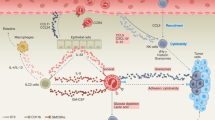
Abstract
Basophils, which are considered as redundant relatives of mast cells and the rarest granulocytes in peripheral circulation, have been neglected by researchers in the past decades. Previous studies have revealed their vital roles in allergic diseases and parasitic infections. Intriguingly, recent studies even reported that basophils might be associated with cancer development, as activated basophils synthesize and release a variety of cytokines and chemokines in response to cancers. However, it is still subject to debate whether basophils function as tumor-protecting or tumor-promoting components; the answer may depend on the tumor biology and the microenvironment. Herein, we reviewed the role of basophils in cancers, and highlighted some potential and promising therapeutic strategies.
概要
嗜碱性粒细胞是外周血中比例最少的粒细胞, 曾一度被认为是肥大细胞的“冗余亲属”, 在过去的几十年里一直被研究人员忽视. 以往的研究揭示了其在过敏性疾病和寄生虫感染中的重要作用, 最新研究则表明嗜碱性粒细胞可能与肿瘤的发生发展同样具有密切关联. 活化的嗜碱性粒细胞合成并释放多种细胞因子和趋化因子参与并调控肿瘤的发生发展. 然而, 嗜碱性粒细胞究竟扮演着抑癌还是促癌的角色至今仍存在较大争议, 这可能取决于肿瘤生物学行为以及微环境的影响. 因此, 我们回顾了嗜碱性粒细胞的生物学功能及其在肿瘤发生发展中的潜在作用, 并展望其在肿瘤治疗中的应用前景.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Allen F, Bobanga ID, Rauhe P, et al., 2018. CCL3 augments tumor rejection and enhances CD8+ T cell infiltration through NK and CD103+ dendritic cell recruitment via IFNγ. Oncoimmunology, 7(3):e1393598. https://doi.org/10.1080/2162402X.2017.1393598
Anthony HM, 1982. Blood basophils in lung cancer. Br J Cancer, 45(2):209–216. https://doi.org/10.1038/bjc.1982.35
Aspord C, Pedroza-Gonzalez A, Gallegos M, et al., 2007. Breast cancer instructs dendritic cells to prime interleukin 13-secreting CD4+ T cells that facilitate tumor development. J Exp Med, 204(5):1037–1047. https://doi.org/10.1084/jem.20061120
Baba T, Tanabe Y, Yoshikawa S, et al., 2016. MIP-1α/CCL3-expressing basophil-lineage cells drive the leukemic hematopoiesis of chronic myeloid leukemia in mice. Blood, 127(21):2607–2617. https://doi.org/10.1182/blood-2015-10-673087
Balam S, Schiechl-Brachner G, Buchtler S, et al., 2019. IL-3 triggers chronic rejection of cardiac allografts by activation of infiltrating basophils. J Immunol, 202(12): 3514–3523. https://doi.org/10.4049/jimmunol.1801269
Borriello F, Longo M, Spinelli R, et al., 2015. IL-3 synergises with basophil-derived IL-4 and IL-13 to promote the alternative activation of human monocytes. Eur J Immunol, 45(7):2042–2051. https://doi.org/10.1002/eji.201445303
Caporali S, Amaro A, Levati L, et al., 2019. miR-126-3p down-regulation contributes to dabrafenib acquired resistance in melanoma by up-regulating ADAM9 and VEGF-A. J Exp Clin Cancer Res, 38:272. https://doi.org/10.1186/s13046-019-1238-4
Chang LY, Lin YC, Mahalingam J, et al., 2012. Tumor-derived chemokine CCL5 enhances TGF-β-mediated killing of CD8+ T cells in colon cancer by T-regulatory cells. Cancer Res, 72(5):1092–1102. https://doi.org/10.1158/0008-5472.CAN-11-2493
Cheng J, He SJ, Wang M, et al., 2019. The caspase-3/PKCδ/Akt/VEGF-A signaling pathway mediates tumor repopulation during radiotherapy. Clin Cancer Res, 25(12): 3732–3743. https://doi.org/10.1158/1078-0432.CCR-18-3001
Clara JA, Monge C, Yang YZ, et al., 2020. Targeting signalling pathways and the immune microenvironment of cancer stem cells—a clinical update. Nat Rev Clin Oncol, 17(4): 204–232. https://doi.org/10.1038/s41571-019-0293-2
Cohen M, Giladi A, Gorki AD, et al., 2018. Lung single-cell signaling interaction map reveals basophil role in macrophage imprinting. Cell, 175(4):1031–1044.e18. https://doi.org/10.1016/j.cell.2018.09.009
