Abstract
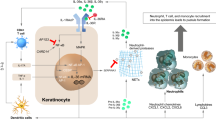
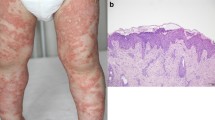
Generalized pustular psoriasis (GPP) is a chronic disease characterized by non-bacterial pustules. Variants in several genes, such as IL36RN, AP1S3, and CARD14, are involved in the pathogenesis of GPP. The prevalence of different gene variants varies among ethnicities, and some variants are related to concurrent psoriasis vulgaris or age at onset. Flares can be triggered by medications (most commonly corticosteroids), infections (possibly due to Toll-like receptor [TLR] and antimicrobial peptides), pregnancy (the onset of GPP has been attributed to endocrine abnormalities such as hypoparathyroidism and hypocalcaemia), hypocalcaemia (presumably due to low levels of calcium and vitamin D regulating the proliferation and differentiation of keratinocytes), and other factors including stress and sun exposure. The mechanisms of pustule formation involve: 1) the LL37/TLR pathway, in which LL37 acts as an alarmin, interacting with TLR and activating the NF-κB and MAPK pathways; 2) the balance between calcium and 1,25(OH)2D levels, and 3) neutrophils and the complement system.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Choon SE, Lai NM, Mohammad NA, Nanu NM, Tey KE, Chew SF. Clinical profile, morbidity, and outcome of adult-onset generalized pustular psoriasis: analysis of 102 cases seen in a tertiary hospital in Johor, Malaysia. Int J Dermatol 2014; 53:676–84.
Takeichi T, Akiyama M. Generalized Pustular Psoriasis: Clinical Management and Update on Autoinflammatory Aspects. Am J Clin Dermatol 2020;21:227–36.
Zelickson BD, Muller SA. Generalized Pustular Psoriasis: A Review of 63 Cases. Arch Dermatol 1991;127:1339–45.
Baker H, Ryan TJ. Generalized pustular psoriasis. A clinical and epidemiological study of 104 cases. Br J Dermatol 1969; 80:771–93.
Jin H, Cho H-H, Kim W-J, et al. Clinical features and course of generalized pustular psoriasis in Korea. J Dermatol 2015;42:674–8.
Kharawala S, Golembesky AK, Bohn RL, Esser D. The clinical, humanistic, and economic burden of generalized pustular psoriasis: a structured review. Expert Rev Clin Immunol 2020; 16: 239–52.
Johnston A, Xing X, Wolterink L, et al. IL-1 and IL-36 are dominant cytokines in generalized pustular psoriasis. J Allergy Clin Immunol 2017; 140:109–20.
Sugiura K, Takemoto A, Yamaguchi M, et al. The majority of generalized pustular psoriasis without psoriasis vulgaris is caused by deficiency of interleukin-36 receptor antagonist. J Invest Dermatol 2013; 133:2514–21.
Körber A, Mössner R, Renner R, et al. Mutations in IL36RN in patients with generalized pustular psoriasis. J Invest Dermatol 2013; 133:2634–7.
Li X, Chen M, Fu X, et al. Mutation analysis of the IL36RN gene in Chinese patients with generalized pustular psoriasis with/without psoriasis vulgaris. J Dermatol Sci 2014; 76: 132–8.
Zhu T, Jin HZ. Pathogenic susceptibility genes for generalized pustular psoriasis. Med J PUMCH 2016; 7:445–9.
Sophie T, Alshimaa M, Nick D, et al. Clinical and genetic differences between pustular psoriasis subtypes. J Allergy Clin Immunol 2019; 143:1021–6.
Li M, Han J, Lu Z, et al. Prevalent and Rare Mutations in IL-36RN Gene in Chinese Patients with Generalized Pustular Psoriasis and Psoriasis Vulgaris. J Invest Dermatol 2013; 133: 2637–9.
Setta-Kaffetzi N, Navarini AA, Patel VM, et al. Rare Pathogenic Variants in IL36RN Underlie a Spectrum of Psoriasis-Associated Pustular Phenotypes. J Invest Dermatol 2013;133:1366–9.
Shu D, Jin HZ. Mutation analysis of IL36RN in patients with generalized pustular psoriasis. J Clin Dermatol 2014;43:531–5.
Marrakchi S, Guigue P, Renshaw BR, Puel A, Smahi A. Interleukin-36-receptor antagonist deficiency and generalized pustular psoriasis. N Engl J Med 2011; 365:620–8.
