Abstract
Background
Enhanced Recovery Protocols (ERPs) provide a multimodal approach to perioperative care, with the aims of improving patient outcomes while decreasing perioperative antiemetic and narcotic requirements. With high rates of post-operative nausea or vomiting (PONV) following total mastectomy (TM), we hypothesized that our institutional designed ERP would reduce PONV while improving pain control and decrease opioid use.
Methods
An ERP was implemented at a single institution for patients undergoing TM with or without implant-based reconstruction. Patients from the first two months of implementation (ERP group, N = 72) were compared with a retrospective usual-care cohort from a three-month period before implementation (UC group, N = 83). Outcomes included PONV incidence, measured with antiemetic use; patient-reported pain scores; perioperative opioid consumption, measured by oral morphine equivalents (OME); and length of stay (LOS).
Results
The characteristics of the two groups were similar. PONV incidence and perioperative opioid consumption were lower in the ERP than the UC group (21% vs. 40%, p 0.011 and mean 44.1 OME vs. 104.3 OME, p < 0.001), respectively. These differences in opioid consumption were observed in the operating room and post-anesthesia care unit (PACU); opioid consumption on the floor was similar between the two groups. Patient-reported pain scores were lower in the ERP than the UC group (mean highest pain score 6.4 vs. 7.4, p 0.003). PACU and hospital LOS were similar between the two groups.
Conclusion
ERP implementation was successful in decreasing PONV following TM with and without reconstruction, while simultaneously decreasing overall opioid consumption without compromising patient comfort.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Enhanced Recovery Protocols (ERPs) utilize a multimodal, multidisciplinary approach to the care of the surgical patient. This approach enables clinical caregivers to design a coordinated protocol, where the components work synergistically to optimize the shared goals of improved patient recovery. While the term fast-track surgery has sometimes been used, the key aim is the quality, rather than the speed, of recovery.1
ERPs developed by various surgical subspecialties are aimed at improving patient outcomes, specifically by decreasing post-operative pain via a multimodality approach to analgesia that also reduces perioperative opioid consumption, postoperative nausea and vomiting (PONV), postoperative pain and hospital length of stay. The subspecialties include but are not limited to colorectal,2,3,4,5,6 other intra-abdominal,7,8,9,10 pelvic,11,12 and orthopedic13,14 surgery. ERPs have also been developed for mastectomy patients undergoing autologous reconstruction.15 However, ERPs have not been well studied in mastectomy patients who are not undergoing autologous reconstruction. This may be particularly important, especially with regard to PONV, which has been reported to be as high as 80% after mastectomy.16,17,18
As part of a system wide effort to reduce narcotic administration and decrease contributions to the opioid epidemic, while using an evidence-based approach with preceding ERPs in other specialties and procedures, we developed an institution specific ERP for patients undergoing total mastectomy, either without reconstruction or with implant-based reconstruction. The protocol was built upon review of other standardized protocols for breast surgery including the published University of California, San Francisco model in 2018.19 The goals were to decrease postoperative pain, perioperative opioid consumption and hospital length of stay. We hypothesize that the ERP would additionally decrease PONV and report our experience with implementation of the protocol and its associated improvements in patient recovery.
Methods
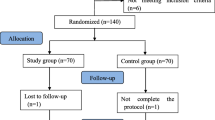
The ERP for total mastectomy patients was initiated at a tertiary referral, academic institution on August 20, 2018. With approval from the Quality Improvement Review Committee, we performed a single-institution chart review of all patients who underwent total mastectomy with implant-based reconstruction or without any reconstruction. Data for the reference group of patients in the usual care (UC) arm were retrospectively collected in the time period before breast ERP initiation, from June to August 2018. Data from the comparison group of patients cared for with the ERP were prospectively collected during the time-period of September to October 2018. We excluded the first several ERP patients from August 20 through August 31, 2018, to eliminate any inconsistencies or deviations in ERP implementation during the early part of transition from UC to ERP based care.
All patients cared for at a single institution by a group of dedicated breast surgeons with coordinated care from dedicated reconstructive surgeons undergoing a mastectomy for a diagnosis of breast cancer or risk reduction surgery were included in the analysis. The cohort also included a small number of transgender patients undergoing a mastectomy as part of their gender affirmation surgery. Patients undergoing concurrent bilateral salpingo-oophorectomy were included. Exclusion criteria included concurrent total hysterectomy with bilateral salpingo-oophorectomy, tissue-based reconstruction, or any other major surgical procedure under general anesthesia.