Costamagna D, Duelen R, Penna F, et al., 2020. Interleukin-4 administration improves muscle function, adult myogenesis, and lifespan of colon carcinoma-bearing mice. J Cachexia Sarcopenia Muscle, 11(3):783–801. https://doi.org/10.1002/jcsm.12539
Crawford G, Hayes MD, Seoane RC, et al., 2018. Epithelial damage and tissue γδ T cells promote a unique tumor-protective IgE response. Nat Immunol, 19(8):859–870. https://doi.org/10.1038/s41590-018-0161-8
Das M, Stephen-Victor E, Bayry J, 2020. Regulatory T cells do not suppress rather activate human basophils by IL-3 and STAT5-dependent mechanisms. Oncoimmunology, 9:1773193. https://doi.org/10.1080/2162402x.2020.1773193
Deepak P, Sanjay K, Acharya A, 2007. IL-13 Ralpha2-mediated interleukin-13 neutralization represses in vivo progressive growth of a T-cell lymphoma. J Exp Clin Cancer Res, 26(3):347–352.
Deepak P, Kumar S, Acharya A, 2008. Interleukin-13 neutralization modulates interleukin-13 induced suppression of reactive oxygen species production in peritoneal macrophages in a murine T-cell lymphoma. Cell Immunol, 251(2):72–77. https://doi.org/10.1016/j.cellimm.2008.03.005
de Monte L, Wörmann S, Brunetto E, et al., 2016. Basophil recruitment into tumor-draining lymph nodes correlates with Th2 inflammation and reduced survival in pancreatic cancer patients. Cancer Res, 76(7): 1792–1803. https://doi.org/10.1158/0008-5472.CAN-15-1801-T
Deng HJ, Kan AN, Lyu N, et al., 2021. Tumor-derived lactate inhibit the efficacy of lenvatinib through regulating PD-L1 expression on neutrophil in hepatocellular carcinoma. J Immunother Cancer, 9:e002305. https://doi.org/10.1136/jitc-2020-002305
de Paulis A, Prevete N, Fiorentino I, et al., 2006. Expression and functions of the vascular endothelial growth factors and their receptors in human basophils. J Immunol, 177(10): 7322–7331. https://doi.org/10.4049/jimmunol.177.10.7322
Didichenko SA, Spiegl N, Brunner T, et al., 2008. IL-3 induces a Pim1-dependent antiapoptotic pathway in primary human basophils. Blood, 112(10):3949–3958. https://doi.org/10.1182/blood-2008-04-149419
di Stefano AB, Iovino F, Lombardo Y, et al., 2010. Survivin is regulated by interleukin-4 in colon cancer stem cells. J Cell Physiol, 225(2):555–561. https://doi.org/10.1002/jcp.22238
Engeroff P, Caviezel F, Mueller D, et al., 2020. CD23 provides a noninflammatory pathway for IgE-allergen complexes. J Allergy Clin Immunol, 145(1):301–311.e4. https://doi.org/10.1016/j.jaci.2019.07.045
Falkensammer C, Jöhrer K, Gander H, et al., 2006. IL-4 inhibits the TNF-α induced proliferation of renal cell carcinoma (RCC) and cooperates with TNF-α to induce apoptotic and cytokine responses by RCC: implications for antitumor immune responses. Cancer Immunol Immunother, 55(10): 1228–1237. https://doi.org/10.1007/S00262-006-0122-1
Ferrara N, Kerbel RS, 2005. Angiogenesis as a therapeutic target. Nature, 438(7070):967–974. https://doi.org/10.1038/nature04483
Galeotti C, Stephen-Victor E, Karnam A, et al., 2019. Intravenous immunoglobulin induces IL-4 in human basophils by signaling through surface-bound IgE. J Allergy Clin Immunol, 144(2):524–535.e8. https://doi.org/10.1016/j.jaci.2018.10.064
Gentinetta T, Pecaric-Petkovic T, Wan D, et al., 2011. Individual IL-3 priming is crucial for consistent in vitro activation of donor basophils in patients with chronic urticaria. J Allergy Clin Immunol, 128(6):1227–1234.e5. https://doi.org/10.1016/j.jaci.2011.07.021
