Abstract
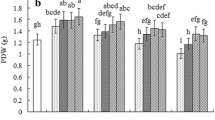
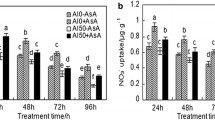
This study investigated the effect of cerium (Ce) on the ascorbate-glutathione (AsA-GSH) cycle in the roots of maize seedlings under salt stress. The results showed that salt stress significantly increased the activities of ascorbate peroxidase (APX) and monodehydroascorbate reductase (MDHAR), malondialdehyde (MDA) content and electrolyte leakage (EL) in the roots of maize seedlings, compared to the control. However, salt stress significantly decreased plant height, dry weight of root and shoot, the activities of glutathione reductase (GR) and dehydroascorbate reductase (DHAR), and the ratios of AsA/DHA and GSH/GSSG in the roots of maize seedlings, compared to the control. Pretreatment with cerium nitrate (Ce(NO3)3) significantly increased plant height, dry weight of root and shoot, the activities of above four enzymes, and the ratios of AsA/DHA and GSH/GSSG under salt stress, compared to salt stress alone. Pretreatment with Ce(NO3)3 significantly decreased MDA content and EL induced by salt stress, compared to salt stress alone. Meanwhile, application of Ce(NO3)3 to the control also significantly increased plant height, dry weight of root and shoot, the activities of above four enzymes and the ratios of AsA/DHA and GSH/GSSG, and decreased MDA content and EL, compared with control. Our results suggested that Ce(NO3)3 alleviated salt stress-induced oxidative damage by improving AsA-GSH cycle in maize roots.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- APX:
-
ascorbate peroxidase
- GR:
-
glutathione reductase
- DHAR:
-
dehydroascorbate reductase
- MDHAR:
-
monodehydroascorbate reductase
- GSH:
-
reduced glutathione
- AsA:
-
reduced ascorbic acid
- MDA:
-
malondialdehyde
- EL:
-
electrolyte leakage
References
Bradford, M.M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72:248–254.
Dai, H., Shan, C., Zhao, H., Jia, G., Chen, D. 2016. Lanthanum improves the cadmium tolerance of Zea mays seedlings by the regulation of ascorbate and glutathione metabolism. Biol. Plant. doi:10.1007/s10535-016-0669-4
Dalton, D.A., Russell, S.A., Hanus, F.J., Pascoe, G.A., Evans, H.J. 1986. Enzymatic reactions of ascorbate and glutathione that prevent peroxide damage in soybean root nodules. Proc. Natl Acad. Sci. USA 83:3811–3815.
Diao, M., Ma, L., Wang, J., Cui, J., Fu, A., Liu, H. 2014. Selenium promotes the growth and photosynthesis of tomato seedlings under salt stress by enhancing chloroplast antioxidant defense system. J. Plant Growth Regul. 33:671–682.
Grace, S.C., Logan, B.A. 1996. Acclimation of foliar antioxidant systems to growth irradiance in three broad-leaved evergreen species. Plant Physiol. 112:1631–1640.
Griffith, O.W. 1980. Determination of glutathione and glutathione disulfide using glutathione reductase and 2-vinylpyridine. Anal. Biochem. 106:207–212.
Hodges, D.M., Andrews, C.J., Johnson, D.A., Hamilton, R.I. 1996. Antioxidant compound responses to chilling stress in differentially sensitive inbred maize lines. Plant Physiol. 98:685–692.
Hodges, M.D., DeLong, J.M., Forney, C.F., Prange, R.K. 1999. Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 207:604–611.
Hu, M., Shi, Z., Zhang, Z., Zhang, Y., Li, H. 2012. Effects of exogenous glucose on seed germination and antioxidant capacity in wheat seedlings under salt stress. Plant Growth Regul. 68:177–188.
Jouyban, Z. 2012. The effects of salt stress on plant growth. Tech. J. Engin. App. Sci. 2:7–10.