Farooq M, Nakai H, Fujimoto A, Fujikawa H, Shimomura Y. Mutation Analysis of the IL36RN Gene in 14 Japanese Patients with Generalized Pustular Psoriasis. Hum Mutat 2013; 34: 176–83.
Zhu T, Jin H, Shu D, Li F, Wu C. Association of IL36RN mutation with clinical features, therapeutic response to acitretin, and frequency of recurrence in patients with generalized pustular psoriasis. Eur J Dermatol 2018;28:217–24.
Liang J, Huang P, Li H, et al. Mutations in IL36RN are associated with geographic tongue. Hum Genet 2017; 136: 241–52.
Jordan CT, Cao L, Roberson EDO, et al. Rare and Common Variants in CARD14, Encoding an Epidermal Regulator of NF-kappaB, in Psoriasis. Am J Hum Genet 2012;90:796–808.
Sugiura K, Muto M, Akiyama M. CARD14 c.526G > C (p.Asp176His) is a significant risk factor for generalized pustular psoriasis with psoriasis vulgaris in the Japanese cohort. J Invest Dermatol 2014; 134:1755–7.
Berki DM, Liu L, Choon SE, et al. Activating CARD14 Mutations Are Associated with Generalized Pustular Psoriasis but Rarely Account for Familial Recurrence in Psoriasis Vulgaris. J Invest Dermatol 2015;135:2964–70.
Mössner R, Wilsmann-Theis D, Oji V, et al. The genetic basis for most patients with pustular skin disease remains elusive. Br J Dermatol 2018;178:740–8.
Qin P, Zhang Q, Chen M, et al. Variant Analysis of CARD14 in a Chinese Han Population with Psoriasis Vulgaris and Generalized Pustular Psoriasis. J Invest Dermatol 2014; 134: 2994–6.
Ammar M, Jordan CT, Cao L, et al. CARD14 alterations in Tunisian patients with psoriasis and further characterization in European cohorts. Br J Dermatol 2016; 174: 330–7.
Akiyama M, Takeichi T, McGrath JA, Sugiura K. Autoinflammatory keratinization diseases: An emerging concept encompassing various inflammatory keratinization disorders of the skin. J Dermatol Sci 2018; 90: 105–11.
Takeichi T, Akiyama M. Familial or sporadic porokeratosis as an autoinflammatory keratinization disease. J Dermatol 2019;46: e125–6. doi: https://doi.org/10.1111/1346-8138.14666 [Epub ahead of print].
Akiyama M. Autoinflammatory Keratinization Diseases (AiKDs): Expansion of Disorders to Be Included. Front Immunol 2020; 11: 280.
Setta-Kaffetzi N, Simpson MA, Navarini AA, et al. AP1S3 Mutations Are Associated with Pustular Psoriasis and Impaired Toll-like Receptor 3 Trafficking. Am J Hum Genet 2014; 94: 790–7.
Naranbhai V, Fairfax BP, Makino S, et al. Genomic modulators of gene expression in human neutrophils. Nat Commun 2015; 6: 7545.
Mahil SK, Twelves S, Farkas K, et al. AP1S3 mutations cause skin autoinflammation by disrupting keratinocyte autophagy and up-regulating IL-36 production. J Invest Dermatol 2016; 136: 2251–9.
Bachelez H. Pustular Psoriasis: The Dawn of a New Era. Acta Derm Venereol 2020; 100: adv00034. doi: https://doi.org/10.2340/00015555-3388 [Epub ahead of print].
Bachelez H. Pustular psoriasis and related pustular skin diseases. Br J Dermatol 2018; 178.
Brenner M, Molin S, Ruebsam K, Weisenseel P, Ruzicka T, Prinz JC. Generalized pustular psoriasis induced by systemic glucocorticosteroids: four cases and recommendations for treatment. Br J Dermatol 2009;161:964–6.
Georgala S, Koumantaki E, Rallis E, Papadavid E. Generalized pustular psoriasis developing during withdrawal of long-term cyclosporin therapy. Br J Dermatol 2000; 142:1057–8.
Sugiura K, Shoda Y, Akiyama M. Generalized Pustular Psoriasis Triggered by Amoxicillin in Monozygotic Twins with Compound Heterozygous IL36RN Mutations: Comment on the Article by Navarini et al. J Invest Dermatol 2014; 134:578–9.
Ozturk G, Turk BG, Karaca N, et al. Generalized pustular eruptions due to terbinafine. J Toxicol Cutaneous Ocul Toxicol 2012;31:814.
