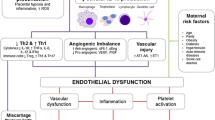
Abstract
Special hormonal and immunological changes are required for normal pregnancy continuation. To escape from rejection by the maternal immune system, pregnancy needs an optimum environment with the integration and the balance of immune factors. As an immunologically unique site that permits allogenic fetus to be tolerated by mother, the maternal–fetal interface has a vital role. Microorganisms may trigger innate immune responses at the maternal–fetal interface and this may have a significant impact on the success of pregnancy. While the presence of inflammatory markers are slightly increased in healthy pregnancies, their significant increase in preeclampsia suggests that the balance between the inflammatory and antiinflammatory mechanisms may be disrupted by a shift towards inflammation. Based on these immunological observations, we aimed to review the literature for the link between the inflammatory response and preeclampsia since its etiology has not yet been clarified.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bonney EA. Immune regulation in pregnancy: a matter of perspective? Obstet Gynecol Clin North Am 2016; 43(4):679–98.
Kutteh WH, Stanic AK, Schust DJ. Immunology and reproduction. Yen and Jaffe’s reproductive endocrinology (eighth edition) physiology, pathophysiology and clinical management 2019; 13: 301–321.e3.
Mor G, Cardenas I, Abrahams V, Guller S. Inflammation and pregnancy: the role of the immune system at the implantation site. Ann N Y Acad Sci 2011; 1221(1):80–7.
Ander SE, Diamond MS, Coyne CB. Immune responses at the maternal-fetal interface. Sci Immunol 2019; 4(31).
Romero R, Espinoza J, Kusanovic JP, et al. The preterm parturition syndrome. BJOG 2006; 113(Suppl 3):17–42.
Romero R, Espinoza J, Gonçalves LF, Kusanovic JP, Friel LA, Nien JK. Inflammation in preterm and term labour and delivery. Semin Fetal Neonatal Med 2006; 11: 317–26.
Kamity R, Sharma S, Hanna N. MicroRNA-mediated control of inflammation and tolerance in pregnancy. Front Immunol 2019; 10: 718.
Phillips TA, Ni J, Pan G, et al. TRAIL (Apo-2L) and TRAIL receptors in human placentas: implications for immune privilege. J Immunol 1999; 162: 6053.
Phillips TA, Ni J, Hunt JS. Death-inducing tumour necrosis factor (TNF) superfamily ligands and receptors are transcribed in human placentae, cytotrophoblasts, placental macrophages and placental cell lines. Placenta 2001; 22: 663.
Austgulen R, Johnsen H, Kjøllesdal AM, et al. Soluble receptors for tumor necrosis factor: occurrence in association with normal delivery at term. Obstet Gynecol 1993; 82: 343.
Payne SG, Smith SC, Davidge ST, et al. Death receptor Fas/Apo-1/CD95 expressed by human placental cytotrophoblasts does not mediate apoptosis. Biol Reprod 1999; 60: 1144.
Yao Y, Xu XH, Jin L. Macrophage polarization in physiological and pathological pregnancy. Front Immunol 2019; 10: 792.
Somerset DA, Zheng Y, Kilby MD, et al. Normal human pregnancy is associated with an elevation in the immune suppressive CD25+ CD4+ regulatory T cell subset. Immunology 2004; 112: 38.
Aluvihare VR, Kallikourdis M, Betz AG. Regulatory T cells mediate maternal tolerance to the fetus. Nat Immunol 2004; 5: 266.
Zenclussen AC, Gerlof K, Zenclussen ML, et al. Abnormal T cell reactivity against paternal antigens in spontaneous abortion: adoptive transfer of pregnancy induced CD4 + CD25+ T regulatory cells prevents fetal rejection in a murine abortion model. Am J Pathol 2005; 166: 811.
Schumacher A, Wafula PO, Bertoja AZ, et al. Mechanisms of action of regulatory T cells specific for paternal antigens during pregnancy. Obstet Gynecol 2007; 110: 1137.
Rolle L, Memarzadeh Tehran M, MorellGarcía A, et al. Cutting edge: IL10 producing regulatory B cells in early human pregnancy. Am J Reprod Immunol 2013; 70: 448.
Ober C, Karrison T, Odem RR, et al. Mononuclear cell immunisation in prevention of recurrent miscarriages: a randomised trial. Lancet 1999; 354: 365.
Lu HQ, Hu R. The role of immunity in the pathogenesis and development of pre-eclampsia. Scand J Immunol 2019; 90(5): e12756.
Amash A, Weintraub AY, Sheiner E, Zeadna A, Huleihel M, Holcberg G. Possible therapeutic effect of magnesium sulfate in pre-eclampsia by the down-regulation of placental tumor necrosis factor-alpha secretion. Eur Cytokine Netw 2010; 21 (1):58–64.
Santulli G, Al-Mallah MH. Pre-eclampsia and future cardiovascular diseases: how to assess the risk? Atherosclerosis 2019; 290: 136–7.
Kushner I. The phenomenon of the acute phase response. Ann N Y Acad Sci 1982; 389: 39.
Raio L, Bersinger NA, Malek A, et al. Ultra-high sensitive C-reactive protein during normal pregnancy and in preeclampsia: a pilot study. J Hypertens 2019; 37(5):1012–7.
Schmidt-Arras D, Rose-John S. IL-6 pathway in the liver: from physiopathology to therapy. J Hepatol 2016; 64(6):1403–15.
Nichols DC, Flannery AH, Magnuson BL, Cook AM. Prealbumin is associated with in-hospital mortality in critically ill patients. Nutr Clin Pract 2019;. [Epub ahead of print].
Syriou V, Papanikolaou D, Kozyraki A, Goulis DG. Cytokines and male infertility. Eur Cytokine Netw 2018; 29(3):73–8.
Stavropoulos-Kalinoglou A, Kitas GD. Could IL-6 inhibition prevent exercise-induced fat loss in RA? Nat Rev Rheumatol 2019; 15: 192–4.
Sproston NR, Ashworth JJ. Role of C-reactive protein at sites of inflammation and infection. Front Immunol 2018; 9: 754.
Mohaupt MG. C-reactive protein and its role in preeclampsia. Hypertension 2015; 65(2):285–6.
Kara AE, Guney G, Tokmak A, Ozaksit G. The role of inflammatory markers hs-CRP, sialic acid and IL-6 in the pathogenesis of preeclampsia and intrauterine growth restriction. Eur Cytokine Netw 2019; 30(1):29–33.
Ali Z, Bokhari FA, Zaki S, Zargham U, Tauseef A, Khakan S. Correlation of CRP levels in third trimester with fetal birth weight in preeclamptic and normotensive pregnant women. J Coll Physicians Surg Pak 2015; 25(2):111–4.
Gandevani SB, Banaem LM, Mohamadi B, Moghadam NA, Asghari M. Association of high-sensitivity C-reactive protein serum levels in early pregnancy with the severity of preeclampsia and fetal birth weight. J Perinat Med 2012; 40 (6):601–5.
Chen H, Zhang J, Qin F, Chen X, Jiang X. Evaluation of the predictive value of high sensitivity C-reactive protein in pregnancy-induced hypertension syndrome. Exp Ther Med 2018; 16(2):619–22.
Rebelo F, Schlüssel MM, Vaz JS, et al. C-reactive protein and later preeclampsia: systematic review and meta-analysis taking into account the weight status. J Hypertens 2013; 31(1):16–26.
Jannesari R, Kazemi E. Level of high sensitive C-reactive protein and procalcitonin in pregnant women with mild and severe preeclampsia. Adv Biomed Res 2017; 6: 140.
Mihu D, Costin N, Mihu CM, Blaga LD, Pop RB. C-reactive protein, marker for evaluation of systemic inflammatory response in preeclampsia. Rev Med Chir Soc Med Nat Iasi 2008; 112(4):1019–25.