The UC group consisted of patients who underwent the procedure prior to initiation of the ERP. There was no existing, standardized pre-operative counseling packet. The choice for intraoperative anesthetic was as the discretion of the anesthesiologist. Intraoperative blocks were not standard part of care for the breast or reconstructive surgeon and the mainstay for post-operative pain control was opioid-based medication both in the hospital and upon discharge.
In contrast, the ERP was designed by a multidisciplinary team, after review of the literature and institutional experience with ERP for other surgical procedures. The protocol consisted of three phases: preoperative, intraoperative, and postoperative. Each phase included medication and counseling components (Fig. 1).
The preoperative phase began in the surgical clinic. Patients received education about the ERP, as well as smoking-cessation and nutrition counseling, to help set stage for the patient and frame the recovery process. They were advised to walk daily for at least 30 min for pre-habilitation. Patients were instructed to avoid prolonged fasting; solid food was allowed up to 8 h before surgery, and clear liquids were allowed up to 3 h before surgery. Preoperative medications in the holding area included oral acetaminophen 1000 mg, oral gabapentin 400 mg, and oral perphenazine 8 mg.
Intraoperative anesthetic management included general anesthesia with propofol-based total intravenous anesthesia, intravenous dexamethasone 4–5 mg after induction, intravenous ondansetron 4 mg during emergence, and minimal use of opioids. Intravenous boluses of ketamine or dexmedetomidine were administered as needed, at the discretion of the anesthesia provider. Following removal of the breasts, the breast surgeon performed a Pecs I and Pecs II block, and a serratus anterior block,20,21,22 using a total of 30 ml of bupivacaine 0.5% with epinephrine 1:200,000. For bilateral cases, the bupivacaine was diluted with an additional 30 ml of saline for a total volume of 60 ml evenly divided between both sides. Intravenous ketorolac 15 mg was administered at the end of the case. Patients were kept euvolemic and normothermic perioperatively.
Postoperatively, patients received multimodal analgesia. This included intravenous acetaminophen 1000 mg Q6H for two doses, before transitioning to oral acetaminophen 1000 mg Q6H; intravenous ketorolac 15 mg Q6H for four doses, before transitioning to oral ibuprofen 600 mg Q6H; oral oxycodone 5–10 mg Q4H PRN pain; and intravenous hydromorphone 0.5 mg Q2H PRN breakthrough pain. Patients could request an alternative oral opioid if they did not wish to take oxycodone, usually due to previous side effects. Patients were encouraged to ambulate on post-operative day (POD) 0, and they were immediately provided with a solid diet. Patients met discharge criteria if they tolerated a solid diet, were independent with mobility and ambulation, had adequate pain control with oral analgesics, and did not have any signs of early complications. Patients were given a discharge goal of POD 1,but could be discharged on POD 0 if they were motivated and met discharge criteria. Patients were kept in-hospital beyond POD 1 if there were any clinical concerns, if additional time was needed to arrange for social services, or if additional time was needed for continued inpatient pain or nausea control.
The electronic health record was reviewed for patient demographic and clinical characteristics, medication administration, numerical rating scale (NRS) pain scores, and length of stay. Outcomes included PONV, postoperative pain, perioperative opioid use, and length of stay.
PONV was defined as the use of any antiemetic, including ondansetron, prochlorperazine, and metoclopramide, in either the post-anesthetic care unit (PACU) or on the hospital floor. Postoperative pain was measured using the NRS pain score, which was on a scale from zero (no pain) to ten (worst pain imaginable). Each patient’s highest NRS pain score and average NRS pain scores were recorded, both in the PACU and on the hospital floor. Perioperative opioid consumption was calculated by converting opioid use into oral morphine equivalents. The calculations were done based on the institutional opioid equivalence table. Patients with a past history of opioid abuse or dependence were excluded from the analysis of perioperative opioid consumption.
Variables between the UC and ERP groups were compared using the student’s t test or χ2 test for continuous and categorical variables, respectively. All measures of statistical significance were measured at an alpha level of 0.05. All statistical analysis was performed using Stata version 15 (College Station, TX).
Results
A total of 83 UC patients and 72 ERP patients were included in the analysis. Demographic and clinical characteristics of the two groups were similar (Table 1).
Overall anti-emetic administration, as a surrogate for PONV, was lower in the ERP group compared to the UC group, 21% vs. 40% (p = 0.011). This trend was statistically significant in the hospital floor setting but not in the PACU (Table 2).