Hoegler KM, John AM, Handler MZ. Generalized pustular psoriasis a review and update on treatment. JEur Acad Dermatol Venereol 2018; 32:1645–51.
Tobin AM, Langan SM, Collins P, Kirby B. Generalized pustular psoriasis (von Zumbusch) following the use of calcipotriol and betamethasone dipropionate ointment: a report of two cases. Clin Exp Dermatol 2009;34:629–30.
Falto-Aizpurua LA, Martin-Garcia RF, Carrasquillo OY, Nevares-Pomales OW, Sánchez-Flores X, Lorenzo-Rios D. Biological therapy for pustular psoriasis: a systematic review. Int J Dermatol 2020; 59: 284–96.
Al-Sharqi A, Jayasekera P, Parslew R. A case of tumour necrosis factor-alpha inhibitor- and rituximab-induced plantar pustular psoriasis that completely resolved with tocilizumab. Br J Dermatol 2014;171:1546–9.
Hay RAS, Pan JY. Paradoxical flare of pustular psoriasis triggered by ustekinumab, which responded to adalimumab therapy. Clin Exp Dermatol 2014;39:751–2.
Dogra S, Bishnoi A, Narang T, Handa S. Secukinumab-induced paradoxical pustular psoriasis. Clin Exp Dermatol 2019; 44: 72–3.
Ohkawara A, Yasuda H, Kobayashi H, Inaba Y, Imamura S. Generalized pustular psoriasis in Japan: Two distinct groups formed by differences in symptoms and genetic background. Acta Derm Venereol 1996; 76: 68–71.
Sbidian E, Eftekahri P, Viguier M, Laroche L, Bachelez H. National Survey of Psoriasis Flares after 2009 Monovalent H1N1/Seasonal Vaccines. Dermatology 2014; 229: 130.
Furue K, Yamamura K, Tsuji G, et al. Highlighting Interleukin-36 Signalling in Plaque Psoriasis and Pustular Psoriasis. Acta Derm Venereol 2018;98:5–13.
Lee EY, Takahashi T, Curk T, Dobnikar J, Gallo RL, Wong GCL. Crystallinity of Double-Stranded RNA-Antimicrobial Peptide Complexes Modulates Toll-Like Receptor 3-Mediated Inflammation. Acs Nano 2017;11:145–55.
Lande R, Botti E, Jandus C, et al. Corrigendum: The antimicrobial peptide LL37 is a T-cell autoantigen in psoriasis. Nat Commun 2014;5:5621.
Ueyama A, Yamamoto M, Tsujii K, et al. Mechanism of pathogenesis of imiquimod-induced skin inflammation in the mouse: A role for interferon-alpha in dendritic cell activation by imiquimod. J. Dermatol 2014;41:135–43.
Sumida H, Yanagida K, Kita Y, et al. Interplay between CXCR2 and BLT1 Facilitates Neutrophil Infiltration and Resultant Keratinocyte Activation in a Murine Model of Imiquimod-Induced Psoriasis. J Immunol 2014;192:4361–9.
Saggini A, Chimenti S, Chiricozzi A. IL-6 as a Druggable Targetin Psoriasis: Focus on Pustular Variants. J Immunol Res 2014;2014: 1–10.
Danesh M, Pomeranz MK, McMeniman E, Murase JE. Dermatoses of Pregnancy: Nomenclature, Misnomers, and Myths. Clin Dermatol 2016;34:314–9.
Flynn A, Burke N, Byrne B, Gleeson N, Wynne B, Barnes L. Two case reports of generalized pustular psoriasis of pregnancy: Different outcomes. Obstet Med 2016;9:55–9.
Shaw CJ, Wu P, Sriemevan A. First trimester impetigo herpetiformis in multiparous female successfully treated with oral cyclosporine. BMJ Case Rep 2011; 2011: bcr0220113915.
Wolf R, Tartler U, Stege H, Megahed M, Ruzicka T. Impetigo herpetiformis with hyperparathyroidism. J Eur Acad Dermatol Venereol 2005; 19:743–6.
Sugiura K, Oiso N, Linuma S, et al. IL36RN mutations underlie impetigo herpetiformis. J Invest Dermatol 2014; 134: 2472–4.
Zeng YP, Liu J, Qu T, et al. Clinical Characteristics of Impetigo Herpetiformis in 14 Patients. Med J PUMCH 2012;3:415–8.
Lee Y, Nam YH, Lee JH, Park JK, Seo YJ. Hypocalcaemia-induced pustular psoriasis-like skin eruption. Br J Dermatol 2005; 152:591–3.
