” [73].
Ethical Approval and/or Informed Consent
The sampling of the anuran specimens was authorized by the ‘Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis—IBAMA’ (Brazilian Institute of Environment and Renewable Natural Resources), authorization # 56612-1, and by the Ethical Committee of Universidade Regional do Cariri (CEUA/URCA, process # 00260/2016.1).
Results
A total of 264 hosts were sampled and necropsied—100 specimens of P. albifrons with prevalence of 20% and mean intensity of infection 2.15 ± 3.06, 93 P. cicada with prevalence of 27% and mean intensity of infection 4.42 ± 5.93, and 71 P. cuvieri with prevalence of 15% and mean intensity of infection 7.63 ± 14.27.
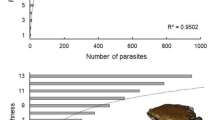
A total of 242 helminths were recovered from the hosts analyzed, with a mean of one to three species by host. The compound community associated with the three anuran species comprised ten taxa: Rhabdias cf. breviensis, Oswaldocruzia cf. mazzai, Cosmocerca parva, Raillietnema spectans, Schrankiana schranki, Oxyascaris oxyascaris, Physaloptera sp., Cylindrotaenia americana, Acanthocephalus cf. saopauloensis, and cystacanths. The nematode Raillietnema spectans [44] showed the highest prevalence, being present in the three host species (Table 1).
Simpson diversity index showed P. cicada (ID = 0.37) with the greatest diversity, P. albifrons (ID = 0.5) with intermediate, and P. cuvieri (ID = 0.62) with the lowest.
There was no significant correlation between parasite abundance (AB) and host size (SVL) for P. albifrons (r = 0.29; p = 0.1), P. cicada (r = 0.2; p = 0.13), and P. cuvieri (r = 0.005; p = 0.96).
The similarity index (S) of the compound community (10 taxa) showed the greatest similarity (57%) between P. albifrons and P. cuvieri (S = 0.57; DE = 1.73), while P. cicada showed the lowest similarity compared to P. albifrons (S = 0.22; DE = 2.64), and P. cuvieri (S = 0.37; DE = 2.44).
Discussion
Nematodes and trematodes are commonly recorded parasitizing amphibians of the Leptodactylidae, with nematodes of the Cosmocercidae, Molineidae, Physalopteridae, Rhabdiasidae, and trematodes of the Diplodiscidae, Glypthelminthidae, Gorgoderidae, and Plagiorchiidae the taxa most frequently found [25, 26, 33, 84].
According to Campião et al. [25], Silva et al. [80] and Teles et al. 84], 26 helminth species are recorded for members of the subfamily Leiuperinae, from which approximately 68% belong to the phylum Nematoda. Anurans of Physalaemus have 20 helminth species recorded, from which 69% are nematodes [2, 25, 60, 85].
The results of the present study represent 38.4% of the known parasite species for members of the subfamily Leiuperinae, and 52.6% for members of Physalaemus. This study also recovered nematodes and acanthocephalans as predominant components, following the same infection patterns of other congeneric hosts. Low host specificity is common for some helminth species [26], which justifies the high number of infection by nematodes of the Cosmocercidae.
The lowest richness among the three host species of this study (R = 5) was observed for P. albifrons, although it showed a great diversity of parasite communities, according to the Simpson index (ID = 0.5). Campião et al. [26] affirm that the geographical locality in association with ecology and life history of host, influence parasite richness, making difficult the contact with infectious forms, which can result in colonization by a less number of parasite species. Nevertheless, the composition of helminth communities of P. albifrons followed the same pattern of P. signifer (R = 4) and P. soaresi (R = 4), which were also composed mainly of nematodes [35, 88].
Among the eight species of Physalaemus already studied, P. cicada (R = 6) was the only one that had parasites belonging to more than two phyla—Nematoda, Platyhelminthes (Cestoda), and Acanthocephala (see [25]). Moreover, P. cicada showed high parasite richness, like P. albonotatus (R = 7; [46]) and P. santafecinus (R = 7; [45]). P. cicada also showed parasite diversity (ID = 0.37) superior to P. cuvieri and P. albifrons, suggesting that the form of habitat exploitation is associated with richness and parasite diversity of P. cicada [69]. Seasonal changes, biotic factors, like immune system and host age, may also affect parasitism, influencing the life of the parasite and its host [67]. Physalaemus cicada follows the same infection patterns as other Neotropical anurans, in which infections formed almost exclusively by nematodes can be observed [25]. Up to date, this is the only surveyed Physalaemus species that showed members of Cestoda as a component of the helminthfauna.