The mean patient-reported NRS pain scores were lower in the ERP group compared to the UC group, both in the PACU (2.6 vs. 4.1, p < 0.001) and in the hospital floor setting (3.3 vs. 4.5, p < 0.001). The mean highest-reported pain score was also lower in the ERP group compared to the UC group, 6.4 vs. 7.4 (p = 0.003) (Table 3).
Overall perioperative opioid consumption was lower in the ERP group compared to the UC group, 44.1 OME vs. 104.3 OME, p < 0.001. When stratified into perioperative phases, the ERP group had lower opioid consumption than the UC group in the intraoperative setting (14.9 OME vs. 50.4 OME, p < 0.001) and PACU (8.7 OME vs. 21.8 OME, p < 0.001). The hospital floor opioid consumption was similar between the two groups (23.1 OME in the ERP group vs. 32.4 OME in the UC group, p = 0.18) (Table 4).
The mean PACU length of stay for the patients was similar in the two groups (154 min in the ERP group and 165 min in the UC group, p = 0.205). The majority of patients were discharged on POD 1 in both groups: 90% of the ERP patients and 88% of the UC patients (Table 5). For those patients who had a delayed discharge beyond POD 1, the reasons for delayed discharge are listed in Table 6.
Discussion
This study confirmed the hypothesis that patients in the ERP group had a lower incidence of PONV, likely secondary to several strategies including multi-modal non-narcotic pre-operative pain control, use of pre-operative and intraoperative anti-emetics, and a deliberate preference for non-opioid medications during the intraoperative phase and avoidance of volatile anesthetics. The pattern of decreased PONV that is more striking in the hospital floor setting but not in the PACU is consistent with findings from implementation of an ERP for total mastectomy patients at the University of California, San Francisco.19 We postulate that this trend could be related to the relatively low baseline incidence of PONV in the PACU compared to the floor, even in the UC group.
This finding is of importance because the incidence of PONV following mastectomy is reported to be as high as 80% and is likely related to the use of volatile anesthetics. As health systems shift focus to value based care, successful reduction in PONV will also result in improved patient satisfaction.16,23,24 Additionally, cost-effectiveness analyses have demonstrated benefits to PONV prophylaxis compared to therapy, when including both the direct and indirect costs.23,25 Some interventions shown to reduce PONV include intraoperative antiemetic use and limited intraoperative opioid use.16,17,18 Our study describes the implementation of an ERP for patients undergoing total mastectomy, either without reconstruction or with implant-based reconstruction, at a single institution. The aim of decreasing procedure-associated PONV while concurrently decreasing overall opioid consumption was achieved by design with the ERP, without compromising patient comfort. There was no difference in hospital length of stay pre- or post-ERP implementation.
These systematic efforts are important as decreasing perioperative opioid consumption has become a critical component in the fight against the opioid epidemic. New persistent opioid use is a common iatrogenic complication in cancer patients undergoing curative-intent surgery, including breast cancer surgery.26 Multimodal analgesic strategies, such as those used in this ERP, can decrease perioperative opioid consumption without compromising patient comfort in the recovery period. These strategies can be used in conjunction with increased prescriber awareness of actual perioperative opioid requirements, that in turn will lead to a decrease in narcotic over-prescription.27
It is interesting to note that there were no statistically significant differences in total OME use for the ERP group compared to the UC group in the hospital floor setting, despite a statistically significant lower patient reported NRS pain score. A potential explanation is the on-demand nature by which opioid medications are administered in the hospital floor setting. Despite parameters that are set for type of medication to be given based upon a patient reported NRS pain score, the retrospective design of this study does not allow for precise identification of the root cause of this finding.
There were no observed difference in length of stay between the UC or ERP groups, either in the PACU or in the overall hospitalization because the majority of patients at baseline were discharged on POD 1. Other modern studies describing ERP for breast cancer surgery with autologous reconstruction have shown either similar findings or do not measure LOS due to the short baseline timeframe.25,28,29
Limitations to our study include the single-institution setting and associated small cohort size. The patients were not formally randomized to treatment groups. Though the two groups were well-balanced with regard to baseline demographic and clinical characteristics, there may have been influence on outcomes related to unmeasurable confounders, such as institutional awareness of patient recovery after the ERP implementation. Furthermore, post-discharge opioid and anti-emetic consumption was not measured.