Stefanović M, Vukomanović P, Milosavljević M, Kutlesić R, Popović J, Tubić-Pavlović A. Insulin resistance and C-reactive protein in preeclampsia. Bosn J Basic Med Sci 2009; 9(3):235–8.
Von Versen-Hoeynck FM, Hubel CA, Gallaher MJ, Gammill HS, Powers RW. Plasma levels of inflammatory markers neopterin, sialic acid, and C-reactive protein in pregnancy and preeclampsia. Am J Hypertens 2009; 22(6):687–92.
Aalami-Harandi R, Karamali M, Asemi Z. The favorable effects of garlic intake on metabolic profiles, hs-CRP, biomarkers of oxidative stress and pregnancy outcomes in pregnant women at risk for pre-eclampsia: randomized, double-blind, placebo-controlled trial. J Matern Fetal Neonatal Med 2015; 28(17):2020–7.
Guven MA, Coskun A, Ertas IE, Aral M, Zencirci B, Oksuz H. Association of maternal serum CRP, IL-6, TNF-alpha, homocysteine, folic acid and vitamin B12 levels with the severity of preeclampsia and fetal birth weight. Hypertens Pregnancy 2009; 28(2):190–200.
Ertas IE, Kahyaoglu S, Yilmaz B, et al. Association of maternal serum high sensitive C-reactive protein level with body mass index and severity of pre-eclampsia at third trimester. J Obstet Gynaecol Res 2010; 36(5):970–7.
Kashanian M, Aghbali F, Mahali N. Evaluation of the diagnostic value of the first-trimester maternal serum high-sensitivity C-reactive protein level for prediction of pre-eclampsia? J Obstet Gynaecol Res 2013; 39(12):1549–54.
Farzadnia M, Ayatollahi H, Hasan-Zade M, Rahimi HR. A comparative study of serum level of vascular cell adhesion molecule-1 (sVCAM-1), intercellular adhesion molecule-1 (ICAM-1) and high sensitive C — reactive protein (hs-CRP) in normal and pre-eclamptic pregnancies. Iran J Basic Med Sci 2013; 16(5):689–93.
Swellam M, Samy N, Wahab SA, Ibrahim MS. Emerging role of endothelial and inflammatory markers in preeclampsia. Dis Markers 2009; 26(3):127–33.
García RG, Celedón J, Sierra-Laguado J, et al. Raised C-reactive protein and impaired flow-mediated vasodilation precede the development of preeclampsia. Am J Hypertens 2007; 20(1):98–103.
Belo L, Santos-Silva A, Caslake M, et al. Neutrophil activation and C-reactive protein concentration in preeclampsia. Hypertens Pregnancy 2003; 22(2):129–41.
Qiu C, Luthy DA, Zhang C, Walsh SW, Leisenring WM, Williams MA. A prospective study of maternal serum C-reactive protein concentrations and risk of preeclampsia. Am J Hypertens 2004; 17(2):154–60.
Mihalceanu E, Nemescu D, Gavrilu¸t M, Dimitriu DC, Pangal A, Onofriescu M. The correlation between markers of systemic inflammation and angiogenic markers in pre-eclampsia. Rev Med Chir Soc Med Nat Iasi 2015; 119(2):473–83.
Ouyang YQ, Li SJ, Zhang Q, Cai HB, Chen HP. Interactions between inflammatory and oxidative stress in preeclampsia. Hypertens Pregnancy 2009; 28(1):56–62.
Sarween N, Drayson MT, Hodson J, et al. Humoral immunity in late-onset pre-eclampsia and linkage with angiogenic and inflammatory markers. Am J Reprod Immunol 2018; 80(5): e13041.
Kucukgoz Gulec U, Tuncay Ozgunen F, Baris Guzel A, et al. An analysis of C-reactive protein, procalcitonin and D-dimer in pre-eclamptic patients. Am J Reprod Immunol 2012; 68(4):331–7.
Best LG, Saxena R, Anderson CM, et al. Two variants of the C-reactive protein gene are associated with risk of pre-eclampsia in an American Indian population. PLoS One 2013; 8(8): e71231.
Paternoster DM, Fantinato S, Stella A, et al. C-reactive protein in hypertensive disorders in pregnancy. Clin Appl Thromb Hemost 2006; 12(3):330–7.
Jääskeläinen T, Heinonen S, Hämäläinen E, Pulkki K, Romppanen J, Laivuori H. Impact of obesity on angiogenic and inflammatory markers in the Finnish Genetics of Pre-eclampsia Consortium (FINNPEC) cohort. Int J Obes 2019; 43 (5):1070–81.
Srinivas SK, Sammel MD, Bastek J, et al. Evaluating the association between all components of the metabolic syndrome and pre-eclampsia. J Matern Fetal Neonatal Med 2009; 22 (6):501–9.
Gammill HS, Powers RW, Clifton RG, et al. Does C-reactive protein predict recurrent preeclampsia? Hypertens Pregnancy 2010; 29(4):399–409.
Donker RB, Molema G, Faas MM, et al. Absence of in vivo generalized pro-inflammatory endothelial activation in severe, early-onset preeclampsia. J Soc Gynecol Investig 2005; 12 (7):518–28.
Ustün Y, Engin-Ustün Y, Kamaci M. Association of fibrinogen and C-reactive protein with severity of preeclamp-sia. Eur J Obstet Gynecol Reprod Biol 2005; 121(2):154–8.
Tjoa ML, van Vugt JM, Go AT, Blankenstein MA, Oudejans CB, van Wijk IJ. Elevated C-reactive protein levels during first trimester of pregnancy are indicative of preeclampsia and intrauterine growth restriction. J Reprod Immunol 2003; 59 (1):29–37.
Chekaoui A, Lahmar K, Belguendouz H, et al. Increased IL-1b levels are associated with an imbalance of “oxidant/antioxi-dant” status during Behçet’s disease. Eur Cytokine Netw 2018; 29(3):95–102.
Duan L, Liu Z, Wang L, et al. C1q and tumor necrosis factor related protein 4 (CTRP4) suppresses caspase-1/IL-1b inflammatory pathway in trophoblasts of rat models with preeclamp-sia. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi 2016; 32(11):1441–5.
Wang X, Jiang F, Liang Y, et al. Interleukin-1b-31C/T and -511T/C polymorphisms were associated with preeclampsia in Chinese Han population. PLoS One 2014; 9(9):e106919.
Weel I, Romão-Veiga M, Matias ML, et al. Increased expression of NLRP3 inflammasome in placentas from pregnant women with severe preeclampsia. J Reprod Immunol 2017; 123: 40–7.
Van Rijn BB, Bruinse HW, Veerbeek JH, et al. Postpartum circulating markers of inflammation and the systemic acute-phase response after early-onset preeclampsia. Hypertension 2016; 67(2):404–14.
Mulla MJ, Myrtolli K, Potter J, et al. Uric acid induces trophoblast IL-1b production via the inflammasome: implications for the pathogenesis of preeclampsia. Am J Reprod Immunol 2011; 65(6):542–8.
Matias ML, Romão M, Weel IC, et al. Endogenous and uric acid-induced activation of NLRP3 inflammasome in pregnant women with preeclampsia. PLoS One 2015; 10(6):e0129095.
WangJ, Huang Y, Huang Y, Zhou J, Liu X. Effect of lipoxin A on IL-1b production of monocytes and its possible mechanism in severe preeclampsia. J Huazhong Univ Sci Technolog Med Sci 2010; 30(6):767–70.
Alvarez-Cabrera MC, Barrientos-Galeana E, Barrera-García A, et al. Secretion of heat shock -60, -70 kD protein, IL-1b and TNFa levels in serum of a term normal pregnancy and patients with pre-eclampsia development. J Cell Mol Med 2018; 22(11):5748–52.
