Abstract
Background
Recent studies have shown that vitiligo is a T-cell mediated autoimmune disease. Skin-homing cytotoxic T lymphocytes expressing cutaneous lymphocyte-associated antigen (CLA) have been suggested to be responsible for the destruction of melanocytes in vitiligo. An aberration in the suppressive function of regulatory T cells (Tregs) has been reported in vitiligo patients. However, whether the weakened suppressive ability of the Tregs contributes to hyper-activated skin homing CD8+CLA+ T cells remains to be determined.
Objectives
To investigate the inhibition of circulating Tregs on the proliferation of autologous CD8+CLA+ T cells in non-segmental vitiligo patients.
Methods
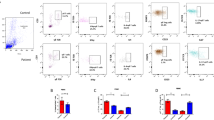
CD8+CLA+ T cells and Tregs were obtained from the peripheral blood of 13 non-segmental vitiligo patients and 7 controls. The proliferative responses of CD8+CLA+ T cells were assessed in the absence or presence of autologous Tregs, and the levels of Transforming Growth Factor β1(TGF-β1) and IL-10 in culture supernatants were detected by enzyme-linked immunosorbent assay.
Results
The proliferative responses of circulating CD8+CLA+ T cells in the presence of Tregs were significantly higher in the active vitiligo than in the stable vitiligo and control groups. Tregs from active vitiligo patients exhibited a lower inhibitory effect on proliferation of CD8+CLA+ T cells. The levels of TGF-β1 produced by Tregs were significantly lower in active vitiligo than other groups and anti-TGF-β1 antibodies could abrogate the suppressive function of Tregs.
Conclusions
The functional activity of Tregs is compromised in active vitiligo patients. TGF-β1 plays an important role in the autoimmune mechanism of the disease.




Similar content being viewed by others
References
Huggins RH, Schwartz RA, Janniger C. Vitiligo. Acta Dermatovenerol Alp Panonica Adriat 2005; 14: 137–45.
Le Poole IC, Wañkowicz-Kaliñska A, van den Wijngaard RM, Nickoloff BJ, Das PK. Autoimmune aspects of depigmentation in vitiligo. J Investig Dermatol Symp Proc 2004; 9: 68–72.
Glassman SJ. Vitiligo, reactive oxygen species and T-cells. Clin Sci (Lond) 2011; 120: 99–120.
Le Gal FA, Avril MF, Bosq J, et al. Direct evidence to support the role of antigen-specific CD8(+) T cells in melanoma-associated vitiligo. J Invest Dermatol 2001; 117: 1464–70.
Palermo B, Campanelli R, Garbelli S, et al. Specific cytotoxic T lymphocyte responses against Melan-A/MART1, tyrosinase and gp100 in vitiligo by the use of major histocompatibility complex/peptide tetramers: the role of cellular immunity in the etiopathogenesis of vitiligo. J Invest Dermatol 2001; 117: 326–32.
Dwivedi M, Laddha NC, Arora P, Marfatia YS, Begum R. Decreased regulatory T-cells and CD4(+) /CD8(+) ratio correlate with disease onset and progression in patients with generalized vitiligo. Pigment Cell Melanoma Res 2013; 26: 586–91.
Lang KS, Caroli CC, Muhm A, et al. HLA-A2 restricted, melanocyte-specific CD8(+) T lymphocytes detected in vitiligo patients are related to disease activity and are predominantly directed against MelanA/MART1. J Invest Dermatol 2001; 116: 891–7.
Mandelcorn-Monson RL, Shear NH, Yau E, et al. Cytotoxic T lymphocyte reactivity to gp100, MelanA/MART-1, and tyrosinase, in HLA-A2-positive vitiligo patients. J Invest Dermatol 2003; 121: 550–6.
Ogg GS, Rod DP, Romero P, Chen JL, Cerundolo V. High frequency of skin-homing melanocyte-specific cytotoxic T lymphocytes in autoimmune vitiligo. J Exp Med 1998; 188: 1203–8.
van den Boorn JG, Konijnenberg D, Dellemijn TA, et al. Autoimmune destruction of skin melanocytes by perilesional T cells from vitiligo patients. J Invest Dermatol 2009; 129: 2220–32.
van den WR, Wankowicz-Kalinska A, Le PC, Tigges B, Westerhof W, Das P. Local immune response in skin of generalized vitiligo patients. Destruction of melanocytes is associated with the prominent presence of CLA+ T cells at the perilesional site. Lab Invest 2000; 80: 1299–309.