Bikle DD, Xie Z, Tu C-L. Calcium regulation of keratinocyte differentiation. Expert Rev Endocrinol Metab 2012; 7: 461–72.
Staberg B, Oxholm A, Klemp P, Christiansen C. Abnormal vitamin D metabolism in patients with psoriasis. Acta Derm Venereol 1987; 67:65–8.
Bechinger B, Gorr SU. Antimicrobial Peptides: Mechanisms of Action and Resistance. J Dent Res 2017;96:254–60.
Zasloff M. Antimicrobial peptides of multicellular organisms. Nature 2002;415:389–95.
Schauber J, Dorschner RA, Yamasaki K, Brouha B, Gallo RL. Control of the innate epithelial antimicrobial response is cell-type specific and dependent on relevant microenvironmental stimuli. Immunology 2006;118:509–19.
Sylwia R. Cathelicidin LL-37 Affects Surface and Intracellular Toll-Like Receptor Expression in Tissue Mast Cells. J Immunol Res 2018;2018:1–18.
Bocchinfuso G, Palleschi A, Orioni B, et al. Different mechanisms of action of antimicrobial peptides: insights from fluorescence spectroscopy experiments and molecular dynamics simulations. J Pept Sci 2009;15:550–8.
Scott A, Weldon S, Buchanan PJ, et al. Evaluation of the Ability of LL-37 to Neutralise LPS In Vitro and Ex Vivo. PLoS One 2011;6:e26525.
Babolewska E, Brzezinska-Blaszczyk E. Human-derived cathelicidin LL-37 directly activates mast cells to proinflammatory mediator synthesis and migratory response. Cell Immunol 2015; 293: 67–73.
Hancock REW. The Human Antimicrobial Peptide LL-37 Is a Multifunctional Modulator of Innate Immune Responses. J Immunol 2002;169:3883–91.
Neumann A, Berends ET, Nerlich A, et al. The antimicrobial peptide LL-37 facilitates the formation of neutrophil extracellular traps. Biochem J 2014; 464:3–11.
Albanesi C, Madonna S, Gisondi P, Girlomoni G. The Interplay Between Keratinocytes and Immune Cells in the Pathogenesis of Psoriasis. Front Immunol 2018; 9: 1549.
Vandamme D, Landuyt B, Luyten W, Schoofs L. A comprehensive summary of LL-37, the factotum human cathelicidin peptide. Cell Immunol 2012;280:22–35.
Schön MP. Adaptive and Innate Immunity in Psoriasis and Other Inflammatory Disorders. Front Immunol 2019; 10: 1764.
Yang D, Han Z, Oppenheim JJ. Alarmins and immunity. Immunol Rev 2017;280:41–56.
Schauber J, Dorschner RA, Coda AB, et al. Injury enhances TLR2 function and antimicrobial peptide expression through a vitamin D-dependent mechanism. J Clin Invest 2007; 117: 803–11.
Na L, Yamasaki K, Saito R, et al. Alarmin Function of Cathelicidin Antimicrobial Peptide LL37 through IL-36γ Induction in Human Epidermal Keratinocytes. J Immunol 2014; 193:5140–8.
Wang L, Yu X, Chao W, et al. RNA sequencing-based longitudinal transcriptomic profiling gives novel insights into the disease mechanism of generalized pustular psoriasis. BMC Med Genomics 2018; 11: 52.
Liang Y, Xing X, Beamer MA, et al. Six-transmembrane epithelial antigens of the prostate comprise a novel inflammatory nexus in patients with pustular skin disorders. J Allergy Clin Immunol 2017;139:1217–27.
Yu XL, Wu C, Wang WM, Li F, Jin HZ. Interleukin (IL)-8 and IL-36γ but not IL-36Ra are related to acrosyringia in pustule formation associated with palmoplantarpustulosis. Clin Exp Dermatol 2019;44: 52–7.
Nestle FO. Plasmacytoid dendritic cells sense self-DNA coupled with antimicrobial peptide. Nature 2007; 449: 564–9.
Gallo RL. Cathelicidin Antimicrobial Peptide LL-37 in Psoriasis Enables Keratinocyte ReactivityagainstTLR9 Ligands. J InvestDermatol 2012;132:135–43.
Ganguly D, Chamilos G, Lande R, et al. Self-RNA-antimicrobial peptide complexes activate human dendritic cells through TLR7 and TLR8. J Exp Med 2009; 206:1983–94.