Physalaemus cuvieri (R = 6), showed the lowest diversity (ID = 0.62), but had the same infection patterns (R = 8) as observed by Aguiar et al. [2], Leivas et al. [60], Santos and Amato [75] and Toledo et al. [85] for component helminth fauna. The present study shows two new records for this host (R. spectans and S. schranki), increasing the known parasite richness for P. cuvieri (10 taxa).
The values of parasite richness found for P. albifrons (R = 5), P. cicada (R = 6), and P. cuvieri (R = 6) in the present study are similar to the results in the literature for the eight Physalaemus species already surveyed [2, 25, 60, 85]. These values indicate that Physalaemus species show low richness of parasite taxa, which seems like a common pattern for this genus. According to Campião et al. [26], parasite richness is linked to geographical distribution, host size, and transmission mode of the parasite.
Inventories of parasites associated with members of the Leptodactylidae, like Pseudopaludicola boliviana from Argentina (R = 9; [33, 47]) Leptodactylus chaquensis and Leptodactylus podicipinus from the Brazilian Pantanal (R = 14; R = 11, [23]), and [65] for 21 species of leptodactylids from Mato Grosso State, Brazil (mean richness = 10 species), suggest infection patterns with high parasite richness for this group. However, such patterns were not xobserved in the present study for P. albifrons, P. cicada, and P. cuvieri, similarly to the results of Teles et al. [84] in Pleurodema diplolister and Silva et al. [80] in Pseudopauludicola pocoto. Thus, leptodactylids from Caatinga might harbor lower richness of endoparasites compared to leptodactylids from other localities.
Lins et al. [61] surveyed the helminths of Leptodactylus syphax from the same locality as the present study, and found a richness of six parasite species (Aplectana membranosa, Schrankiana formosula, Physaloptera sp., Lophosicyadiplostomum sp., cystacanths, and no identified larvae). Such richness is similar to what was found in this study for P. albifrons, P. cicada, and P. cuvieri, demonstrating that the environment is a key factor to predict parasite richness in Neotropical anurans. However, the parasitic composition differs from the observed for the Physalaemus spp. of this study. The composition of helminth communities in anurans depend on several factors, like body size, gender, diet, infection site, behavior, host species, and interactions with the environment [18, 26]. According to Barton [9] and Bursey et al. [19], such communities are composed of generalist species of direct transmission, favoring parasites with infectious stage in soil, like nematodes [22]. Communities richness can also vary according to environmental conditions, due to the way that species respond to biotic factors [71]. The absence of trematode species in our study is noteworthy, since species of Physalaemus are semiaquatic. Trematodes in general require aquatic intermediate hosts in addition to snails. Therefore, anurans which live in aquatic habitats are more easily infected with digeneans than terrestrial species [48]. The Caatinga has a marked water deficit and this may be related to the low diversity or even the absence of trematodes in parasitological studies conducted in this domain.
The results of the present study follow the common pattern of parasite communities found in leptodactylids of South America, with dominance of nematodes [25, 47, 65, 76, 84]. For the total richness (R = 10) of P. albifrons, P. cicada, and P. cuvieri studied herein, only the cosmocercid R. spectans and cystacanths were present in the three host species.
Raillietnema spectans had the highest prevalence (11%), similar to the patterns found by Morais [65] for 21 species of leptodactylids from Mato Grosso State, Brazil. This result shows that Physalaemus spp. present an interaction web of parasites composed of generalist species of direct infection, a common pattern found in the literature for Neotropical anurans.
According to Baker [7], acanthocephalans are parasites with indirect life cycle, with arthropods (usually crustaceans) as intermediate host, and fishes or aquatic birds as final hosts. The presence of cystacanths in P. albifrons, P. cicada, and P. cuvieri indicate that these species are serving as paratenic hosts of acanthocephalans. The cystacanths were probably acquired through the diet of Physalaemus spp. that is composed mainly of ants and coleopterans [30, 60].
Given the increase of studies on parasites of amphibians [6, 22,23,24,25,26,28, 51, 60, 61, 74], new host records are becoming common, as well as records of parasite taxa that are still new to science [1, 6].
The parasite R. spectans was reported before in Rhinella crucifer, R. icterica, Leptodactylus latrans [25], and Leptodactylus mystaceus [65]. The present study is a new host record of R. spectans parasitizing P. albifrons, P. cicada, and P. cuvieri from Caatinga area.