Giorgi VS, Witkin SS, Bannwart-Castro CF, et al. Elevated circulatingadenosine deaminase activity in women with preeclampsia: association with pro-inflammatory cytokine production and uric acid levels. Pregnancy Hypertens 2016; 6 (4):400–5.
Romão-Veiga M, Matias ML, Ribeiro VR, et al. Induction of systemic inflammation by hyaluronan and hsp70 in women with pre-eclampsia. Cytokine 2018; 105: 23–31.
Alanbay I, Coksuer H, Ercan CM, et al. Chitotriosidase, interleukin-1 beta and tumor necrosis factor alpha levels in mild preeclampsia. Arch Gynecol Obstet 2012; 285(6):1505–11.
Kalinderis M, Papanikolaou A, Kalinderi K, et al. Elevated serum levels of interleukin-6, interleukin-1b and human chorionic gonadotropin in pre-eclampsia. Am J Reprod Immunol 2011; 66(6):468–75.
Amash A, Holcberg G, Sapir O, Huleihel M. Effect placental secretion of interleukin-1 and interleukin-1 receptor antagonist in preeclampsia of magnesium sulfate. J Interferon Cytokine Res 2012; 32(9):432–41.
Liu L, Zhang Y, Wang Y, Peng W, Zhang N, Ye Y. Progesterone inhibited endoplasmic reticulum stress associated apoptosis inducedbyinterleukin-1bvia the GRP78/PERK/CHOP pathway in BeWo cells. J Obstet Gynaecol Res 2018; 44(3):463–73.
Conrad KP, Miles TM, Benyo DF. Circulating levels of immunoreactive cytokines in women with preeclampsia. Am J Reprod Immunol 1998; 40(2):102–11.
Pinheiro MB, Martins-Filho OA, Mota AP, et al. Severe preeclampsia goes along with a cytokine network disturbance towards a systemic inflammatory state. Cytokine 2013; 62 (1):165–73.
Szarka A,, Rigó Jr J, Lázár L, Beko G, Molvarec A. Circulating cytokines, chemokines and adhesion molecules in normal pregnancy and preeclampsia determined by multiplex suspension array. BMC Immunol 2010; 11: 59.
Munno I, Chiechi LM, Lacedra G, et al. Spontaneous and induced release of prostaglandins, interleukin (IL)-1beta, IL-6, and tumor necrosis factor-alpha by placental tissue from normal and preeclamptic pregnancies. Am J Reprod Immunol 1999; 42(6):369–74.
Greer IA, Lyall F, Perera T, Boswell F, Macara LM. Increased concentrations of cytokines interleukin-6 and interleukin-1 receptor antagonist in plasma of women with preeclampsia: a mechanism for endothelial dysfunction? Obstet Gynecol 1994; 84(6):937–40.
Mosimann B, Wagner M, Poon LC, Bansal AS, Nicolaides KH. Maternal serum cytokines at 30–33 weeks in the prediction of preeclampsia. Prenat Diagn 2013; 33(9):823–30.
Benyo DF, Smarason A, Redman CW, Sims C, Conrad KP. Expression of inflammatory cytokines in placentas from women with preeclampsia. J Clin Endocrinol Metab 2001; 86(6):2505–12.
Benyo DF, Miles TM, Conrad KP. Hypoxia stimulates cytokine production by villous explants from the human placenta. J Clin Endocrinol Metab 1997; 82(5):1582–8.
Siljee JE, Wortelboer EJ, Koster MP, et al. Identification of interleukin-1 beta, but no other inflammatory proteins, as an early onset pre-eclampsia biomarker in first trimester serum by bead-based multiplexed immunoassays. Prenat Diagn 2013; 33(12):1183–8.
Moreno-Eutimio MA, Tovar-Rodríguez JM, Vargas-Avila K, et al. Increased serum levels of inflammatory mediators and low frequency of regulatory T cells in the peripheral blood of preeclamptic Mexican women. Biomed Res Int 2014; 2014: 413249.
Lockwood CJ, Huang SJ, Krikun G, et al. Decidual hemostasis, inflammation, and angiogenesis in pre-eclampsia. Semin Thromb Hemost 2011; 37(2):158–64.
Jabalie G, Ahmadi M, Koushaeian L, et al. Metabolic syndrome mediates proinflammatory responses of inflammatory cells in preeclampsia. Am J Reprod Immunol 2019; 81(3): e13086.
Giorgi VS, Peracoli MT, Peracoli JC, Witkin SS, Bannwart-Castro CF. Silibinin modulates the NF-kb pathway and pro-inflammatory cytokine production by mononuclear cells from preeclamptic women. J Reprod Immunol 2012; 95(1–2):67–72.
Van Nieuwenhoven AL, Moes H, Heineman MJ, Santema J, Faas MM. Cytokine production by monocytes, NK cells, and lymphocytes is different in preeclamptic patients as compared with normal pregnant women. Hypertens Pregnancy 2008; 27 (3):207–24.
Kronborg CS, Gjedsted J, Vittinghus E, Hansen TK, Allen J, Knudsen UB. Longitudinal measurement of cytokines in pre-eclamptic and normotensive pregnancies. Acta Obstet Gynecol Scand 2011; 90(7):791–6.
Djurovic S, Clausen T, Wergeland R, Brosstad F, Berg K, Henriksen T. Absence of enhanced systemic inflammatory response at 18 weeks of gestation in women with subsequent pre-eclampsia. BJOG 2002; 109(7):759–64.
Giorgi VS, Bannwart-Castro CF, Peracoli JC, Peracoli MT. PP062 Silibinin modulates NF-kB pathway and proinflamma-tory cytokines production by mononuclear cells of preeclamp-tic women. Pregnancy Hypertens 2012; 2(3):275–6.
Tanaka T, Narazaki M, Kishimoto T. IL-6 in inflammation, immunity and disease. Cold Spring Harb Perspect Biol 2014; 6 (10):a016295.
Taylor BD, Tang G, Ness RB, et al. Mid-pregnancy circulating immune biomarkers in women with preeclampsia and normotensive controls. Pregnancy Hypertens 2016; 6: 72–8.
Bernardi FC, Guolo F, Bortolin T, Petronilho F, Dal-Pizzol F. Oxidative stress and inflammatory markers in normal pregnancy and preeclampsia. J ObstetGynaecol Res 2008; 34: 948–51.
Bernardi FC, Felisberto F, Vuolo F, et al. Oxidative damage, inflammation, and toll-like receptor 4 pathway are increased in preeclamptic patients: a case–control study. Oxidat Med Cell Long 2012; 2012: 636419.
Catarino C, Santos-Silva A, Belo L, et al. Inflammatory disturbances in preeclampsia: relationship between maternal and umbilical cord blood. J Pregnancy 2012; 2012: 684384.
Daneva AM, Hadzˇi-Lega M, Stefanovic M. Correlation of the system of cytokines in moderate and severe preeclampsia. Clin Exp Obstet Gynecol 2016; 43: 220–4.
Jonsson Y, Rubèr M, Matthiesen L, et al. Cytokine mapping of sera from women with preeclampsia and normal pregnancies. J Reprod Immunol 2006; 70: 83–91.
Luppi P, Deloia JA. Monocytes of preeclamptic women spontaneously synthesize pro-inflammatory cytokines. Clin Immunol 2006; 118: 268–75.
Molvarec A, Szarka A, Walentin S, et al. Serum leptin levels in relation to circulating cytokines, chemokines, adhesion molecules and angiogenic factors in normal pregnancy and preeclampsia. Reprod Biol Endocr 2011; 9: 124.
Pinheiro MB, Martins-Filho OA, Mota AP, et al. Severe preeclampsia goes along with a cytokine network disturbance towards a systemic inflammatory state. Cytokine 2013; 62: 165–173.