Gharib AF, Shalaby SM, Raafat N, et al., 2017. Assessment of neutralizing interleukin-4 effect on CD133 gene expression in colon cancer cell line. Cytokine, 97:66–72. https://doi.org/10.1016/j.cyto.2017.05.022
Gocheva V, Wang HW, Gadea BB, et al., 2010. IL-4 induces cathepsin protease activity in tumor-associated macrophages to promote cancer growth and invasion. Genes Dev, 24(3): 241–255. https://doi.org/10.1101/gad.1874010
Hagmann BR, Odermatt A, Kaufmann T, et al., 2017. Balance between IL-3 and type I interferons and their interrelationship with FasL dictates lifespan and effector functions of human basophils. Clin Exp Allergy, 47(1):71–84. https://doi.org/10.1111/cea.12850
He XD, Cao YF, Gu Y, et al., 2021. Clinical outcomes and immune metrics in intratumoral basophil-enriched gastric cancer patients. Ann Surg Oncol, 28:6439–6450. https://doi.org/10.1245/s10434-021-09815-0
Herbst T, Esser J, Prati M, et al., 2012. Antibodies and IL-3 support helminth-induced basophil expansion. Proc Natl Acad Sci USA, 109(37):14954–14959. https://doi.org/10.1073/pnas.1117584109
Ho VW, Hofs E, Elisia I, et al., 2016. All trans retinoic acid, transforming growth factor β and prostaglandin E2 in mouse plasma synergize with basophil-secreted interleukin-4 to M2 polarize murine macrophages. PLoS ONE, 11(12): e0168072. https://doi.org/10.1371/journal.pone.0168072
Ito SE, Shirota H, Kasahara Y, et al., 2017. IL-4 blockade alters the tumor microenvironment and augments the response to cancer immunotherapy in a mouse model. Cancer Immunol Immunother, 66(11): 1485–1496. https://doi.org/10.1007/s00262-017-2043-6
Jiang X, 2019. Lymphatic vasculature in tumor metastasis and immunobiology. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 21(1):3–11. https://doi.org/10.1631/jzus.B1800633
Karasuyama H, Yamanishi Y, 2014. Basophils have emerged as a key player in immunity. Curr Opin Immunol, 31:1–7. https://doi.org/10.1016/j.coi.2014.07.004
Karasuyama H, Miyake K, Yoshikawa S, et al., 2018. Multi-faceted roles of basophils in health and disease. J Allergy Clin Immunol, 142(2):370–380. https://doi.org/10.1016/j.jaci.2017.10.042
Keklikoglou I, Kadioglu E, Bissinger S, et al., 2018. Periostin limits tumor response to VEGFA inhibition. Cell Rep, 22(10): 2530–2540. https://doi.org/10.1016/j.celrep.2018.02.035
Kodama T, Koma YI, Arai N, et al., 2020. CCL3-CCR5 axis contributes to progression of esophageal squamous cell carcinoma by promoting cell migration and invasion via Akt and ERK pathways. Lab Invest, 100(9):1140–1157. https://doi.org/10.1038/s41374-020-0441-4
Koh YJ, Kim HZ, Hwang SI, et al., 2010. Double antiangiogenic protein, DAAP, targeting VEGF-A and angiopoietins in tumor angiogenesis, metastasis, and vascular leakage. Cancer Cell, 18(2):171–184. https://doi.org/10.1016/j.ccr.2010.07.001
Kreso A, Dick JE, 2014. Evolution of the cancer stem cell model. Cell Stem Cell, 14(3):275–291. https://doi.org/10.1016/j.stem.2014.02.006
Lantz CS, Boesiger J, Song CH, et al., 1998. Role for interleukin-3 in mast-cell and basophil development and in immunity to parasites. Nature, 392(6671):90–93. https://doi.org/10.1038/32190
Lavin Y, Kobayashi S, Leader A, et al., 2017. Innate immune landscape in early lung adenocarcinoma by paired single-cell analyses. Cell, 169(4):750–765.e17. https://doi.org/10.1016/j.cell.2017.04.014
Li H, Sim TC, Alam R, 1996. IL-13 released by and localized in human basophils. J Immunol, 156(12):4833–4838.
