Abstract
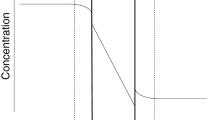
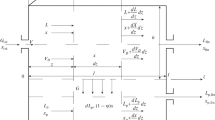
The presented paper deals with the numerical analysis of pseudo-steady state conditions used in the modelling of the batch dialysis process. First, under specified conditions, time dependences of component concentrations and liquid volumes in both compartments were generated using a rigorous model based on Fick’s second law. From these data, the diffusion coefficient of the component in the membrane was calculated using a simplified model based on Fick’s first law. The specified coefficient was then compared with the calculated one. Numerical analysis revealed that in case of not too thick membranes, sufficiently high values of the diffusion coefficient, high intensity of mixing and considering the concentration and volume data in compartments I and II, the pseudo-steady state conditions can be considered as a good approximation of the real state.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Akgemci, E. G., Ersöz, M., & Atalay, T. (2005). Transport of formic acid through anion exchange membranes by diffusion dialysis and electro-electro dialysis. Separation Science and Technology, 39, 165–184. DOI: 10.1081/ss-120027407.
Alexandrova, I., & Iordanov, G. (2005). Transport of cadmium and iron through a carboxylic membrane based on poly(vinyl chloride)/poly(methyl methacrylate-co-divinyl benzene) system. Journal of Applied Polymer Science, 95, 705–707. DOI: 10.1002/app.21230.
Allen, C. A., Cummings, D. G., & McCaffrey, R. R. (1989). Separation of Cr ions from Co and Mn ions by poly[bis(trifluoroethoxy)phosphazene] membranes. Journal of Membrane Science, 43, 217–228. DOI: 10.1016/s0376-7388(00)85099-1.
Audinos, R. & Pichelin, G. (1988). Characterization of electrodialysis membranes by chronopotentiometry. Desalination, 68, 251–263. DOI: 10.1016/0011-9164(88)80059-6.
Elmidaoui, A., Cherif, A. T., Molenat, J., & Gavach, C. (1995). Transfer of H2SO4,Na2SO4 and ZnSO4 by dialysis through an anion exchange membrane. Desalination, 101, 39–46. DOI: 10.1016/0011-9164(95)00006-n.
Ersoz, M., Gugul, I. H., & Sahin, A. (2001). Transport of acids through polyether-sulfone anion-exchange membrane. Journal of Colloid and Interface Science, 237, 130–135. DOI: 10.1006/jcis.2001.7487.
Hao, J. W., Gong, M., Wu, Y. H., Wu, C. M., Luo, J.Y., & Xu, T. W. (2013a). Alkali recovery using PVA/SiO2 cation exchange membranes with different —COOH contents. Journal of Hazardous Materials, 244–245, 348–356. DOI: 10.1016/j.jhazmat.2012.11.056.
Hao, J. W., Wu, Y. H., & Xu, T. W. (2013b). Cation exchange hybrid membranes prepared from PVA and multisilicon copolymer for application in alkali recovery Journal of Membrane Science, 425–426, 156–162. DOI: 10.1016/j.memsci.2012.09.024.
Heintz, A., & Illenberger, C. (1996). Diffusion coefficients of Br2 in cation exchange membranes. Journal of Membrane Science, 113, 175–181. DOI: 10.1016/0376-7388(95)00026-7.
Kaczmarek, M. S., Ładziński, P., Woźniak, Z., Błaszczak, Z., Surma, M., & Pochylski, M. (2008). Dynamics of transportation of glucose and urea water solutions through a porous polyethyleneterephthalate membrane studied by optical interference. Journal of Molecular Liquids, 138, 168–172. DOI: 10.1016/j.molliq.2007.10.002.
Kang, M. S., Yoo, K. S., Oh, S. J., & Moon, S. H. (2001). A lumped parameter model to predict hydrochloric acid recovery in diffusion dialysis. Journal of Membrane Science, 188, 61–70. DOI: 10.1016/s0376-7388(01)00372-6.
Luo, J. Y., Wu, C. M., Wu, Y. H., & Xu, T. W. (2010). Diffusion dialysis of hydrochloric acid at different temperatures using PPO-SiO2 hybrid anion exchange membranes. Journal of Membrane Science, 347, 240–249. DOI: 10.1016/j.memsci.2009.10.029.
Narebska, A., & Warszawski, A. (1992). Diffusion dialysis. Effect of membrane composition on acid/salt separation. Separation Science and Technology, 27, 703–715. DOI: 10.1080/01496399208019719.
Narebska, A., & Staniszewski, M. (1997). Separation of fermentation products by membrane techniques. I. Separation of lactic acid/lactates by diffusion dialysis. Separation Science and Technology, 32, 1669–1682. DOI: 10.1080/01496399708000727.
Narebska, A., & Staniszewski, M. (2008). Separation of carboxylic acids from carboxylates by diffusion dialysis. Separation Science and Technology, 43, 490–501. DOI: 10.1080/01496390701787388.
Palatý, Z., & Žáková, A. (2000). Apparent diffusivity of some inorganic acids in anion-exchange membrane. Journal of Membrane Science, 173, 211–223. DOI: 10.1016/s0376-7388(00)00363-x.
Palatý, Z., Stoček, P., Žáková, A., & Bendová, H. (2007). Transport characteristics of some carboxylic acids in the polymeric anion-exchange membrane Neosepta-AMH: Batch experiments. Journal of Applied Polymer Science, 106, 909–916. DOI: 10.1002/app.26516.
Palatý, Z., Kaláb, J., & Bendová, H. (2010). Transport properties of propionic acid in anion-exchange membrane Neosepta-AFN. Journal of Membrane Science, 349, 90–96. DOI: 10.1016/j.memsci.2009.11.034.
Sudoh, M., Kamei, H., & Nakamura, S. (1987). Donnan dialysis concentration of cupric ions. Journal of Chemical Engineering of Japan, 20, 34–40. DOI: 10.1252/jcej.20.34.
Suhara, M., Suzuki, K., Horie, H., & Shimohira, T. (1989). Transport numbers in perfluorocarboxylate cation-exchange membrane. Journal of Membrane Science, 41, 143–153. DOI: 10.1016/s0376-7388(00)82397-2.
Wu, C. M., Gu, J. J., Wu, Y. H., Luo, J. Y., Xu, T. W., & Zhang, Y. P. (2012). Carboxylic acid type PVA-based hybrid membranes for alkali recovery using diffusion dialysis. Separation and Purification Technology, 92, 21–29. DOI: 10.1016/j.seppur.2012.03.014.
Xu, T. W., & Yang, W. H. (2001). Sulfuric acid recovery from titanium white (pigment) waste liquor using diffusion dialysis with a new series of anion exchange membranes — static runs. Journal of Membrane Science, 183, 193–200. DOI: 10.1016/s0376-7388(00)00590-1.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Palatý, Z., Bendová, H. Determination of diffusivity from mass transfer measurements in a batch dialyzer: numerical analysis of pseudo-steady state approximation. Chem. Pap. 69, 560–568 (2015). https://doi.org/10.1515/chempap-2015-0070
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1515/chempap-2015-0070