Sharma A, Satyam A, Sharma JB. Leptin, IL-10 and inflammatory markers (TNF-alpha, IL-6 and IL-8) in pre-eclamptic, normotensive pregnant and healthy non-pregnant women. Am J Reprod Immunol 2007; 58: 21–30.
Silva DM, Marreiro Ddo N, Moita Neto JM, et al. Oxidative stress and immunological alteration in women with preeclamp-sia. Hypertens Pregnancy 2013; 32: 304–11.
Singh A, Sharma D, Raghunandan C, Bhattacharjee J. Role of inflammatory cytokines and eNOS gene polymorphism in pathophysiology of pre-eclampsia. Am J Reprod Immunol 2010; 63: 244–51.
Teran E, Escudero C, Moya W, Flores M, Vallance P, Lopez-Jaramillo P. Elevated C-reactive protein and pro-inflammatory cytokines in Andean women with preeclampsia. Int J Gynaecol Obstet 2001; 75: 243–9.
Toldi G, Bíró E, Szalay B, et al. Soluble urokinase plasminogen activator receptor (suPAR) levels in healthy pregnancy and preeclampsia. Clin Chem Lab Med 2011; 49: 1873–6.
Wang B, Koga K, Osuga Y, et al. High mobility group box 1 (HMGB1) levels in the placenta and in serum in preeclampsia. Am J Reproductive Immunol 2011; 66: 143–8.
Xiao JP, Yin YX, Gao YF, et al. The increased maternal serum levels of IL-6 are associated with the severity and onset of preeclampsia. Cytokine 2012; 60(3):856–60.
Xie F, Hu Y, Speert DP, et al. Toll-like receptor gene polymorphisms and preeclampsia risk: a case–control study and data synthesis. Hypertens Pregnancy 2010; 29: 390–8.
Wu LZ, Xiao XM. Evaluation of the effects of Uncaria rhynchophylla alkaloid extract on LPS-induced preeclampsia symptoms and inflammation in a pregnant rat model. Braz J Med Biol Res 2019; 52(6):e8273.
Ma Y, Ye Y, Zhang J, Ruan CC, Gao PJ. Immune imbalance is associated with the development of preeclampsia. Medicine 2019; 98(14):e15080.
Gu S, Shen H, Zhou Y, et al. Tetramethylpyrazine reduces the consequences of nitric oxide inhibition in pregnant rats. J Cell Physiol 2019; 234(11):19799–806.
Zák P, Souček M. Correlation of tumor necrosis factor alpha, interleukin 6 and interleukin 10 with blood pressure, risk of preeclampsia and low birth weight in gestational diabetes. Physiol Res 2019; 68(3):395–408.
Sljivancanin Jakovljevic T, Kontic-Vucinic O, Nikolic N, Carkic J, Soldatovic I, Milasin J. Glutathione-S-transferase M1 polymorphism and pro-inflammatory cytokines tumour necrosis factor-a and interleukin-1b are associated with preeclamp-sia in Serbian women. Am J Reprod Immunol 2019; 81(5): e13105.
Aggarwal R, Jain AK, Mittal P, Kohli M, Jawanjal P, Rath G. Association of pro- and anti-inflammatory cytokines in preeclampsia. J Clin Lab Anal 2019; 33(4):e22834.
Zhang JY, Cao XX, Wen HX, Zhang HY. Correlation analysis of levels of inflammatory cytokines and nitric oxide in peripheral blood with urine proteins and renal function in patients with gestational hypertension. Exp Ther Med 2019; 17 (1):657–62.
Cottrell JN, Amaral LM, Harmon A, et al. Interleukin-4 supplementation improves the pathophysiology of hypertension in response to placental ischemia in RUPP rats. Am J Physiol Regul Integr Comp Physiol 2019; 316(2):R165–71.
Chen J, Zhao L, Wang D, et al. Contribution of regulatory T cells to immune tolerance and association of microRNA-210 and Foxp3 in preeclampsia. Mol Med Rep 2019; 19(2):1150–8.
Ding H, Dai Y, Lei Y, et al. Upregulation of CD81 in trophoblasts induces an imbalance of Treg/Th17 cells by promoting IL-6 expression in preeclampsia. Cell Mol Immunol 2019; 16(1):302–12.
Eghbal-Fard S, Yousefi M, Heydarlou H, et al. The imbalance of Th17/Treg axis involved in the pathogenesis of preeclampsia. J Cell Physiol 2019; 234(4):5106–16.
Heydarlou H, Eghabl-Fard S, Ahmadi M, et al. Investigation of follicular helper T cells, as a novel player, in preeclampsia. J Cell Biochem 2019; 120(3):3845–52.
Tanz LJ, Stuart JJ, Missmer SA, et al. Cardiovascular biomarkers in the years following pregnancies complicated by hypertensive disorders or delivered preterm. Pregnancy Hypertens 2018; 13: 14–21.
Sagrillo-Fagundes L, Assunção Salustiano EM, Ruano R, Markus RP, Vaillancourt C. Melatonin modulates autophagy and inflammation protecting human placental trophoblast from hypoxia/reoxygenation. J Pineal Res 2018; 65(4):e12520.
Yang MY, Diao ZY, Wang ZY, et al. Pravastatin alleviates lipopolysaccharide-induced placental TLR4 over-activation and promotes uterine arteriole remodeling without impairing rat fetal development. J Biomed Res 2018; 32(4):288–97.
Valencia-Ortega J, Zárate A, Saucedo R, Hernández-Valencia M, Cruz JG, Puello E. Placental Proinflammatory State and Maternal Endothelial Dysfunction in Preeclampsia. Gynecol Obstet Invest 2019; 84(1):12–9.
Sun J, Zhang H, Liu F, Tang D, Lu X. Ameliorative effects of aspirin against lipopolysaccharide-induced preeclampsia-like symptoms in rats by inhibiting the pro-inflammatory pathway. Can J Physiol Pharmacol 2018; 96(11):1084–91.
Ding L, Bai C, Liu Y. Interleukin-6 contributes to myocardial damage in pregnant rats with reduced uterine perfusion pressure. Braz J Med Biol Res 2018; 51(8):e6921.
Ciampa E, Li Y, Dillon S, et al. Cerebrospinal fluid protein changes in preeclampsia. Hypertension 2018; 72(1):219–26.
Krasnyi AM, Gracheva MI, Sadekova AA, et al. Complex analysis of total and fetal DNA and cytokines in blood plasma of pregnant women with preeclampsia. Bull Exp Biol Med 2018; 164(6):721–5.
Bashir SO, Morsy MD, Elkarib AO, et al. Impact of high altitude on maternal serum leptin level and its correlation with oxidative stress and endothelial inflammatory markers in preeclamptic women. Chin J Physiol 2018; 61(1):50–6.
Freitas NA, Santiago LTC, Kurokawa CS, Meira Junior JD, Corrente JE, Rugolo LMSS. Effect of preeclampsia on human milk cytokine levels. J Matern Fetal Neonatal Med 2019; 32 (13):2209–13.
Martinez-Fierro ML, Castruita-De La Rosa C, Garza-Veloz I, et al. Early pregnancy protein multiplex screening reflects circulating and urinary divergences associated with the development of preeclampsia. Hypertens Pregnancy 2018; 37 (1):37–50.
Allam HIG, Masri AAA. The potential therapeutic role of peroxisome proliferatoractivated receptors agonist in pre-eclamptic pregnant rats. J Coll Physicians Surg Pak 2018; 28 (1):31–5.
Kemse N, Sundrani D, Kale A, Joshi S. Maternal micro-nutrients, omega-3 fatty acids and gene expression of angiogenic and inflammatory markers in pregnancy induced hypertension rats. Arch Med Res 2017; 48(5):414–22.