Liu, R., Shan, C., Gao, Y., Wang, J., Xu, Z., Zhang, L., Ma, W., Tan, R. 2016. Cerium improves the copper tolerance of turf grass Poa pratensis by affecting the regeneration and biosynthesis of ascorbate and glutathione in leaves. Braz. J. Bot. 39:779–785.
Liu, S.G., Zhu, D.Z., Chen, G.H., Gao, X.Q., Zhang, X.S. 2012. Disrupted actin dynamics trigger an increment in the reactive oxygen species levels in the Arabidopsis root under salt stress. Plant Cell Rep. 31:1219–1226.
Mao, C.X., Chen, M.M., Wang, L., Zou, H., Liang, C.J., Wang, L.H., Zhou, Q. 2012. Protective effect of cerium ion against ultraviolet B radiation-induced water stress in soybean seedlings. Biol. Trace Elem. Res. 146:381–387.
Miyake, C., Asada, K. 1992. Thylakoid-bound ascorbate peroxidase in spinach chloroplasts and photoreduction of its primary oxidation product monodehydroascorbate radicals in thylakoids. Plant Cell Physiol. 33:541–553.
Muchate, N.S., Nikalje, G.C., Rajurkar, N.S., Suprasanna, P., Nikam, T.D. 2016. Plant salt stress: adaptive responses, tolerance mechanism and bioengineering for salt tolerance. Bot. Rev. 82:371–406.
Nakano, Y., Asada, K. 1981. Hydrogen peroxide is scavenged by ascorbate specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 22:867–880.
Qu, C., Liu, C., Guo, F., Hu, C., Ze, Y., Li, C., Zhou, Q., Hong, F. 2013. Improvement of cerium on photosynthesis of maize seedlings under a combination of potassium deficiency and salt stress. Biol. Trace Elem. Res. 155:104–113.
Rossi, L., Zhang, W., Lombardini, L., Ma, X. 2016. The impact of cerium oxide nanoparticles on the salt stress responses of Brassica napus L. Environmental Pollution 219:28–36.
Shan, C., Liu, H., Zhao, L., Wang, X. 2014. Effects of exogenous hydrogen sulfide on the redox states of ascorbate and glutathione in maize leaves under salt stress. Biol. Plant. 58:169–173.
Shan, C., Zhao, X. 2014. Effects of lanthanum on the ascorbate and glutathione metabolism of Vigna radiate seedlings under salt stress. Biol. Plant. 58:595–599.
Shan, C., Zhou, Y., Liu, M. 2015. Nitric oxide participates in the regulation of the ascorbate-glutathione cycle by exogenous jasmonic acid in the leaves of wheat seedlings under drought stress. Protoplasma 252:1397–1405.
Wu, M., Wang, P.Y., Sun, L.G., Zhang, J.J., Yu, J., Wang, Y.W., Chen, G.X. 2014. Alleviation of cadmium toxicity by cerium in rice seedlings is related to improved photosynthesis, elevated antioxidant enzymes and decreased oxidative stress. Plant Growth Regul. 74:251–260.
Yousuf, P.Y., Ahmad, A., Ganie, A.H., Sareer, O., Krishnapriya, V., Aref, I.M., Iqbal, M. 2017. Antioxidant response and proteomic modulations in Indian mustard grown under salt stress. Plant Growth Regul. 81:31–50.
Zhao, L.Q., Zhang, F., Guo, J.K., Yang, Y.L., Li, B.B., Zhang, L.X. 2004. Nitric oxide functions as a signal in salt resistance in the calluses from two ecotypes of reed. Plant Physiol. 134:849–857.
Acknowledgement
This study was funded by the Science and Technology Program of Henan Province (142102110041).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Gottwald
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Hu, H., Shan, C. Effect of Cerium (Ce) on the Redox States of Ascorbate and Glutathione through Ascorbate-glutathione Cycle in the Roots of Maize Seedlings under Salt Stress. CEREAL RESEARCH COMMUNICATIONS 46, 31–40 (2018). https://doi.org/10.1556/0806.45.2017.057
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/0806.45.2017.057