Lien MY, Tsai HC, Chang AC, et al., 2018. Chemokine CCL4 induces vascular endothelial growth factor C expression and lymphangiogenesis by miR-195-3p in oral squamous cell carcinoma. Front Immunol, 9:412. https://doi.org/10.3389/fimmu.2018.00412
Lima-Fernandes E, Murison A, da Silva Medina T, et al., 2019. Targeting bivalency de-represses Indian Hedgehog and inhibits self-renewal of colorectal cancer-initiating cells. Nat Commun, 10:1436. https://doi.org/10.1038/s41467-019-09309-4
Lin XB, Wang SY, Sun M, et al., 2019. miR-195-5p/NOTCH2-mediated EMT modulates IL-4 secretion in colorectal cancer to affect M2-like TAM polarization. J Hematol Oncol, 12:20. https://doi.org/10.1186/s13045-019-0708-7
Liou GY, Bastea L, Fleming A, et al., 2017. The presence of interleukin-13 at pancreatic ADM/PanIN lesions alters macrophage populations and mediates pancreatic tumorigenesis. Cell Rep, 19(7):1322–1333. https://doi.org/10.1016/j.celrep.2017.04.052
Liu JY, Li F, Wang LP, et al., 2015. CTL-vs Treg lymphocyte-attracting chemokines, CCL4 and CCL20, are strong reciprocal predictive markers for survival of patients with oesophageal squamous cell carcinoma. Br J Cancer, 113(5): 747–755. https://doi.org/10.1038/bjc.2015.290
Long HX, Xie RK, Xiang T, et al., 2012. Autocrine CCL5 signaling promotes invasion and migration of CD133+ ovarian cancer stem-like cells via NF-κB-mediated MMP-9 upregulation. Stem Cells, 30(10):2309–2319. https://doi.org/10.1002/stem.1194
Ma HL, Whitters MJ, Jacobson BA, et al., 2004. Tumor cells secreting IL-13 but not IL-13Rα2 fusion protein have reduced tumorigenicity in vivo. Int Immunol, 16(7):1009–1017. https://doi.org/10.1093/intimm/dxh105
Malveaux FJ, Conroy MC, Adkinson NF, et al., 1978. IgE receptors on human basophils. Relationship to serum IgE concentration. J Clin Invest, 62(1):176–181. https://doi.org/10.1172/JCI109103
Marone G, Varricchi G, Loffredo S, et al., 2016. Mast cells and basophils in inflammatory and tumor angiogenesis and lymphangiogenesis. Eur J Pharmacol, 778:146–151. https://doi.org/10.1016/j.ejphar.2015.03.088
Marone G, Gambardella AR, Mattei F, et al., 2020a. Basophils in tumor microenvironment and surroundings. Adv Exp Med Biol, 1224:21–34. https://doi.org/10.1007/978-3-030-35723-8_2
Marone G, Schroeder JT, Mattei F, et al., 2020b. Is there a role for basophils in cancer? Front Immunol, 11:2103. https://doi.org/10.3389/fimmu.2020.02103
Mayer P, Valent P, Schmidt G, et al., 1989. The in vivo effects of recombinant human interleukin-3: demonstration of basophil differentiation factor, histamine-producing activity, and priming of GM-CSF-responsive progenitors in nonhuman primates. Blood, 74(2):613–621. https://doi.org/10.1182/blood.V74.2.613.613
Mgrditchian T, Arakelian T, Paggetti J, et al., 2017. Targeting autophagy inhibits melanoma growth by enhancing NK cells infiltration in a CCL5-dependent manner. Proc Natl Acad Sci USA, 114(44):E9271–E9279. https://doi.org/10.1073/pnas.1703921114