Schrankiana schranki [86] was reported before in Leptodactylus latinasus, L. pentadactylus, L. rhodomystax, L. vastus, L. labyrinthicus, L. mystaceus, L. fuscus, and L. pustulatus [25, 43, 65], which is a new record of S. schranki parasitizing P. cuvieri and P. cicada from the Caatinga area. Oswaldocruzia cf. mazzai [87] was reported before by Morais [65] in Rhinella icterica, R. margaritifera, R. marina, Leptodactylus bufonius, L. fuscus, L. chaquensis, L. knudseni, L. labyrinthicus, L. latrans, and L. rhodomystax. In the present study, this nematode is a new host record for P. albifrons and P. cuvieri.
The cestode Cylindrotaenia americana [56] was recorded before in the amphibians R. icterica [83] and P. olfersii [85] in Brazil, R. marina in Colombia [17], L. latrans in Argentina [78], Lithobates septentrionalis in the United States [14], Hyla arborea in the former Czechoslovakia [72], and in the reptile Scincella lateralis [16, 54]. In the present study, we present the first record of C. americana parasitizing P. cicada from the Caatinga area in Brazil. The low mean abundance found in this study for S. schranki corroborate the findings of Hamann et al. [53] for L. latinasus in Argentina, but not the findings of Dyer and Altig [34] for L. pentadactylus in Ecuador. For mean abundance of the cestode C. americana, this study corroborates the findings of Bouchard [14], Brooks [16, 17], Harwood [54], Prokopic [72], Savazzini [78], and Stumpf [83] that also presented low mean abundance values for this parasite. Low abundance of parasite can be an occasional finding since there is not a common pattern among amphibians. Infection abundance is related to environmental factors, influencing parasite or host species in different ways, which can alter infection rates [57, 68]. The ecology, life history, and geographical location of the hosts can also influence parasite infection rates [26].
Host body size is suggested as a determinant to predict species richness, affecting the composition of parasite communities [26]. However, the values of SVL and parasite abundance did not show any correlation in the present study. As proposed by George-Nascimento et al. [40], larger hosts (body mass and SVL) can harbor greater parasitic loads and species richness [26] because they can offer more diversity of microhabitat, favoring the development and reproduction of parasites. Even though body size influences the acquisition of parasites, other aspects like physiology, behavior, and seasonality can also explain variations in the abundance of parasites [15].
In the present study, the analyses of similarity of parasite composition showed that the greatest similarity was between P. albifrons and P. cuvieri (S = 0.57; DE = 1.73), while P. cicada was more similar to P. cuvieri (S = 0.37; DE = 2.44) than P. albifrons (S = 0.22; DE = 2.64). Such proximity can be explained by the phylogenetic relationship among species of this group [63]. Krasnov et al. [59] affirm that phylogeny is a determinant factor for structuring webs between parasites and hosts since the phylogenetic proximity is reflected on phenotypic similarities, leading parasite species to explore particular hosts. This result can also be explained by niche occupation, given that hosts sharing the same habitat are exposed to the same web of interactions [58]. Moreover, Martin et al. [64] suggest that host species with similar diet show more similarity of parasite composition, as observed for P. albifrons and P. cuvieri.
The present study evaluated the helminth associated with three species of Physalaemus from the Caatinga area recovering four new host records, which highlights the importance of parasite inventories for host species and poorly researched areas. The pattern presented in this study indicates that the parasite communities associated with the studied hosts are composed mainly of generalist parasites with direct life cycle, which is a common standard for the species of Physalaemus. No relation was observed between mean host size and average parasite abundance. In addition, the highest similarity of the parasite composition associated with P. albifrons and P. cuvieri in relation to P. cicada shows this to be related to the phylogenetic proximity of the species.
References
Aguiar, A. 2013. Helmintofauna associada à anfíbios da Ilha Anchieta, Litoral norte do estado de São Paulo, Brasil. Masters dissertation, Botucatu, SP. Universidade Estadual Paulista—UNESP. (In Portuguese).
Aguiar, A., Toledo, G.M., Anjos, L.A., Silva, R.J. 2015. Helminth parasite communities of two Physalaemus cuvieri Fitzinger, 1826 (Anura: Leiuperidae) populations under different conditions of habitat integrity in the Atlantic Rain Forest of Brazil. Brazilian Journal of Biology 75(4): 963–968. https://doi.org/10.1590/1519-6984.03614.
Amato, F.R., Boeger, W.A., Amato, S.B. 1991. Protocolos para laboratório—Coleta e processamento de parasitos de pescado. Rio de janeiro: Imprensa Universitária-UFRRJ, 81p. (In Portuguese).
Andrade, C.M. 2000. Meios e soluções comumente empregados em laboratórios. Editora Universidade Rural, Rio de Janeiro, pp.353. (In Portuguese).
Andrade, G., Flora J. 2004. Physalaemus albifrons. In: IUCN Red List of Threatened Species. Version 2011.1. Retrieved August 25, 2016 from http://www.iucnredlist.org.