Our results are consistent with other recently reported studies describing multimodal ERP for breast cancer surgery.25,28,29 However, one of the unique characteristics of this study is the observation that not only can post-operative analgesia be achieved with a decreased opioid requirement, it in turn results in a decrease in PONV, a significant problem in breast surgery.18 To our knowledge, there is only one other study from UCSF that has reported this observation.19
With the increasing prevalence of ERP for breast cancer surgery, collaborative adjustments to these protocols can further improve the recovery of breast cancer patients. With our expanded experience with this ERP protocol, some of the improvements that have since been implemented include providing the regional anesthetic block prior to surgery start instead of intraoperatively. It is expected that these refinements will continue to improve the patient experience with breast surgery, as we aim to eliminate PONV while providing appropriate pain control for an extended period of time in the post-operative setting.
References
Ljungqvist O, Scott M, Fearon KC. Enhanced recovery after surgery a review. JAMA Surg. 2017;152(3):292–8. https://doi.org/10.1001/jamasurg.2016.4952
Badram L, Funch-Jensen P, Jensen P, Crawford ME KH. Recovery after laparoscopic colonic surgery with epidural analgesia, and early. Lancet. 1995;345:763–4.
Kehlet H, Mogensen T. Hospital stay of 2 days after open sigmoidectomy with a multimodal rehabilitation programme. Br J Surg. 1999;86(2):227–30.
Rix T, Jourdan L, Delaney CP, Senagore AJ, Remzi FH, Fazio VW. “Fast track” postoperative management protocol for patients with high co-morbidity undergoing complex abdominal and pelvic colorectal surgery. Br J Surg. 2001;88:15331538.
Varadhan KK, Neal KR, Dejong CHC, Fearon KCH, Ljungqvist O, Lobo DN. The enhanced recovery after surgery (ERAS) pathway for patients undergoing major elective open colorectal surgery: a meta-analysis of randomized controlled trials. Clin Nutr. 2010;29(4):434–40. https://doi.org/10.1016/j.clnu.2010.01.004
Gotlib Conn L, McKenzie M, Pearsall EA, McLeod RS. Successful implementation of an enhanced recovery after surgery programme for elective colorectal surgery: a process evaluation of champions’ experiences. Implement Sci. 2015;10(1):1–11. https://doi.org/10.1186/s13012-015-0289-y
Hughes MJ, Ventham NT, McNally S, Harrison E, Wigmore S. Analgesia after open abdominal surgery in the setting of enhanced recovery surgery: a systematic review and meta-analysis. JAMA Surg. 2014;149(12):1224–30. https://doi.org/10.1001/jamasurg.2014.210
Kagedan DJ, Ahmed M, Devitt KS, Wei AC. Enhanced recovery after pancreatic surgery: A systematic review of the evidence. Hpb. 2015;17(1):11–6. https://doi.org/10.1111/hpb.12265
Melloul E, Hübner M, Scott M, et al. Guidelines for perioperative care for liver surgery: enhanced recovery after surgery (ERAS) society recommendations. World J Surg. 2016;40(10):2425–40. https://doi.org/10.1007/s00268-016-3700-1
Visioni A, Shah R, Gabriel E, Attwood K, Kukar M, Nurkin S. Enhanced recovery after surgery for noncolorectal surgery? Ann Surg. 2018;267(1):57–65. https://doi.org/10.1097/sla.0000000000002267
Azhar RA, Bochner B, Catto J, et al. Enhanced recovery after urological surgery: a contemporary systematic review of outcomes, key elements, and research needs. Eur Urol. 2016;70(1):176–87. https://doi.org/10.1016/j.eururo.2016.02.051
Nelson G, Altman AD, Nick A, et al. Guidelines for postoperative care in gynecologic/oncology surgery: enhanced recovery after surgery (ERAS®) Society recommendations—Part II. Gynecol Oncol. 2016;140(2):323–32. https://doi.org/10.1016/j.ygyno.2015.12.019
Jørgensen CC, Madsbad S, Kehlet H. Postoperative morbidity and mortality in type-2 diabetics after fast-track primary total hip and knee arthroplasty. Anesth Analg. 2015;120(1):230–8. https://doi.org/10.1213/ane.0000000000000451
Stowers MDJ, Manuopangai L, Hill AG, Gray JR, Coleman B, Munro JT. Enhanced recovery after surgery in elective hip and knee arthroplasty reduces length of hospital stay. ANZ J Surg. 2016;86(6):475–9. https://doi.org/10.1111/ans.13538
Batdorf NJ, Lemaine V, Lovely JK, et al. Enhanced recovery after surgery in microvascular breast reconstruction. J Plast Reconstr Aesthetic Surg. 2015;68(3):395–402. https://doi.org/10.1016/j.bjps.2014.11.014
Sadhasivam S, Saxena A, Kathirvel S, Kannan TR, Trikha A, Mohan V. The safety and efficacy of prophylactic ondansetron in patients undergoing modified radical mastectomy. Anesth Analg. 1999;89(6):1340.