Nakano A, Yoneyama H, Ueha S, et al., 2007. Intravenous administration of MIP-1α with intra-tumor injection of P. acnes shows potent anti-tumor effect. Int Immunopharmacol, 7(6):845–857. https://doi.org/10.1016/j.intimp.2007.02.005
Nakashima C, Otsuka A, Kitoh A, et al., 2014. Basophils regulate the recruitment of eosinophils in a murine model of irritant contact dermatitis. J Allergy Clin Immunol, 134(1):100–107. https://doi.org/10.1016/j.jaci.2014.02.026
Nakasone Y, Fujimoto M, Matsushita T, et al., 2012. Host-derived MCP-1 and MIP-1α regulate protective anti-tumor immunity to localized and metastatic B16 melanoma. Am J Pathol, 180(1):365–374. https://doi.org/10.1016/j.ajpath.2011.09.005
Nakayama T, Hirahara K, Onodera A, et al., 2017. Th2 cells in health and disease. Annu Rev Immunol, 35:53–84. https://doi.org/10.1146/annurev-immunol-051116-052350
Nesbeth YC, Martinez DG, Toraya S, et al., 2010. CD4+ T cells elicit host immune responses to MHC class II− ovarian cancer through CCL5 secretion and CD40-mediated licensing of dendritic cells. J Immunol, 184(10):5654–5662. https://doi.org/10.4049/jimmunol.0903247
Nigro EA, Brini AT, Yenagi VA, et al., 2016. Cutting edge: IgE plays an active role in tumor immunosurveillance in mice. J Immunol, 197(7):2583–2588. https://doi.org/10.4049/jimmunol.1601026
Obata K, Mukai K, Tsujimura Y, et al., 2007. Basophils are essential initiators of a novel type of chronic allergic inflammation. Blood, 110(3):913–920. https://doi.org/10.1182/blood-2007-01-068718
Oda Y, Fukunaga A, Washio K, et al., 2019. Low responsiveness of basophils via FcεRI reflects disease activity in chronic spontaneous urticaria. J Allergy Clin Immunol Pract, 7(8):2835–2844.e7. https://doi.org/10.1016/j.jaip.2019.05.020
Oh K, Shen T, le Gros G, et al., 2007. Induction of Th2 type immunity in a mouse system reveals a novel immunoregulatory role of basophils. Blood, 109(7):2921–2927. https://doi.org/10.1182/blood-2006-07-037739
Oliver JM, Tarleton CA, Gilmartin L, et al., 2010. Reduced FcεRI-mediated release of asthma-promoting cytokines and chemokines from human basophils during omalizumab therapy. Int Arch Allergy Immunol, 151(4):275–284. https://doi.org/10.1159/000250436
Petty AJ, Dai R, Lapalombella R, et al., 2021. Hedgehog-induced PD-L1 on tumor-associated macrophages is critical for suppression of tumor-infiltrating CD8+ T cell function. JCI Insight, 6(6):e146707. https://doi.org/10.1172/jci.insight.146707
Prokopchuk O, Liu Y, Henne-Bruns D, et al., 2005. Interleukin-4 enhances proliferation of human pancreatic cancer cells: evidence for autocrine and paracrine actions. Br J Cancer, 92(5):921–928. https://doi.org/10.1038/sj.bjc.6602416
Redrup AC, Howard BP, Macglashan DW, et al., 1998. Differential regulation of IL-4 and IL-13 secretion by human basophils: their relationship to histamine release in mixed leukocyte cultures. J Immunol, 160(4): 1957–1964.