Charkiewicz K, Jasinska E, Goscik J, et al. Angiogenic factor screening in women with mild preeclampsia–New and significant proteins in plasma. Cytokine 2018; 106: 125–30.
Xu J, Gu Y, Sun J, Zhu H, Lewis DF, Wang Y. Reduced CD200 expression is associated with altered Th1/Th2 cytokine production in placental trophoblasts from preeclampsia. Am J Reprod Immunol 2018; 79(1).
Chen Q, Wang Y, Li Y, Zhao M, Nie G. Serum podocalyxin is significantly increased in early-onset preeclampsia and may represent a novel marker of maternal endothelial cell dysfunction. J Hypertens 2017; 35(11):2287–94.
Huda SS, Jordan F, Bray J, et al. Visceral adipose tissue activated macrophage content and inflammatory adipokine secretion is higher in pre-eclampsia than in healthy pregnancys. Clin Sci 2017; 131(13):1529–40.
Ribeiro VR, Romao-Veiga M, Romagnoli GG, et al. Association between cytokine profile and transcription factors produced by T-cell subsets in early- and late-onset pre-eclampsia. Immunology 2017; 152(1):163–73.
Hannan NJ, Brownfoot FC, Cannon P, et al. Resveratrol inhibits release of soluble fms-like tyrosine kinase (sFlt-1) and soluble endoglin and improves vascular dysfunction -implications as a preeclampsia treatment. Sci Rep 2017; 7(1):1819.
Yin Y, Feng Y, Zhao H, et al. SIRT1 inhibits releases of HMGB1 and HSP70 from human umbilical vein endothelial cells caused by IL-6 and the serum from a preeclampsia patient and protects the cells from death. Biomed Pharmacother 2017; 88: 449–58.
Al-Othman S, Omu AE, Diejomaoh FM, Al-Yatama M, Al-Qattan F. Differential levels of interleukin 6 in maternal and cord sera and placenta in women with preeclampsia. Gynecol Obstet Investig 2001; 52: 60–5.
Boij R, Svensson J, Nilsson-Ekdahl K, et al. Biomarkers of coagulation, inflammation and angiogenesis are independently associated with preeclampsia. Am J Reprod Immunol 2012; 68: 258–70.
Carty DM, Anderson LA, Freeman DJ, et al. Early pregnancy soluble E-selectin concentrations and risk of preeclampsia. J Hypertens 2012; 30: 954–9.
Montagnana M, Lippi G, Albiero A, Salvagno GL, Franchi M, Guidi GC. Serum pro-inflammatory cytokines in physiological and pre-eclamptic pregnancies. Gynecol Endocr 2008; 24: 113–116.
Ozkan ZS, Simsek M, Ilhan F, Deveci D, Godekmerdan A, Sapmaz E. Plasma IL-17, IL-35, interferon-g, SOCS3 and TGF-b levels in pregnant women with preeclampsia, and their relation with severity of disease. J Matern Fetal Neonat Med 2014; 27: 1513–7.
Vitoratos N, Economou E, Iavazzo C, Panoulis K, Creatsas G. Maternal serum levels of TNF-alpha and IL-6 long after delivery in preeclamptic and normotensive pregnant women. Mediat Inflamm 2010; 2010: 908649.
Maharaj NR, Phulukdaree A, Nagiah S, Ramkaran P, Tiloke C, Chuturgoon AA. Pro-inflammatory cytokine levels in HIV infected and uninfected pregnant women with and without preeclampsia. PLoS One 2017; 12(1):e0170063.
Keaton SA, Heilman P, Bryleva EY, et al. Altered tryptophan catabolism in placentas from women with pre-eclampsia. Int J Tryptophan Res 2019; 12: 1178646919840321.
Capobianco MP, Cassiano GC, da Cruz Furini AA, et al. Human” interleukin 2 (IL-2) promotion of immune regulation and clinical outcomes: a review. J Cytokine Biol 2016; 1: 10.
Sharma D, Singh A, Trivedi SS, Bhattacharjee J. Role of endothelin and inflammatory cytokines in preeclampsia-A pilot North Indian study. Am J Reprod Immunol 2011; 65: 428–32.
Sharma D, Singh A, Trivedi SS, Bhattacharjee J. Intergeno-typic variation of nitric oxide and inflammatory markers in preeclampsia: A pilot study in a north indian population. Hum Immunol 2011; 72: 436–9.
Ibrahim T, Przybyl L, Harmon AC, et al. Proliferation of endogenous regulatory T cells improve the pathophysiology associated with placental ischaemia of pregnancy. Am J Reprod Immunol 2017; 78(5).
Maharaj NR, Ramkaran P, Pillay S, Chuturgoon AA. MicroRNA-146a rs2910164 is associated with severe preeclampsia in Black South African women on HAART. BMC Genet 2017; 18(1):5.
Brünnert D, Piccenini S, Ehrhardt J, Zygmunt M, Goyal P. Sphingosine 1-phosphate regulates IL-8 expression and secretion via S1PR1 and S1PR2 receptors-mediated signaling in extravillous trophoblast derived HTR-8/SVneo cells. Placenta 2015; 36(10):1115–21.
Cemgil Arikan D, Aral M, Coskun A, Ozer A. Plasma IL-4, IL-8, IL-12, interferon-y and CRP levels in pregnant women with preeclampsia, and their relation with severity of disease and fetal birth weight. J Matern Fetal Neonat Med 2012; 25(9): 1569–73.
Luppi P, Deloia JA. Monocytes of preeclamptic women spontaneously synthesize pro-inflammatory cytokines. Clin Immunol 2006; 118: 268–75.
Molvarec A, Szarka A, Walentin S, et al. Serum heat shock protein 70 levels in relation to circulating cytokines, chemokines, adhesion molecules and angiogenic factors in women with preeclampsia. Clinica Chimica Acta 2011; 412: 1957–62.
Sahin S, Ozakpinar OB, Eroglu M, et al. The impact of platelet functions and inflammatory status on the severity of preeclampsia. J Matern Fetal Neonat Med 2015; 28: 643–8.
Berryman K, Buhimschi CS, Zhao G, Axe M, Locke M, Buhimschi IA. Proteasome levels and activity in pregnancies complicated by severe preeclampsia and hemolysis, elevated liver enzymes, and thrombocytopenia (HELLP) syndrome. Hypertension 2019; 73(6):1308–18.
Williamson RD, McCarthy FP, Kenny LC, McCarthy CM. Activation of a TLR9 mediated innate immune response in preeclampsia. Sci Rep 2019; 9(1):5920.
Khanabdali R, Shakouri-Motlagh A, Wilkinson S, et al. Low-dose aspirin treatment enhances the adhesion of preeclamptic decidual mesenchymal stem/stromal cells and reduces their production of pro-inflammatory cytokines. J MolMed2018; 96 (11):1215–25.
Arlier S. Endothelial cell leptin receptors, leptin and interleukin-8 in the pathogenesis of preeclampsia: an in vitro study. Turk J Obstet Gynecol 2017; 14(4):220–7.
Docheva N, Romero R, Chaemsaithong P, et al. The profiles of soluble adhesion molecules in the “great obstetrical syndromes”. J Matern Fetal Neonat Med 2019; 32(13):2113–36.
Salazar Garcia MD, Mobley Y, Henson J, et al. Early pregnancy immune biomarkers in peripheral blood may predict preeclampsia. J Reprod Immunol 2018; 125: 25–31.
Al-Ofi EA, Anumba DO. Ligands of toll-like receptors 2/4 differentially alter markers of inflammation, adhesion and angiogenesis by monocytes from women with pre-eclampsia in co-culture with endothelial cells. J Reprod Immunol 2017; 121: 26–33.