Wu J, Zhou M, Wan Y, Xu A. CD8+ T cells from vitiligo perilesional margins induce autologous melanocyte apoptosis. Mol Med Rep 2013; 7: 237–41.
Zhang BX, Lin M, Qi XY, et al. Characterization of circulating CD8+T cells expressing skin homing and cytotoxic molecules in active non-segmental vitiligo. Eur J Dermatol 2013; 23: 331–8.
Dubois B, Chapat L, Goubier A, Kaiserlian D, CD4+CD25+ D. T cells as key regulators of immune responses. Eur J Dermatol 2003; 13: 111–6.
Ben AM, Zaraa I, Rekik R, et al. Functional defects of peripheral regulatory T lymphocytes in patients with progressive vitiligo. Pigment Cell Melanoma Res 2012; 25: 99–109.
Lili Y, Yi W, Ji Y, Yue S, Weimin S, Ming L. Global activation of CD8+ cytotoxic T lymphocytes correlates with an impairment in regulatory T cells in patients with generalized vitiligo. PLoS One 2012; 7: e37513-.
Richetta A, D’Epiro S, Salvi M, et al. Serum levels of functional T-regs in vitiligo: our experience and mini-review of the literature. Eur J Dermatol 2013; 23: 154–9.
Nakamura K, Kitani A, Strober W. Cell contact-dependent immunosuppression by CD4(+)CD25(+) regulatory T cells is mediated by cell surface-bound transforming growth factor beta. J Exp Med 2001; 194: 629–44.
Thornton AM, Shevach EM. CD4+CD25+ immunoregulatory T cells suppress polyclonal T cell activation in vitro by inhibiting interleukin 2 production. J Exp Med 1998; 188: 287–96.
Tu CX, Jin WW, Lin M, Wang ZH, Man MQ. Levels of TGF-beta(1) in serum and culture supernatants of CD4(+)CD25 (+) T cells from patients with non-segmental vitiligo. Arch Dermatol Res 2011; 303: 685–9.
Yano S, Nakamura K, Okochi H, Tamaki K. Analysis of the expression of cutaneous lymphocyte-associated antigen on the peripheral blood and cutaneous lymphocytes of alopecia areata patients. Acta Derm Venereol 2002; 82: 82–5.
Sigmundsdottir H, Gudjonsson JE, Jonsdottir I, Ludviksson BR, Valdimarsson H. The frequency of CLA+ CD8+ T cells in the blood of psoriasis patients correlates closely with the severity of their disease. Clin Exp Immunol 2001; 126: 365–9.
Gregg RK, Nichols L, Chen Y, Lu B, Engelhard VH. Mechanisms of spatial and temporal development of autoimmune vitiligo in tyrosinase-specific TCR transgenic mice. J Immunol 2010; 184: 1909–17.
Hilchey SP, Bernstein SH. Use of CFSE to monitor ex vivo regulatory T-cell suppression of CD4+ and CD8+ T-cell proliferation within unseparated mononuclear cells from malignant and non-malignant human lymph node biopsies. Immunol Invest 2007; 36: 629–48.
Antiga E, Kretz CC, Klembt R, et al. Characterization of regulatory T cells in patients with dermatomyositis. J Autoimmun 2010; 35: 342–50.
Miyara M, Amoura Z, Parizot C, et al. Global natural regulatory T cell depletion in active systemic lupus erythematosus. J Immunol 2005; 175: 8392–400.
Klarquist J, Denman CJ, Hernandez C, et al. Reduced skin homing by functional Treg in vitiligo. Pigment Cell Melanoma Res 2010; 23: 276–86.
Oida T, Xu L, Weiner HL, Kitani A, Strober W. TGF-beta-mediated suppression by CD4+CD25+ T cells is facilitated by CTLA-4 signaling. J Immunol 2006; 177: 2331–9.
Fahlen L, Read S, Gorelik L, et al. T cells that cannot respond to TGF-beta escape control by CD4(+)CD25(+) regulatory T cells. J Exp Med 2005; 201: 737–46.
von BH. Mechanisms of suppression by suppressor T cells. Nat Immunol 2005; 6: 338–44.
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
About this article
Cite this article
Lin, M., Zhang, BX., Shen, N. et al. Regulatory T cells from active non-segmental vitiligo exhibit lower suppressive ability on CD8+CLA+ T cells. Eur J Dermatol 24, 676–682 (2014). https://doi.org/10.1684/ejd.2014.2436
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1684/ejd.2014.2436