Rohner L, Reinhart R, Hagmann B, et al., 2018. FcεRI cross-linking and IL-3 protect human basophils from intrinsic apoptotic stress. J Allergy Clin Immunol, 142(5):1647–1650.e3. https://doi.org/10.1016/j.jaci.2018.06.040
Romagnani S, 2002. Cytokines and chemoattractants in allergic inflammation. Mol Immunol, 38(12–13):881–885. https://doi.org/10.1016/S0161-5890(02)00013-5
Romero JM, Grünwald B, Jang GH, et al., 2020. A four-chemokine signature is associated with a T-cell-inflamed phenotype in primary and metastatic pancreatic cancer. Clin Cancer Res, 26(8):1997–2010. https://doi.org/10.1158/1078-0432.Ccr-19-2803
Russo RC, Garcia CC, Teixeira MM, et al., 2014. The CXCl8/IL-8 chemokine family and its receptors in inflammatory diseases. Expert Rev Clin Immunol, 10(5):593–619. https://doi.org/10.1586/1744666X.2014.894886
Saito H, Hatake K, Dvorak AM, et al., 1988. Selective differentiation and proliferation of hematopoietic cells induced by recombinant human interleukins. Proc Natl Acad Sci USA, 85(7):2288–2292. https://doi.org/10.1073/pnas.85.7.2288
Sakaguchi S, Yamaguchi T, Nomura T, et al., 2008. Regulatory T cells and immune tolerance. Cell, 133(5):775–787. https://doi.org/10.1016/j.cell.2008.05.009
Salmiheimo ANE, Mustonen HK, Vainionpää SAA, et al., 2016. Increasing the inflammatory competence of macrophages with IL-6 or with combination of IL-4 and LPS restrains the invasiveness of pancreatic cancer cells. J Cancer, 7(1):42–49. https://doi.org/10.7150/jca.12923
Salter BM, Oliveria JP, Nusca G, et al., 2015. Thymic stromal lymphopoietin activation of basophils in patients with allergic asthma is IL-3 dependent. J Allergy Clin Immunol, 136(6):1636–1644. https://doi.org/10.1016/j.jaci.2015.03.039
Salter BM, Oliveria JP, Nusca G, et al., 2016. IL-25 and IL-33 induce type 2 inflammation in basophils from subjects with allergic asthma. Respir Res, 17:5. https://doi.org/10.1186/s12931-016-0321-z
Sasaki S, Baba T, Nishimura T, et al., 2016. Essential roles of the interaction between cancer cell-derived chemokine, CCL4, and intra-bone CCR5-expressing fibroblasts in breast cancer bone metastasis. Cancer Lett, 378(1):23–32. https://doi.org/10.1016/j.canlet.2016.05.005
Schneider E, Petit-Bertron AF, Bricard R, et al., 2009. IL-33 activates unprimed murine basophils directly in vitro and induces their in vivo expansion indirectly by promoting hematopoietic growth factor production. J Immunol, 183(6): 3591–3597. https://doi.org/10.4049/jimmunol.0900328
Schroeder JT, 2011. Basophils: emerging roles in the pathogenesis of allergic disease. Immunol Rev, 242(1):144–160. https://doi.org/10.1111/j.1600-065X.2011.01023.x
Schroeder JT, Bieneman AP, 2017. Activation of human basophils by A549 lung epithelial cells reveals a novel IgE-dependent response independent of allergen. J Immunol, 199(3):855–865. https://doi.org/10.4049/jimmunol.1700055
Sektioglu IM, Carretero R, Bulbuc N, et al., 2017. Basophils promote tumor rejection via chemotaxis and infiltration of CD8+ T cells. Cancer Res, 77(2):291–302. https://doi.org/10.1158/0008-5472.CAN-16-0993
Shan MM, Carrillo J, Yeste A, et al., 2018. Secreted IgD amplifies humoral T helper 2 cell responses by binding basophils via galectin-9 and CD44. Immunity, 49(4):709–724.e8. https://doi.org/10.1016/j.immuni.2018.08.013
Shankaranarayanan P, Nigam S, 2003. IL-4 induces apoptosis in A549 lung adenocarcinoma cells: evidence for the pivotal role of 15-hydroxyeicosatetraenoic acid binding to activated peroxisome proliferator-activated receptor γ transcription factor. J Immunol, 170(2):887–894. https://doi.org/10.4049/jimmunol.170.2.887
Sharma M, Das M, Stephen-Victor E, et al., 2018. Regulatory T cells induce activation rather than suppression of human basophils. Sci Immunol, 3(23):eaan0829. https://doi.org/10.1126/sciimmunol.aan0829