Adela R, Borkar RM, Mishra N, et al. Lower serum vitamin D metabolite levels in relation to circulating cytokines/chemo-kines and metabolic hormones in pregnant women with hypertensive disorders. Front Immunol 2017; 8: 273.
Sun L, Mao D, Cai Y, et al. Association between higher expression of interleukin-8 (IL-8) and haplotype -353A/-251A/ + 678T of IL-8 gene with preeclampsia: a case-control study. Medicine 2016; 95(52):e5537.
Pestka S, Krause CD, Walter MR. Interferons, interferon-like cytokines and their receptors. Immunol Rev 2004; 202: 8–32.
Huang SJ, Chen CP, Buchwalder L, et al. Regulation of CX3CL1 expression in human first-trimester decidual cells: implications for preeclampsia. Reprod Sci 2019; 26(9):1256–65.
Lee CL, Veerbeek JHW, Rana TK, Van Rijn BB, Burton GJ, Yung HW. Role of endoplasmic reticulum stress in proin-flammatory cytokine-mediated inhibition of trophoblast invasion in placenta-related complications of pregnancy. Am J Pathol 2019; 189(2):467–78.
Sharma K, Singh R, Kumar M, Gupta U, Rohil V, Bhattacharjee J. First-trimester inflammatory markers for risk evaluation of pregnancy hypertension. J Obstet Gynaecol 2018; 68(1):27–32.
Scroggins SM, Santillan DA, Lund JM, et al. Elevated vasopressin in pregnant mice induces T-helper subset alterations consistent with human preeclampsia. Clin Sci 2018; 132 (3):419–36.
Elfarra J, Amaral LM, McCalmon M, et al. Natural killer cells mediate pathophysiology in response to reduced uterine perfusion pressure. Clin Sci 2017; 131(23):2753–62.
Li X, Li T, Liu Y, et al. Association of IFNG gene polymorphisms with susceptibility to preeclampsia among pregnant woman from Shaanxi. Zhonghua Yi Xue Yi Chuan Xue Za Zhi 2017; 34(5):726–30.
Hashemi V, Dolati S, Hosseini A, Gharibi T, Danaii S, Yousefi M. Natural killer T cells in preeclampsia: an updated review. Biomed Pharmacother 2017; 95: 412–8.
Bueno-Sánchez JC, Pen˜a-Alzate S, Pen˜a RB, et al. Sera from early-onset, severely preeclamptic women directly modulate HLA-E expression in the EA. hy296 endothelial cell line. J Reprod Immunol 2014; 104–105: 68–79.
Giurgescu C, Sanguanklin N, Engeland C G, et al. Relationships among psychosocial factors, biomarkers, preeclampsia, and preterm birth in African American women: a pilot. Appl Nurs Res 2015; 28: e1–6.
Cemgil Arikan D, Aral M, Coskun A, Ozer A. Plasma IL-4, IL-8, IL-12, interferon-g and CRP levels in pregnant women with preeclampsia and their relation with severity of disease and fetal birth weight. J Matern Fetal Neonat Med 2012; 25: 1569–73.
Haedersdal S, Salvig JD, Aabye M, et al. Inflammatory markers in the second trimester prior to clinical onset of preeclampsia, intrauterine growth restriction and spontaneous preterm birth. Inflammation 2013; 36: 907–13.
Soobryan N, Murugesan S, Phoswa W, Gathiram P, Moodley J, Mackraj I. The effects of sildenafil citrate on uterine angiogenic status and serum inflammatory markers in an L-NAME rat model of pre-eclampsia. Eur J Pharmacol 2017; 795: 101–7.
Ren M, Li X, Hao L, Zhong J. Role of tumor necrosis factor alpha in the pathogenesis of atrial fibrillation: a novel potential therapeutic target? Ann Med 2015; 47(4):316–24.
Cackovic M, Buhimschi CS, Zhao G, et al. Fractional excretion of tumor necrosis factor-alpha in women with severe preeclampsia. Obstet Gynecol 2008; 112: 93–100.
Ferguson KK, McElrath TF, Chen YH, Mukherjee B, Meeker JD. Longitudinal profiling of inflammatory cytokines and C-reactive protein during uncomplicated and preterm pregnancy. Am J Reprod Immunol 2014; 72(3):326–36.
Hou L, Zhu Y, Ma X, Li J, Zhang W. Serum protein microarray analysis of patients with preeclampsia. Mol Med Rep 2012; 6: 83–7.
Shu C, Yan D, Chen C, et al. Metformin exhibits its therapeutic effect in the treatmentof pre-eclampsia via modulating the Met/ H19/miR-148a-5p/P28 and Met/H19/miR-216–3p/EBI3 signaling pathways. Int Immunopharmacol 2019; 74: 105693.
Liu Z, Zhao X, Shan H, Gao H, Wang P. microRNA-520c-3p suppresses NLRP3 inflammasome activation and inflammatory cascade in preeclampsia by downregulating NLRP3. Inflamm Res 2019; 68(8):643–54.
Doganlar ZB, Güçlü H, Oztopuz O, et al. The role of melatonin in oxidative stress, DNA damage, apoptosis and angiogenesis in fetal eye under preeclampsia and melatonin deficiency stress. Curr Eye Res 2019; 44: 1–13.
Cox AG, Gurusinghe S, Abd Rahman R, et al. Sulforaphane improves endothelial function and reduces placental oxidative stress in vitro. Pregnancy Hypertens 2019; 16: 1–10.
Matias ML, Gomes VJ, Romao-Veiga M, et al. Silibinin Downregulates the NF-kB Pathway and NLRP1/NLRP3 Inflammasomes in Monocytes from Pregnant Women with Preeclampsia. Molecules 2019; 24(8):E1548.
Ahmed M, Alqosaibi A, Mohamed MA, Soliman MG. Evaluation of some cytokines and gene expressions in pre-eclampsia. Pak J Biol Sci 2019; 22(3):148–53.
Zhang QL, Wang L, Xu MJ, Wang TL. Protective effect of dexmedetomidine on kidney injury of parturients with preeclampsia undergoing cesarean section: a randomized controlled study. Biosci Rep 2019; 39(5):BSR20190352.
Harmon AC, Ibrahim T, Cornelius DC, et al. Placental CD4 T cells isolated from preeclamptic women cause preeclampsia-like symptoms in pregnant nude-athymic rats. Pregnancy Hypertens 2019; 15: 7–11.
Ampey AC, Boeldt DS, Clemente L, et al. TNF-alpha inhibits pregnancy-adapted Ca signaling in uterine artery endothelial cells. Mol Cell Endocrinol 2019; 488: 1424.
Li ZH, Wang LL, Liu H, et al. Galectin-9 alleviates LPS-induced preeclampsia-like impairment in rats via switching decidual macrophage polarization to M2 subtype. Front Immunol 2019; 9: 3142.
Turbeville HR, Taylor EB, Garrett MR, Didion SP, Ryan MJ, Sasser JM. Superimposed preeclampsia exacerbates postpar-tum renal injury despite lack of long-term blood pressure difference in the dahl salt-sensitive rat. Hypertension 2019; 73 (3):650–8.
Lin Y, Wang L, Yan Y, Zhou W, Chen Z. A meta-analysis of tumor necrosis factor-a and FAS/FASL polymorphisms with risk of pre-eclampsia. Hypertens Pregnancy 2019; 38(1):20–31.
Wang Z, Wang P, Wang Z, et al. MiRNA-548c-5p down-regulates inflammatory response in preeclampsia via targeting PTPRO. J Cell Physiol 2019; 234(7):11149–55.
Kim S, Lee KS, Choi S, et al. NF-kB-responsive miRNA-31–5p elicits endothelial dysfunction associated with preeclampsia via down-regulation of endothelial nitric-oxide synthase. J Biol Chem 2018; 293(49):18989–9000.