Terhorst D, Chelbi R, Wohn C, et al. Dynamics and Transcriptomics of Skin Dendritic Cells and Macrophages in an Imiquimod-Induced, Biphasic Mouse Model of Psoriasis. J Immunol 2015;195:4953–61.
Lande R, Botti E, Jandus C, et al. The antimicrobial peptide LL37 is a T-cell autoantigen in psoriasis. Nat Commun 2014; 5: 5621.
Bell TD, Demay MB, Burnett-Bowie SAM. The biology and pathology of vitamin D control in bone. J Cell Biochem 2010; 111: 7–13.
Henry HL. Regulation of vitamin D metabolism. Best Pract Res Clin Endocrinol Metab 2011; 25:531–41.
Barrea L, Savanelli MC, Di Somma C, et al. Vitamin D and its role in psoriasis: An overview of the dermatologist and nutritionist. Rev Endocr Metab Disord 2017; 18: 195–205.
Ruan X, Tey HL. Hypocalcemia: low incidence in flares of pustular and chronic plaque psoriasis. Int J Dermatol 2017; 56: e133–5.
Knuever J, Tantcheva-Poor I. Generalized pustular psoriasis: A possible association with severe hypocalcaemia due to primary hypoparathyroidism. J Dermatol 2017;44: 1416–7.
Bikle DD, Xie ZJ, Tu CL. Calcium regulation of keratinocyte differentiation. Expert Rev Endocrinol Metab 2012; 7: 461–72.
Bikle DD, Oda Y, Xie Z. Calcium and 1,25(OH)2D: interacting drivers of epidermal differentiation. J. Steroid Biochem. Mol Biol 2004; 89–90:355–60.
Zhang ZH. 1,25(OH) 2 D 3 inhibited Th17 cells differentiation via regulating the NF-kB activity and expression of IL-17. Cell Prolif 2018;51:e12461.
Navarro-Triviño FJ, Arias-Santiago S, Gilaberte-Calzada Y. Vitamin D and the Skin: A Review for Dermatologists. Actas Dermo Sifiliográficas 2019; 110: 262–72.
Stewart AF, Battaglini-Sabetta J, Millstone L. Hypocalcemia-Induced Pustular Psoriasis of von Zumbusch. Ann Intern Med 1984; 100: 677–80.
Carmona-Rivera C, Kaplan MJ. Low-density granulocytes: a distinct class of neutrophils in systemic autoimmunity. Semin Immunopathol 2013;35:455–63.
Villanueva E, Yalavarthi S, Berthier CC, et al. Netting Neutrophils Induce Endothelial Damage, Infiltrate Tissues, and Expose Immunos-timulatory Molecules in Systemic Lupus Erythematosus. J Immunol 2011;187:538–52.
Lin AM, Rubin CJ, Khandpur R, et al. Mast Cells and Neutrophils Release IL-17 through Extracellular Trap Formation in Psoriasis. J Immunol 2011; 187:490–500.
Chen K, Bao Z, Gong W, Tang P, Yoshimura T, Wang JM. Regulation of inflammation by members of the formyl-peptide receptor family. J Autoimmun 2017; 85:64–77.
Elssner A, Duncan M, Gavrilin M, Wewers MD. A Novel P2X7 Receptor Activator, the Human Cathelicidin-Derived Peptide LL37, Induces IL-1 beta Processing and Release. J Immunol 2004; 172: 4987–94.
Takematsu H, Ohkohchi K, Tagami H. Demonstration of anaphylatoxins C3a, C4a and C5a in the scales of psoriasis and inflammatory pustular dermatoses. Br J Dermatol 1986; 114:1–6.
Kanazawa N, Nakamura T, Mikita N, Furukawa F. Novel IL36RN mutation in a Japanese case of early onset generalized pustular psoriasis. J Dermatol 2013; 40: 749–51.
Sugiura K, Takeichi T, Kono M, et al. A novel IL36RN/IL1F5 homozygous nonsense mutation, p.Arg10X, in a Japanese patient with adult-onset generalized pustular psoriasis. Br J Dermatol 2012; 167:699–701.
Funding
Funding: CAMS Initiative for Innovative Medicine (2017-I2M-3-020), CAMS Initiative for Innovative Medicine (2017-I2M-B&R-01). National Natural Science Foundation of China (81773331).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Wang, H., Jin, H. Update on the aetiology and mechanisms of generalized pustular psoriasis. Eur J Dermatol 31, 602–608 (2021). https://doi.org/10.1684/ejd.2021.4047
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1684/ejd.2021.4047