Shibata S, Miyake K, Tateishi T, et al., 2018. Basophils trigger emphysema development in a murine model of COPD through IL-4-mediated generation of MMP-12-producing macrophages. Proc Natl Acad Sci USA, 115(51):13057–13062. https://doi.org/10.1073/pnas.1813927115
Sinha P, Clements VK, Ostrand-Rosenberg S, 2005. Interleukin-13-regulated M2 macrophages in combination with myeloid suppressor cells block immune surveillance against metastasis. Cancer Res, 65(24):11743–11751. https://doi.org/10.1158/0008-5472.CAN-05-0045
Siracusa MC, Saenz SA, Hill DA, et al., 2011. TSLP promotes interleukin-3-independent basophil haematopoiesis and type 2 inflammation. Nature, 477(7363):229–233. https://doi.org/10.1038/nature10329
Siracusa MC, Kim BS, Spergel JM, et al., 2013. Basophils and allergic inflammation. J Allergy Clin Immunol, 132(4): 789–801. https://doi.org/10.1016/j.jaci.2013.07.046
Smithgall MD, Comeau MR, Yoon BRP, et al., 2008. IL-33 amplifies both Th1-and Th2-type responses through its activity on human basophils, allergen-reactive Th2 cells, iNKT and NK cells. Int Immunol, 20(8):1019–1030. https://doi.org/10.1093/intimm/dxn060
Spranger S, Bao RY, Gajewski TF, 2015. Melanoma-intrinsic β-catenin signalling prevents anti-tumour immunity. Nature, 523(7559):231–235. https://doi.org/10.1038/nature14404
Sullivan BM, Liang HE, Bando JK, et al., 2011. Genetic analysis of basophil function in vivo. Nat Immunol, 12(6):527–535. https://doi.org/10.1038/ni.2036
Tang S, Xiang T, Huang S, et al., 2016. Ovarian cancer stem-like cells differentiate into endothelial cells and participate in tumor angiogenesis through autocrine CCL5 signaling. Cancer Lett, 376(1):137–147. https://doi.org/10.1016/j.canlet.2016.03.034
Terme M, Tartour E, Taieb J, 2013. VEGFA/VEGFR2-targeted therapies prevent the VEGFA-induced proliferation of regulatory T cells in cancer. Oncoimmunology, 2(8):e25156. https://doi.org/10.4161/onci.25156
Todaro M, Zerilli M, Ricci-Vitiani L, et al., 2006. Autocrine production of interleukin-4 and interleukin-10 is required for survival and growth of thyroid cancer cells. Cancer Res, 66(3):1491–1499. https://doi.org/10.1158/0008-5472.CAN-05-2514
Todaro M, Alea MP, di Stefano AB, et al., 2007. Colon cancer stem cells dictate tumor growth and resist cell death by production of interleukin-4. Cell Stem Cell, 1(4):389–402. https://doi.org/10.1016/j.stem.2007.08.001
Varricchi G, Pecoraro A, Marone G, et al., 2018. Thymic stromal lymphopoietin isoforms, inflammatory disorders, and cancer. Front Immunol, 9:1595. https://doi.org/10.3389/fimmu.2018.01595
Volonté A, di Tomaso T, Spinelli M, et al., 2014. Cancer-initiating cells from colorectal cancer patients escape from T cell-mediated immunosurveillance in vitro through membrane-bound IL-4. J Immunol, 192(1):523–532. https://doi.org/10.4049/jimmunol.1301342
Wada T, Ishiwata K, Koseki H, et al., 2010. Selective ablation of basophils in mice reveals their nonredundant role in acquired immunity against ticks. J Clin Invest, 120(8): 2867–2875. https://doi.org/10.1172/jci42680
Walens A, Dimarco AV, Lupo R, et al., 2019. CCL5 promotes breast cancer recurrence through macrophage recruitment in residual tumors. eLife, 8:e43653. https://doi.org/10.7554/eLife.43653
Wanderley CW, Colón DF, Luiz JPM, et al., 2018. Paclitaxel reduces tumor growth by reprogramming tumor-associated macrophages to an M1 profile in a TLR4-dependent manner. Cancer Res, 78(20):5891–5900. https://doi.org/10.1158/0008-5472.Can-17-3480
Wang FR, Li B, Wei YC, et al., 2018. Tumor-derived exosomes induce PD1+ macrophage population in human gastric cancer that promotes disease progression. Oncogenesis, 7(5): 41. https://doi.org/10.1038/s41389-018-0049-3
Webb LM, Oyesola OO, Früh SP, et al., 2019. The Notch signaling pathway promotes basophil responses during helminth-induced type 2 inflammation. J Exp Med, 216(6): 1268–1279. https://doi.org/10.1084/jem.20180131