Hammas B, Thörn SE, Wattwil M. Superior prolonged antiemetic prophylaxis with a four-drug multimodal regimen: comparison with propofol or placebo. Acta Anaesthesiol Scand. 2002;46(3):232–7. https://doi.org/10.1034/j.1399-6576.2002.460302.x
Fernandez-Guisasola J, Gómez-Arnau JI, Cabrera Y, Del Valle SG. Association between nitrous oxide and the incidence of postoperative nausea and vomiting in adults: A systematic review and meta-analysis: review article. Anaesthesia. 2010;65(4):379–87. https://doi.org/10.1111/j.1365-2044.2010.06249.x
Chiu C, Aleshi P, Esserman LJ, et al. Improved analgesia and reduced post-operative nausea and vomiting after implementation of an enhanced recovery after surgery (ERAS) pathway for total mastectomy. BMC Anesthesiol. 2018. https://doi.org/10.1186/s12871-018-0505-9
Blanco R. The “pecs block”: a novel technique for providing analgesia after breast surgery. Anaesthesia. 2011;66(9):847–8. https://doi.org/10.1111/j.1365-2044.2011.06846.x
Blanco R, Fajardo M, Parras Maldonado T. Ultrasound description of Pecs II (modified Pecs I): a novel approach to breast surgery. Rev Esp Anestesiol Reanim. 2012;59(9):470–5. https://doi.org/10.1016/j.redar.2012.07.003
Blanco R, Parras T, McDonnell JG, Prats-Galino A. Serratus plane block: a novel ultrasound-guided thoracic wall nerve block. Anaesthesia. 2013;68(11):1107–13. https://doi.org/10.1111/anae.12344
Tang J, Wang B, White PF, Watcha MF, Qi J, Wender RH. The effect of timing of ondansetron administration on its efficacy, cost-effectiveness, and cost-benefit as a prophylactic antiemetic in the ambulatory setting. Anesth Analg. 1998;86(2):274–82.
Eberhart L, Mauch M, Morin A, Wulf H, Geldner G. Impact of a multimodal anti-emetic prophylaxis on patient satisfaction in high-risk patients for postoperative nausea and vomiting. Anaesthesia. 2002;57(10):1022–7.
Watcha MF, Smith I. Cost-effectiveness analysis of antiemetic therapy for ambulatory surgery. J Clin Anesth. 1994;6(5):370–7.
Lee JS, Hu HM, Edelman AL, et al. New persistent opioid use Among patients with cancer after curative-intent surgery. J Clin Oncol. 2017;35(36):4042–9. https://doi.org/10.1200/jco.2017.74.1363
Park KU, Kyrish K, Terrell J, et al. Surgeon perception versus reality: opioid use after breast cancer surgery. J Surg Oncol. 2019;119(7):909–15. https://doi.org/10.1002/jso.25395
Rojas KE, Manasseh DM, Flom PL, et al. A pilot study of a breast surgery enhanced recovery after surgery (ERAS) protocol to eliminate narcotic prescription at discharge. Breast Cancer Res Treat. 2018;171(3):621–6. https://doi.org/10.1007/s10549-018-4859-y
Wahab TA, Uwakwe H, Jumah M, Aransi R, Khan HK. A modified enhanced recovery after surgery (ERAS): use and surgical outcome in breast cancer patients. J Biosci Med. 2018;6:15–25. https://doi.org/10.4236/jbm.2018.63002
Acknowledgment
We appreciate the efforts of Adrian Tudose with data collection.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
There are no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dinh, K.H., McAuliffe, P.F., Boisen, M. et al. Post-operative Nausea and Analgesia Following Total Mastectomy is Improved After Implementation of an Enhanced Recovery Protocol. Ann Surg Oncol 27, 4828–4834 (2020). https://doi.org/10.1245/s10434-020-08880-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-020-08880-1