Feng J, Wang X, Li H, Wang L, Tang Z. SilencingofAnnexinA1 suppressed the apoptosis and inflammatory response of preecla-mpsia rat trophoblasts. Int J Mol Med 2018; 42(6):3125–34.
Canfield J, Arlier S, Mong EF, et al. Decreased LIN28B in preeclampsia impairs human trophoblast differentiation and migration. FASEB J 2019; 33(2):2759–69.
Wang L, Qu G, Wu W, Tang X, Sun Y. Association between tumor necrosis factor-a-308G/A gene polymorphism and susceptibility to pre-eclampsia: an updated meta-analysis. Cytokine 2018; 111: 278–86.
Chen Y, Xue F, Han C, et al. Ferulic acid ameliorated placental inflammation and apoptosis in rat with preeclampsia. Clin Exp Hypertens 2019; 41(6):524–30.
Wen Z, Chen Y, Long Y, Yu J, Li M. Tumor necrosis factor-alpha suppresses the invasion of HTR-8/SVneo trophoblast cells through microRNA-145–5p-mediated downregulation of Cyr61. Life Sci 2018; 209: 132–9.
Zou AX, Chen B, Li QX, Liang YC. MiR-134 inhibits infiltration of trophoblast cells in placenta of patients with preeclampsia by decreasing ITGB1 expression. Eur Rev Med Pharmacol Sci 2018; 22(8):2199–206.
Li G, Ma L, Lin L, Wang YL, Yang H. The intervention effect of aspirin on a lipopolysaccharide-induced preeclampsia-like mouse model by inhibiting the nuclear factor-kB pathway. Biol Reprod 2018; 99(2):422–32.
Brunacci F, Rocha VS, De Carli E, Espósito BP, Ruano R, Colli C. Increased serum iron in preeclamptic women is likely due to low hepcidin levels. Nutr Res 2018; 53: 32–9.
Agostinis C, Rami D, Zacchi P, et al. Pre-eclampsia affects procalcitonin production in placental tissue. Am J Reprod Immunol 2018; 79(4):e12823.
Quan LM, Xu QL, Zhang GQ, Wu LL, Xu H. An analysis of the risk factors of preeclampsia and prediction based on combined biochemical indexes. Kaohsiung J Med Sci 2018; 34 (2):109–12.
Uckan K, Sahin HG. Serum amyloid A, procalcitonin, highly sensitive C reactive protein and tumor necrosis factor alpha levels and acute inflammatory response in patients with hemolysis, elevated liver enzymes, low platelet count (HELLP) and eclampsia. J Obstet Gynaecol Res 2018; 44(3):440–7.
Shi DD, Wang Y, Guo JJ, Zhou L, Wang N. Vitamin D enhances efficacy of oral nifedipine in treating preeclampsia with severe features: a double blinded, placebo-controlled and randomized clinical trial. Front Pharmacol 2017; 8: 865.
Bakrania BA, Spradley FT, Satchell SC, et al. Heme oxygenase-1 is a potent inhibitor of placental ischemia-mediated endothelin-1 production in cultured human glomer-ular endothelial cells. Am J Physiol Regul Integr Comp Physiol 2018; 314(3):R427–32.
Gurusinghe S, Cox AG, Rahman R, et al. Resveratrol mitigates trophoblast and endothelial dysfunction partly via activation of nuclear factor erythroid 2-related factor-2. Placenta 2017; 60: 74–85.
Amaral LM, Faulkner JL, Elfarra J, et al. Continued investigation into 17-OHPC: results from the preclinical RUPP rat model of preeclampsia. Hypertension 2017; 70(6):1250–5.
Xu B, Shanmugalingam R, Chau K, Pears S, Hennessy A, Makris A. The effect of acetyl salicylic acid (Aspirin) on trophoblast-endothelial interaction in vitro. J Reprod Immunol 2017; 124: 54–61.
Yu J, Jia J, Guo X, Chen R, Feng L. Modulating circulating sFlt1 in an animal model of preeclampsia using PAMAM nanoparticles for siRNA delivery. Placenta 2017; 58: 1–8.
Kaitu’u-Lino TJ, Brownfoot FC, Hastie R, et al. Activating transcription factor 3 is reduced in preeclamptic placentas and negatively regulates sFlt-1 (soluble fms-like tyrosine kinase 1), soluble endoglin, and proinflammatory cytokines in placenta. Hypertension 2017; 70(5):1014–24.
Li W, Cui N, Mazzuca MQ, Mata KM, Khalil RA. Increased vascular and uteroplacental matrix metalloproteinase-1 and -7 levels and collagen type I deposition in hypertension in pregnancy: role of TNF-a. Am J Physiol Heart Circ Physiol 2017; 313(3):H491–507.
Kim J, Lee KS, Kim JH, et al. Aspirin prevents TNF-a-induced endothelial cell dysfunction by regulating the NF-kB-depen-dent miR-155/eNOS pathway: Role of a miR-155/eNOS axis in preeclampsia. Free Radic Biol Med 2017; 104: 185–98.
Alijotas-Reig J, Esteve-Valverde E, Ferrer-Oliveras R, Llurba E, Gris JM. Tumor necrosis factor-alpha and pregnancy: focus on biologics, an updated and comprehensive review. Clin Rev Allergy Immunol 2017; 53(1):40–53.
Ferguson KK, Meeker JD, McElrath TF, Mukherjee B, Cantonwine DE. Repeated measures of inflammation and oxidative stress biomarkers in preeclamptic and normotensive pregnancies. Am J Obstet Gynecol 2017; 216(5):527.e1–527.e9.
Tavakkol Afshari Z, Rahimi HR, Ehteshamfar SM, Ganjali R, Tara F, Shapouri Moghadam A. Tumor necrosis factor-α and interleukin-1-β polymorphisms in pre-eclampsia. Iran J Immunol 2016; 13(4):309–16.
Xu B, Bobek G, Makris A, Hennessy A. Antihypertensive methyldopa, labetalol, hydralazine and clonidine reversed tumour necrosis factor-α inhibited endothelial nitric oxide synthase expression in endothelial-trophoblast cellular networks. Clin Exp Pharmacol Physiol 2017; 44(3):421–7.
Taylor BD, Ness RB, Klebanoff MA, et al. First and second trimester immune biomarkers in preeclamptic and normoten-sive women. Pregnancy Hypertens 2016; 6(4):388–93.
Song J, Li Y, An R. Vitamin D restores angiogenic balance and decreases tumor necrosis factor-α in a rat model of pre-eclampsia. J Obstet Gynaecol Res 2017; 43(1):42–9.
Dong X, Shi D. Simvastatin alleviates pathology in a rat model of preeclampsia involving ERK/MAPK pathway. Reprod Sci 2017; 24(7):1053–61.
Shaw J, Tang Z, Schneider H, Saljé K, Hansson SR, Guller S. Inflammatory processes are specifically enhanced in endothelial cells by placental-derived TNF-α: Implications in preeclampsia (PE). Placenta 2016; 43: 1–8.
Weel IC, Baergen RN, Romão-Veiga M, et al. Association between placental lesions, cytokines and angiogenic factors in pregnant women with preeclampsia. PLoS One 2016; 11(6): e0157584.
Zhou L, Cheng L, He Y, Gu Y, Wang Y, Wang C. Association of gene polymorphisms of FV, FII, MTHFR, SERPINE1, CTLA4, IL10 and TNF alpha with preeclampsia in Chinese women. Inflamm Res 2016; 65(9):717–24.
Mohammadpour-Gharehbagh A, Jahantigh D, Eskandari M, et al. The role of TNF-a and TLR4 polymorphisms in the placenta of pregnant women complicated by preeclampsia and in silico analysis. Int J Biol Macromol 2019; 134: 1205–15.
