Abstract
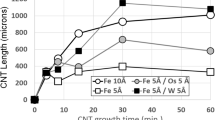
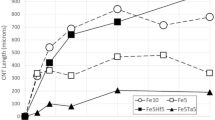
Catalytic growth of substantial amounts of Carbon Nanotubes (CNTs) to lengths greater than 1–2 cm is currently limited by several factors, including especially the deactivation of the catalyst particles due to erosion of catalyst atoms from the catalyst particles at elevated CNT growth temperatures. Inclusion of refractory metals in the CNT growth catalyst has recently been proposed as a method to prevent this catalytic particle erosion and deactivation, allowing the CNT to grow for greater times and reach substantially greater lengths. Here are presented results of recent investigations into this method. The system investigated employs Molybdenum as the erosion inhibitor and Iron as the CNT growth catalyst. Results show that inclusion of Mo leads to substantially longer catalyst particle lifetimes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
B. Yacobson and R. Smalley. American Scientist 85, 324 (1997).
M. Meo and M. Rossi. Composites Science and Technology 66, 1597 (2006).
B. Peng, M. Locascio, P. Zapol, S. Y. Li, S. L. Mielke, G. C. Schatz, H. D. Espinosa. Nature Nanotechnology 3, 626 (2008).
K. R. Atkinson, S. C. Hawkins, C. Huynh, C. Skourtis, J. Dai, M. Zhang, S. L. Fang, A. A. Zakhidov, S. B. Lee, A. E. Aliev, C. D. Williams, R. H. Baughman. Physica B: Condensed Matter 394, 339 (2007).
M. Zhang, K. R. Atkinson, R. H. Baughman. Science 306, 1358 (2004).
M. J. Bronikowski. Carbon 107, 297 (2016).
L. Ratke and P. Voorhees. (Springer-Verlag, Berlin, 2002), pp. 117, 118. ISBN 3-540-42563-2
R. D. Vengrenovich, Y. V. Gudyma, and S. V. Yarema. Semiconductors 35, 1378 (2001).
R. F. Zhang, H. H. Xie, Y. Y. Zhang, Q. Zhang, Y. G. Jin, P. Li, W. Z. Qian and F. Wei. Carbon 52, 232 (2013).
M. J. Bronikowski. J. Phys Chem. C 111, 17705 (2007).
J. H. Hafner, M. J. Bronikowski, B. R. Azamian, P. Nikolaev, A. G. Rinzler, D. T. Colbert, K. A. Smith and R. E. Smalley. Chem. Phys. Lett. 296, 195 (1998).
S. Huang, M. Woodson, R. E. Smalley, J. Liu. Nano Letters 4, 1025 (2004).
W. Cho, M. Schulz, and V. Shanov. Carbon 72, 264 (2014).
B. Kitiyanan, W. Alvarez, D. Harwell, and D. Resasco. Chem. Phys Lett. 317, 498 (2000).
G. Y. Xiong, D. Z. Wang, and Z. F. Ren. Carbon 44, 969 (2006).
Y. Yun, V. Shanov, Y. Tu, S. Subramaniam, and M. Schulz. J. Phys. Chem. B 110, 23920 (2006).
A. Puretzky, D. Geohegan, S. Jesse, I. Ivanov, G. Eres. Appl. Phys. A 81, 223 (2005).
Q. W. Li, X. F. Zhang, R. F. DePaula, L. X. Zheng, Y. H. Zhao, L. Stan, T. Holesinger, P. N. Arendt, D. E. Peterson, Y. T. Zhu. Adv. Mater. 18, 3160 (2006).
D. Futaba, K. Hata, T. Yamada, K. Mizuno, M. Yumura, S. Iijima. Phys. Rev. Lett. 95, 056104 (2005).
A. M. Cassell, J. A. Raymakers, J. Kong, H. J. Dai. J. Phys. Chem. B 103, 6484 (1999).
D. Venegoni, D. P. Serp, R. Feurer, Y. Kihn, C. Vahlas, P. Kalck, P. Carbon 40, 1799 (2002).
H. Cui, G. Eres, J. Y. Howe, A. Puretkzy, M. Varela, D. B. Geohegan, D. H. Lowndes. Chem. Phys. Lett. 374, 222 (2003).
H. Dai, A. G. Rinzler, P. Nikolaev, A. Thess, D. T. Colbert, R. E. Smalley. Chem. Phys. Lett. 260, 471 (1996).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bronikowski, M.J., King, M. Refractory-Metal Diffusion Inhibitors Slow Erosion of Catalytic Metal Particles in the growth of Carbon Nanotubes. MRS Advances 4, 197–204 (2019). https://doi.org/10.1557/adv.2018.666
Published:
Issue Date:
DOI: https://doi.org/10.1557/adv.2018.666