Araujo-Filho, J.A., Brito, S.V., Oliveira, W.O., Morais, D.H., Ávila, R.W. 2015. A new species of Parapharyngodon (Nematoda: Pharyngodonidae) infecting Dermatonotus muelleri (Anura: Microhylidae) from Caatinga. Northeastern Brazil. Zootaxa, 4012(2): 386–390. https://doi.org/10.11646/zootaxa.4012.2.10.
Baker, D.G. 2007. Acanthocephala In: Flynn’s Parasites of Laboratory Animals: Second Edition. Second ed. Oxford, UK: Blackwell Publishing Ltd, pp. 193–193.
Baker, M.R. 1980. Revision of World species of the genus Aplectana (Railliet and Henry, 1916) (Nematoda, Cosmocercidae). Adansonia, 2(4): 955–998.
Barton, D.P. 1999. Ecology of helminth communities in tropical Australian amphibians. International Journal for Parasitology. 29: 921–926. https://doi.org/10.1016/s0020-7519(99)00057-0.
Bernarde, P.S. 2012. Anfíbios e Répteis: Introdução ao Estudo da Herpetofauna Brasileira. 1. ed. Curitiba: Anolis Books, v. 1. 320p. (In Portuguese).
Bezerra, C.H., Pinheiro, L.T., Melo, G.C., Zanchi-Silva, D., Queiroz, M.S., Anjos, L.A., et al. 2016. Assessing the influence of geographic distance in parasite communities of an exotic lizard. Acta Parasitologica, 61(1): 136–143. https://doi.org/10.1515/ap-2016-0018.
Bokermann, W.C.A. 1966. Dos nuevas especies de Physalaemus de Espiritu Santo, Brasil (Amphibia, Leptodactylidae). Physis, 26(71), 193-202. (In portuguese).
Bolek, M.G., Coggins. J.R. 2003. Helminth community structure of sympatric eastern American toad, Bufo americanus, northern leopard frog, Rana pipiens, and blue-spotted salamander, Ambystoma laterale, from southeastern Wisconsin. Journal of Parasitology, 89: 673–680. https://doi.org/10.1645/ge-70r.
Bouchard, J.L. 1951. Platyhelminthes parasitizing some northern Maine Amphibia. Transactions of the American Microscopical Society. 70: 245–250. https://doi.org/10.2307/3223054.
Brito, S.V., Corso, G., Almeida, A.M., Ferreira, F.S., Almeida, W.O.; Anjos, L.A., Vasconcellos, A. 2014. Phylogeny and micro-habitats utilized by lizards determine the composition of their endoparasites in the semiarid Caatinga of Northeast Brazil. Parasitology Research, 113: 3963–3972.
Brooks, D.R. 1972. Intestinal parasites of the lizard Lygosoma laterale. journal of the Florida Academy of Sciences. 35: 4–8.
Brooks, D.R. 1976. Five species of platyhelminths from Bufo marinus L. (Anura: Bufonidae) in Colombia with descriptions of Creptotrema lynchi sp. n. (Digenea: Allocreadiidae) and Clypthelmius robustus sp. n. (Digenea: Macroderoididae). Journal of Parasitology, 62: 429–433.
Brooks, D.R., León-Regagnon, V., McLennan, D.A., Zelmer, D. 2006. Ecological fitting as a determinant of the community structure of platyhelminth parasites of anurans. Ecology, 87: 76–85. https://doi.org/10.1890/0012-9658(2006)87%5b76:efaado%5d2.0.co;2.
Bursey, C.R., Goldberg, S.R., Parmelee, J.R. 2001. Gastrointestinal helminths of 51 species of Anurans from Reserva Cuzco Amazónico, Peru. Comparative Parasitology 68: 21–35.
Bush, A.O., Lafferty, K.D., Lotz, J.M., Shostak, A.W. 1997. Parasitology Meets Ecology on Its Own Terms: Margolis et al. Revisited. Journal of Parasitology, 83(4): 575–583. https://doi.org/10.2307/3284227.
Caldas, F.L.S., De-Carvalho, C.B., Santana, D.O., Santos, R.A., Silva, B.D., Faria, R.G. 2010. Amphibia, Anura, Leiuperidae, Physalaemus cicada Bokermann, 1966: First records fort the state of Sergipe. Check List 6(3): 427–428. https://doi.org/10.15560/6.3.427.
Campião, K.M. 2010. Influência das características ambientais na comunidade de helmintos parasitas de Leptodactylus podicipinus (Anura, Leptodactylidae) de lagoas do Pantanal da Nhecolândia, Corumbá, Mato Grosso do Sul. Masters dissertation, Campo Grande. Federal University of Mato Grosso do Sul. (In Portuguese).