Artunc-Ulkumen B, Guvenc Y, Goker A, Gozukara C. Relationship of neutrophil gelatinase-associated lipocalin (NGAL) and procalcitonin levels with the presence and severity of the preeclampsia. J Matern Fetal Neonat Med 2015; 28: 1895–900.
Couper KN, Blount DG, Riley EM. IL-10: the master regulator of immunity to infection. J Immunol 2008; 180(9):5771–7.
Bachmayer N, Rafik Hamad R, Liszka L, Bremme K, Sverremark-Ekström E. Aberrant uterine natural killer (NK)-cell expression and altered placental and serum levels of the NK-cell promoting cytokine interleukin-12 in pre-eclampsia. Am J Reprod Immunol 2006; 56: 292–301.
Makris A, Xu B, Yu B, Thornton C, Hennessy A. Placental deficiency of interleukin-10 (IL-10) in preeclampsia and its relationship to an IL-10 promoter polymorphism. Placenta 2006; 27: 445–51.
Yu J, Qian L, Wu F, Li M, Chen W, Wang H. Decreased frequency of peripheral blood CD8CD25FoxP3regulatory T cells correlates with IL-33 levels in pre-eclampsia. Hypertens Pregnancy 2017; 36(2):217–25.
Cui S, Gao Y, Zhang L, et al. Combineduseofserum MCP-1/IL-10 ratio and uterine artery Doppler index significantly improves the predictionofpreeclampsia. Clin Chim Acta 2017; 473: 228–36.
Azizieh FY, Raghupathy R. IL-10 and pregnancy complications. Clin Exp Obstet Gynecol 2017; 44(2):252–8.
Cubro H, Kashyap S, Nath MC, Ackerman AW, Garovic VD. The role of interleukin-10 in the pathophysiology of pre-eclampsia. Curr Hypertens Rep 2018; 20(4):36.
Raguema N, Gannoun MBA, Zitouni H, et al. Interleukin-10 rs1800871 (-819C/T) and ATA haplotype are associated with preeclampsia in a Tunisian population. Pregnancy Hypertens 2018; 11: 105–10.
Zhang Z, Liu H, Shi Y, et al. Increased circulating Th22 cells correlated with Th17 cells in patients with severe preeclampsia. Hypertens Pregnancy 2017; 36(1):100–7.
Xu H, Shi Q, Mo Y, Wu L, Gu J, Xu Y. Downregulation of a7 nicotinic acetylcholine receptors in peripheral blood monocytes is associated with enhanced inflammation in preeclampsia. BMC Pregnancy Childbirth 2019; 19(1):188.
Nunes PR, Romão-Veiga M, Peraçoli JC, et al. Downregulation of CD163 in monocytes and its soluble form in the plasma is associated with a pro-inflammatory profile in pregnant women with preeclampsia. Immunol Res 2019; 67(2–3): 194–201.
Yang SW, Cho EH, Choi SY, et al. DC-SIGN expression in Hofbauer cells may play an important role in immune tolerance in fetal chorionic villi during the development of preeclampsia. J Reprod Immunol 2017; 124: 30–7.
Heyward CY, Sones JL, Lob HE, et al. The decidua of preeclamptic-like BPH/5 mice exhibits an exaggerated inflammatory response during early pregnancy. J Reprod Immunol 2017; 120: 27–33.
Cornelius DC, Cottrell J, Amaral LM, LaMarca B. Inflammatory mediators: a causal link to hypertension during pre-eclampsia. Br J Pharmacol 2019; 176(12):1914–21.
Choi P, Reiser H. IL-4: role in disease and regulation of production. Clin Exp Immunol 1998; 113: 317–9.
Chen J, Zhong M, Yu YH. Association between interleukin-4 polymorphisms and risk of pre-eclampsia in a population of Chinese pregnant women. Genet Mol Res 2017; 16(2).
Chatterjee P, Chiasson VL, Seerangan G, et al. Cotreatment with interleukin 4 and interleukin 10 modulates immune cells and prevents hypertension in pregnant mice. Am J Hypertens 2015; 28(1):135–42.
Salimi S, Mohammadoo-Khorasani M, Yaghmaei M, Mokh-tari M, Moossavi M. Possible association of IL-4 VNTR polymorphism with susceptibility to preeclampsia. Biomed Res Int 2014; 2014: 497031.
Omu AE, Makhseed M, Al-Qattan F. The comparative value of interleukin-4 in sera of women with preeclampsia and cord sera. Nutrition 1995; 11(5):688–91.
Hwang JH, Lee MJ, Seok OS, et al. Cytokine expression in placenta-derived mesenchymal stem cells in patients with pre-eclampsia and normal pregnancies. Cytokine 2010; 49(1):95–101.
Chatterjee P, Kopriva SE, Chiasson VL, et al. Interleukin-4 deficiency induces mild preeclampsia in mice. J Hypertens 2013; 31(7):1414–23.
Fraser R, Walker JJ, Ekbote UV, Martin KL, McShane P, Orsi NM. Interleukin- 4–590 (C > T), toll-like receptor-2 +2258 (G > A) and matrix metalloproteinase-9–1562 (C > T) polymorphisms in pre-eclampsia. BJOG 2008; 115(8):1052–6.
Azizieh F, Raghupathy R, Makhseed M. Maternal cytokine production patterns in women with pre-eclampsia. Am J Reprod Immunol 2005; 54(1):30–7.
Kang L, Chen CH, Yu CH, Chang CH, Chang FM. An association study of interleukin-4 gene and preeclampsia in Taiwan. Taiwan J Obstet Gynecol 2014; 53(2):215–9.
Henriques CU, Rice GE, Wong MH, Bendtzen K. Immuno-localisation of interleukin-4 and interleukin-4 receptor in placenta and fetal membranes in association with pre-term labour and pre-eclampsia. Gynecol Obstet Invest 1998; 46 (3):172–7.
Omu AE, Makhseed M, Al-Qattan F. Effect of antihyperten-sive therapy in preeclampsia on levels of serum interleukin-4. Gynecol Obstet Invest 1996; 42(4):230–6.
Shima Y. The benefits and prospects of interleukin-6 inhibitor on systemic sclerosis. Mod Rheumatol 2019; 29(2):294–301.
Tangerås LH, Austdal M, Skråstad RB, et al. Distinct first trimester cytokine profiles for gestational hypertension and preeclampsia. Arterioscler Thromb Vasc Biol 2015; 35(11): 2478–2485.
Peixoto AB, Araujo Jünior E, Ribeiro JU, et al. Evaluation of inflammatory mediators in the deciduas of pregnant women with pre-eclampsia/eclampsia. J Matern Fetal Neonatal Med 2016; 29(1):75–9.
Bueno-Sánchez JC, Agudelo-Jaramillo B, Escobar-Aguilerae LF, et al. Cytokine production by non-stimulated peripheral blood NK cells and lymphocytes in early-onset severe pre-eclampsia without HELLP. J Reprod Immunol 2013; 97(2):223–31.
Brewster JA, Orsi NM, Gopichandran N, Ekbote UV, Cadogan E, Walker JJ. Host inflammatory response profiling in preeclampsia using an in vitro whole blood stimulation model. Hypertens Pregnancy 2008; 27(1):1–16.
Jonsson Y, Matthiesen L, Berg G, Ernerudh J, Nieminen K, Ekerfelt C. Indications of an altered immune balance in preeclampsia: a decrease in in vitro secretion of IL-5 and IL-10 from blood mononuclear cells and in blood basophil counts compared with normal pregnancy. J Reprod Immunol 2005; 66 (1):69–84.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Guney, G., Taskin, M.I. & Tokmak, A. Increase of circulating inflammatory molecules in preeclampsia, an update. Eur Cytokine Netw 31, 18–31 (2020). https://doi.org/10.1684/ecn.2020.0443
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1684/ecn.2020.0443