Williford JM, Ishihara J, Ishihara A, et al., 2019. Recruitment of CD103+ dendritic cells via tumor-targeted chemokine delivery enhances efficacy of checkpoint inhibitor immunotherapy. Sci Adv, 5(12):eaay1357. https://doi.org/10.1126/sciadv.aay1357
Yoshikawa S, Oh-Hora M, Hashimoto R, et al., 2019. Pivotal role of STIM2, but not STIM1, in IL-4 production by IL-3-stimulated murine basophils. Sci Signal, 12(576): eaav2060. https://doi.org/10.1126/scisignal.aav2060
Yoshimoto T, Yasuda K, Tanaka H, et al., 2009. Basophils contribute to TH2-IgE responses in vivo via IL-4 production and presentation of peptide-MHC class II complexes to CD4+ T cells. Nat Immunol, 10(7):706–712. https://doi.org/10.1038/ni.1737
Zellweger F, Buschor P, Hobi G, et al., 2018. IL-3 but not monomeric IgE regulates FcεRI levels and cell survival in primary human basophils. Cell Death Dis, 9(5): 510. https://doi.org/10.1038/s41419-018-0526-9
Zhang QF, Qin JL, Zhong L, et al., 2015. CCL5-mediated Th2 immune polarization promotes metastasis in luminal breast cancer. Cancer Res, 75(20):4312–4321. https://doi.org/10.1158/0008-5472.CAN-14-3590
Zhang SB, Zhong M, Wang C, et al., 2018. CCL5-deficiency enhances intratumoral infiltration of CD8+ T cells in colorectal cancer. Cell Death Dis, 9(7): 766. https://doi.org/10.1038/s41419-018-0796-2
Zhang WJ, Li BH, Yang XZ, et al., 2008. IL-4-induced Stat6 activities affect apoptosis and gene expression in breast cancer cells. Cytokine, 42(1):39–47. https://doi.org/10.1016/j.cyto.2008.01.016
Zhang Y, He SJ, Mei RM, et al., 2018. miR-29a suppresses IL-13-induced cell invasion by inhibiting YY1 in the AKT pathway in lung adenocarcinoma A549 cells. Oncol Rep, 39(6):2613–2623. https://doi.org/10.3892/or.2018.6352
Zhang Y, Meng WB, Yue P, et al., 2020. M2 macrophage-derived extracellular vesicles promote gastric cancer progression via a microRNA-130b-3p/MLL3/GRHL2 signaling cascade. J Exp Clin Cancer Res, 39:134. https://doi.org/10.1186/s13046-020-01626-7
Zhao N, Dang H, Ma LC, et al., 2021. Intratumoral γδ T-cell infiltrates, chemokine (C-C motif) ligand 4/chemokine (C-C motif) ligand 5 protein expression and survival in patients with hepatocellular carcinoma. Hepatology, 73(3): 1045–1060. https://doi.org/10.1002/hep.31412
Zheng ZY, Jia SF, Shao CS, et al., 2020. Irradiation induces cancer lung metastasis through activation of the cGAS-STING-CCL5 pathway in mesenchymal stromal cells. Cell Death Dis, 11(5):326. https://doi.org/10.1038/s41419-020-2546-5
Ziegler SF, Artis D, 2010. Sensing the outside world: TSLP regulates barrier immunity. Nat Immunol, 11(4):289–293. https://doi.org/10.1038/ni.1852
Acknowledgments
This work was supported by the Shanghai Sailing Program (No. 21YF1407100), the China Postdoctoral Science Foundation (No. 2021M690037), the National Natural Science Foundation of China (Nos. 82103409 and 81773068), and the National Key R&D Program of China (No. 2019YFC1315902).
Author information
Authors and Affiliations
Contributions
Jicheng ZHANG, Ning PU, and Wenchuan WU came up with the idea of the manuscript. Jicheng ZHANG, Hanlin YIN, Qiangda CHEN, and Guochao ZHAO wrote the manuscript. Ning PU, Wenhui LOU, and Wenchuan WU revised the manuscript. All authors have read and approved the final manuscript, and therefore, take responsibility for the integrity of the study.
Corresponding authors
Ethics declarations
Jicheng ZHANG, Hanlin YIN, Qiangda CHEN, Guochao ZHAO, Wenhui LOU, Wenchuan WU, and Ning PU declare that they have no conflicts of interests.
This article does not contain any studies with human or animal subjects performed by any of the authors.
Rights and permissions
About this article
Cite this article
Zhang, J., Yin, H., Chen, Q. et al. Basophils as a potential therapeutic target in cancer. J. Zhejiang Univ. Sci. B 22, 971–984 (2021). https://doi.org/10.1631/jzus.B2100110
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2100110