Campião, K.M., Dias, O.T., Silva, R.J., Ferreira, V.L., Tavares, L.E.R. 2016a. Living apart and having similar trouble: are frog helminth parasites determined by the host or by the habitat? Canadian Journal of Zoology, 94: 761–765. https://doi.org/10.1139/cjz-2016-0066.
Campião, K.M., Silva, I.C.O., Dalazen, G.T., Paiva, F., Tavares, L.E.R. 2016b. Helminth Parasites of 11 Anuran Species from the Pantanal Wetland, Brazil. Comparative Parasitology, 83(1): 92–100. https://doi.org/10.1654/1525-2647-83.1.92.
Campião, K.M., Morais, D.H, Dias, O.T., Aguiar, A., Toledo, G., Tavares, L.E.R., Da Silva, R.J. 2014. Checklist of Helminth parasites of Amphibians from South America. Zootaxa 3843(1): 1–93. https://doi.org/10.1016/s0020-7519(99)00057-0.
Campião, K.M, Ribas, A.C.A., Morais, D.H, Dias, O.T, Silva, R.J., Tavares, L.E.R. 2015a. How Many Parasites Species a Frog Might Have? Determinants of Parasite Diversity in South American Anurans. PloSone, 10(10): e0140577. https://doi.org/10.1371/journal.pone.0140577.
Campião, K.M., Ribas, A., Tavares, L.E.R. 2015b. Diversity and patterns of interaction of an anuran-parasite network in a neotropical wetland. Parasitology, 142: 1751–1757. https://doi.org/10.1017/s0031182015001262.
Campião, K.M., Ribas, A.C.A., Cornell, S.J., Begon, M. Tavares, L.E.R. 2015c. Estimates of coextinction risk: how anuran parasites respond to the extinction of their hosts? International Journal for Parasitology, 45: 885–889. https://doi.org/10.1016/j.ijpara.2015.08.010.
Cfmv. Conselho Federal de Medicina Veterinária. 2013. Métodos de eutanásia. In: Guia brasileiro de boas práticas de eutanásia em animais, pp. 28-29. Comissão de ética, Bioética e bem-estar animal. Brasília, Distrito Federal. (In Portuguese).
Cruz, L.C. 2000. Utilização de Recursos Espaciais e Alimentares por Physalaemus albifrons (Anura: Leptodactylidae) das Dunas do Rio São Francisco, Bahia. Bachelor’s Degree, Instituto de Ciências Biológicas da UFBA, Salvador. (In Portuguese).
Da Graça, R.J., Oda, F.H., Lima, F.S., Guerra, V., Gambale, P.G., Takemoto, R.M. 2017. Metazoan endoparasites of 18 anuran species from the mesophytic semi deciduous Atlantic Forest in southern Brazil, Journal of Natural History, 51: 705–729. https://doi.org/10.1080/00222933.2017.1296197.
Dobson, A., Lafferty, K.D., Kuris, A.M., Hechinger, R.F., Jetz, W. 2008. Homage to Linnaeus: How many parasites? How many hosts? Proceedings of the National Academy of Sciences of the United States of America, 105 (1): 11482–11489. https://doi.org/10.1073/pnas.0803232105.
Duré, M.I., Schaefer, E.F., Hamann, M.I., Kehr, A.I. 2004. Consideraciones ecológicas sobre la dieta, lareprodución y el parasitismo de Pseudopaludicola boliviana (Anura, Leptodactylidae) de Corrientes, Argentina. Phylomedusa, 3 (2), 121–131. (In Spanish).
Dyer, W.G., Altig, R. 1977. Helminths of some ecuadorian anurans. Herpetologica, 33: 293–296.
Fabio, S. P. 1978. Helmintos de populações simpátricas de algumas espécies de anfíbios anuros da família Leptodactylidae. Arquivos da Universidade Federal Rural do Rio de Janeiro, 5, 69–83. (In Portuguese).
Fabio, S.P. 1982. Helmintos de populações simpátricas de algumas espécies de anfíbios anuros da família Leptodactylidae. Arquivos da Universidade Federal Rural do Rio de Janeiro, 5, 69–83. (In portuguese).
Fitzinger, L.J.F.J., 1826. Neue Classification der Reptilien nach ihren naturlichen Verwandtscaften. Viena (In German).
Franco, F.L., Salomão, M.G. 2002. Répteis. Coleta e preparação de répteis para coleções científicas: considerações iniciais In: P. AURICCHIO; SALOMÃO, M. G. (Eds). Técnicas de coleta e preparação de vertebrados para fins científicos e didáticos. São Paulo: Pau Brazil Institute of Natural History, cap.3, p.77–123.
Frost, D.R. 2018. Amphibian Species of the World: an Online Reference. Version 6.0. In: American Museum of Natural History, New York, USA. Retrieved March 03, 2018 from http://research.amnh.org/herpetology/amphibia/index.html.
George-Nascimento, M., Muñoz, G., Marquet, P.A., Poulin, R. 2004. Testing the energetic equivalence rule with helminth endoparasites of vertebrates. Ecology Letters, 7: 527–531. https://doi.org/10.1111/j.1461-0248.2004.00609.x.
Goater, C.P., Baldwin, R.E., Scrimgeour, G. J. 2005. Physico-chemical determinant community structure in whitefish (Coregonus clupeaformes) from adjacent lakes in Northern Alberta, Canada. Parasitology, 131: 713–722. https://doi.org/10.1017/s0031182005008371.
Goldberg, S.R., Bursey, C.R., Trujillo, D.J., Kaiser, H. 2002. Intestinal helminths of seven frog species from Trinidad and Tobago. Caribbean Journal of Science, 38: 147–150.
Goldberg, S.R., Bursey, C.R., Caldwell, J.P., Vitt, L.J., Costa, G.C. 2007. Gastrointestinal helminths from six species of frogs and three species of lizards, sympatric in Pará state, Brazil. Comparative Parasitology, 74: 327–342. https://doi.org/10.1654/4268.1.
Gomes, D.C. 1964. Sobre uma nova espécie do gênero Raillietnema Travassos, 1927 (Nematoda, Cosmocercidae). Atas Sociedade de Biologia do Rio de Janeiro, 8(5), 53–55. (In portuguese).
González, C.E. Hamann, M.I. 2010. First report of nematode parasites of Physalaemus santafecinus (Anura: leiuperidae) from corrientes, Argentina. Revista Mexicana de Biodiversidad, 81: 677–687.
González, C.E. Hamann, M.I. 2012a. First report of nematode of Physalaemus albonotatus (Stundachner 1864) (Anura: leiuperidae) from corrientes, Argentina. Neotropical Helminthology, 6: 9–23.
González, C.E., Hamann, M.I. 2012b. Seasonal occurrence of Cosmocerca podicipinus (Nematoda: Cosmocercidae) in Pseudopaludicola boliviana (Anura: Leiuperidae) from natural environments in Corrientes Province, Argentina and aspects of its population structure. Parasitology Research, 111: 1923–1928.
Guillén-Hernández, S., Salgado-Maldonado, G., Lamothe-Argumedo, R. 2000. Digeneans (Plathelminthes: Trematoda) of seven sympatric species of anurans from Los Tuxtlas, Veracruz, Mexico. Studies on Neotropical Fauna and Environment, 35:10–13.
Gutierrez, C., Attademo, A., Guerrero, S., Peltzer, P., Lajmanovich, R. 2005. Physalaemus biligonigerus (False-eyed Frog). Endoparasites. Herpetological Review, 36: 161–162.
Haddad, C.F.B., Toledo, L.F., Prado, C.P.A. 2008. Anfíbios da Mata Atlântica. São Paulo: Editora Neotropica. (In Portuguese).
Hamann, M.I., Kehr, A.I., González, C.E. 2010. Helminth community structure of Scinax nasicus (Anura: Hylidae) from a South American subtropical area. Diseases of Aquatic Organisms, 93(1): 71–82. https://doi.org/10.3354/dao02276.
Hamann, M.I., González, C.E. 2009. Larval digenetic trematodes in tadpoles of six amphibian species from Northeastern Argentina. Journal of Parasitology, 95(3): 623–628. https://doi.org/10.1645/ge-1738.1.
Hamann, M.I., González, C.E., Kehr, A.I. 2006. Helminth community structure of the oven frog Leptodactylus latinasus (Anura, Leptodactylidae) from Corrientes, Argentina. Acta Parasitologica, 51(4): 294–299. https://doi.org/10.2478/s11686-006-0045-1.
Harwood, P.D. 1936. The effect of soil types on the helminths parasitic in the ground lizard Leiolopisma laterale (Say). Ecology, 17: 694–698.
Ipece—Instituto de Pesquisa e Estratégia Econômica do Ceará. 2017. Perfil municipal 2016. Retrieved October 10, 2017 from http://www.ipece.ce.gov.br/index.php/2016-12-16-13-09-40 (In Portuguese).
Jewell, M.E. 1916. Cylindrotaenia americana nov. spec. from the cricket frog. The Journal of Parasitology, 2(4): 181-192.
Koprivnikar, J., Baker, R.L., Forbes, M.R. 2006. Environmental factors influencing trematode prevalence in grey tree frog (Hyla versicolor) tadpoles in Southern Ontario. Journal of Parasitology, 92: 997–1001. https://doi.org/10.1645/ge-771r.1.
Krause, A.E., Frank, K.A., Mason, D.M., Ulanowicz, R.E., Taylor, W.W. 2003. Compartments revealed in food-web structure. Nature, 426(6964): 282–285.
Krasnov, B.R., Fortuna, M.A., Mouillot, D., Khokhlova, I.S., Shenbrot, G I., Poulin, R. 2012. Phylogenetic signal in module composition and species connectivity in compartmentalized host-parasite networks. American Naturalist, 179(4): 501–511.
Leivas, P.T., Leivas, F.W.T., Campião, K.M. 2018. Diet and parasites of the anuran Physalaemus cuvieri Fitzinger, 1826 (Leiuperidae) from an Atlantic Forest fragment. Herpetology Notes, 11: 109–113.
Lins, A.G.S., Aguiar, A., Morais, D.H., Da Silva, L.A.F., Ávila, R.W., Silva, R.J. 2017. Helmintofauna de Leptodactylus syphax (Anura: Leptodactylidae) do bioma da Caatinga, Nordeste do Brasil. Revista Brasileira De Parasitologia Veterinaria . 26: 74–80. https://doi.org/10.1590/s1984-29612017013 (in Portuguese).
Lisboa, B.S., Haddad, C.F.B. 2009. Amphibia, Anura, Leiuperidae, Physalaemus cicada Bokermann, 1966: Distribution extension and geographic distribution map. Check List 5(3): 699–701.
Lourenço, L.B., Targueta, C.P., Baldo, D., Nascimento, J., Garcia, P.C., Andrade, G.V., et al. 2015. Phylogeny of frogs from the genus Physalaemus (Anura, Leptodactylidae) inferred from mitochondrial and nuclear gene sequences. Molecular phylogenetics and evolution, 92: 204–216. https://doi.org/10.1016/j.ympev.2015.06.011.
Martin, J.E., Llorente, G.A., Roca, V., Carretero, M.A., Montori, A., Santos, X., Romeu, R. 2005. Relationship between diet and helminths in Gallotia caesaris (Sauria: Lacertidae). Zoology, 108(2): 121–130. https://doi.org/10.1016/j.zool.2005.03.002.
Morais, D.H. 2013. Aspectos ecológicos da helmintofauna de anfíbios Leptodactylidae (ANURA) no Estado do Mato Grosso, Brasil. Doctorate thesis, UNESP—Campus de Botucatu, Botucatu-SP. (In Portuguese).
Nascimento, L.B., Caramaschi, U., Cruz, C.A.G. 2005. Taxonomic review of the species groups of the genus Physalaemus Fitzinger, 1826. Arquivos do Museu Nacional do Rio de Janeiro, 63(2): 297–320.
Pietrock, M., Marcogliese, D.J. 2004. Response to Morley and Lewis: Freeliving endohelminths: the influence of multiple factors. Trends Parasitology, 20(3): 115–116.
Pizzato, L., Kelehear, C., Shine, R. 2013. Seasonal dynamics of the lungworm, Rhabdias pseudosphaerocephala, in recently colonised cane toad (Rhinella marina) populations in tropical Australia. International journal for parasitology, 43: 753–761. https://doi.org/10.1016/j.ijpara.2013.05.002.
Poulin, E., Morand, S. 2004. Parasite Biodiversity. Smithsonian Books, Washington Price P.W. 1980. Evolutionary Ecology of Parasites. Princetown University Press, Princetown, 237p.
Poulin, R., Blanar, C.A., Thieltges, D.W., Marcogliese, D.J. 2011. The biogeography of parasitism in sticklebacks: distance, habitat differences and the similarity in parasite occurrence and abundance. Ecography, 34: 540–551 https://doi.org/10.1111/j.1600-0587.2010.06826.x.
Poulin, R., Krasnov, B.R. 2010. Similarity and variability of parasite assemblages across geographical space. In: Morand S, Krasnov BR. The biogeography of host-parasite interactions. New York: Oxford University. p. 115–128.
Prokopic, J.K. 1957. Helmintofaune Nasich Zab. Folia Parasitologica, 4: 249–262.
R Core Team. 2013. R: A language and environment for statistical computing. R Foundation for Statistical Computing.
Santos, V.G.T., Amato, S.B. 2010. Helminth fauna of Rhinella fernandezae (Anura: Bufonidae) from the Rio Grande do Sul coastland, Brazil: analysis of the parasite community. Journal of Parasitology, 96(4): 823–826. https://doi.org/10.1645/ge-2388.1.
Santos, V.G.T., Amato, S.B. 2012. Polystoma cuvieri (Monogenea, Polystomatidae) in physalaemus cuvieri (Anura, Leiuperidae) in southern Brazil. Neotropical Helminthology, 6(1): 1–8.
Santos, V.G.T., Amato, S.B. 2013. Species of Cosmocerca (Nematoda, Cosmocercidae) in anurans from Southern Santa Catarina State, Brazil. Comparative Parasitology, 80: 123–129. https://doi.org/10.1654/4608.1.
Santos, V.G.T., Amato, S.B., Borges-Martins, M. 2013. Community structure of helminth parasites of the “Cururu” toad, Rhinella icterica (Anura: Bufonidae) from southern Brazil. Parasitology Research, 112: 1097–1103.
Savazzini, L.A. 1929. La Cylindrotaenia americana en nuestro Leotodactulus occellatus. Semana Médica Buenos Aires. 36: 868–870. (In Spanish).
Segalla, M.V., Caramaschi, U., Cruz, C.A.G., Grant, T., Haddad, C.F.B., Garcia, P.C.A., et al. 2016. Brazilian Amphibians: List of Species, Herpetologia Brasileira—5:2. Retrieved September 28, 2016 from http://www.sbherpetologia.org.br.
Silva, C.S., Ávila, R.W., Morais, D.M. 2018. Helminth community dynamics in a population of Pseudopaludicola pocoto (Leptodactylidae: Leiuperinae) from Brazilian Northeast. Helminthologia, in press.
Silva, M.C., Oliveira, D.B., Oliveira, H.F., Roberto, I.J., Morais, D.H., Brito, S.V., Ávila, R.W. 2013. Geographic distribution of Physalaemus cicada Bokermann, 1966 (Anura: Leiuperidae) in Northeastern Brazil. Check List, 9(5): 1119–1121.
Silveira, A. L. 2006. Anfíbios do município de João Pinheiro, uma área de cerrado no noroeste de Minas Gerais, Brasil. Arquivos do Museu Nacional, 64(2): 134–139. (In Portuguese).
Stumpf, I.V.K. 1981. Biological aspects of Cylindrotaenia americana Jewell, 1916 (Cyclophyllidae: Nematotaeniidae) in Bufo ictericus Spix, 1824. Acta Biológica Paranaense. Curitiba. 10: 41–52.
Teles, D.A., Sousa, J.G.G., Teixeira, A.A.M., Silva, M.C., Oliveira, R.H., Silva, M.R.M., Ávila, R.W. 2015. Helminths of the frog Pleurodema diplolister (Anura, Leiuperidae) from the Caatinga in Pernambuco State, Northeast Brazil. Brazilian Journal of Biology, 75: 251–253.
Toledo, G.M., Aguiar, A., Silva, R.J., Anjos, L.A. 2013. Helminth Fauna of Two Species of Physalaemus (Anura: Leiuperidae) from an Undisturbed Fragment of the Atlantic Rainforest, Southeastern Brazil. Journal of Parasitology, 99(5): 919–922. https://doi.org/10.1645/ge-3212.1.
Travassos, L. 1925. Contribuições para o conhecimento da fauna helmintológica dos batráchios do Brasil. Nematódeos intestinais. Sciencia Medica, 3(1): 673–687. (In portuguese).
Travassos, L. 1935. Alguns novos gêneros e espécies de Trichostrongylideos. Revista de Medicina e Cirurgia Brasileira, 43(11): 345–361. (In portuguese).
Vicente, J.J., Rodrigues, H.O., Gomes, D.C., Pinto, R.M. 1991. Nematoides do Brasil. 2ª parte: Nematóides de anfíbios. Revista Brasileira de Zoologia, 7: 549–626.
Walton, A.C. 1935. The Nematoda as parasites of Amphibia. II. Journal of Parasitology, 21(1): 27–50. https://doi.org/10.2307/3271792.
Acknowledgements
The authors thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for providing a research fellowship to RWA (# 303622/2015-6) and Fundação Cearense de Apoio ao Desenvolvimento Científico e Tecnológico (FUNCAP) for providing the Master fellowship to CRO. Top level staffing coordination provided to research fellowship to DHM (CAPES / PNPD 22005013001P4).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
de Oliveira, C.R., Ávila, R.W. & Morais, D.H. Helminths Associated with Three Physalaemus Species (Anura: Leptodactylidae) from Caatinga Biome, Brazil. Acta Parasit. 64, 205–212 (2019). https://doi.org/10.2478/s11686-018-00022-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11686-018-00022-8