Abstract
Background
Following surgery, a significant proportion of patients develop postoperative complications that are associated with poorer long-term survival. Stereotypical markers of the systemic inflammatory response (SIR) have been shown to identify patients at increased risk of developing such complications. The aim of the present study was to examine the prognostic value of a postoperative systemic inflammation-based score in patients undergoing potentially curative surgery for colorectal cancer.
Methods
Patients with histologically proven colorectal cancer undergoing resection between 1999 and 2013 (n = 813) were grouped into two cohorts—a retrospective test cohort (n = 402) and a prospective validation cohort (n = 411). Patients were assessed for postoperative complications and had routine blood samples taken daily. The relationship between markers of the postoperative SIR and survival was examined using Cox regression analysis.
Results
In the test cohort, 87 patients developed an infective complication, while in the validation cohort, 106 patients developed an infective complication. In both cohorts, the postoperative SIR (C-reactive protein and albumin thresholds of >150 mg/L and <25 g/L, respectively) were associated with the development of infective complications (all p < 0.01). Using these thresholds, a scoring system [postoperative Glasgow prognostic score (poGPS)] was created, and on days 3 and 4 was associated with an incremental increase in the infective complication rate (all p < 0.001) and complication severity (p < 0.001). In the overall cohort, there were 175 cancer and 139 non-cancer deaths. The poGPS was also significantly associated with overall survival (p < 0.05).
Conclusions
The postoperative SIR, evidenced by the poGPS, was associated with increased complication rates and severity and a reduction in survival.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Colorectal cancer is the fourth most common cancer in the UK and the second most common cause of cancer death.1 Despite death rates from colorectal cancer falling by approximately 14% over the last decade, approximately 40% of those diagnosed with colorectal cancer will die from their disease.2
In those deemed to have non-metastatic disease, surgery is the primary modality of cure; however, there has been a long-standing concern that although surgery provides the means of a potential cure for patients with colorectal cancer, the injury associated with surgery may stimulate tumor growth and dissemination.3–7 Despite various infection control measures and the use of preoperative antibiotic prophylaxis following surgery, a significant proportion of patients develop postoperative complications, with the majority of these being infective in nature. Recently, it has become apparent that these complications, as well as having an adverse effect on patients quality of life,8 are associated with both increased risk of cancer recurrence and poorer long-term survival.6,9–12
Therefore, there has been considerable interest in objectively identifying, early in the postoperative phase, which patients are at increased risk of developing infective complications in order to facilitate prompt investigation, treatment or, alternatively, safe discharge. In particular, the stereotypical marker of the systemic inflammatory response (SIR), C-reactive protein (CRP) has been extensively examined and concentrations greater than approximately 150 mg/L on days 3–5 have been shown to be useful.13 In particular, it has been proposed that, in patients undergoing resection for colorectal cancer, CRP concentrations of ≤150 mg/L on postoperative days 3–5 are unlikely to develop infective complications, facilitating safe early discharge.14
McSorley et al. recently reported, in 377 patients, that the postoperative SIR, evidenced by CRP concentrations >150 mg/L, were associated with both complication severity and long-term outcome;15 however, whether this observation provides the basis for a postoperative scoring system to predict both short- and long-term outcomes is not clear.
Therefore, the aim of the present study was to examine whether the combination of postoperative markers of the SIR, namely CRP and albumin, are useful in predicting the development of postoperative infective complications and long-term survival in a large cohort of patients undergoing potentially curative surgery for colorectal cancer.
Patients and Methods
Patients with histologically proven colorectal cancer who, on the basis of intraoperative findings and preoperative computed tomography, were considered to have undergone potentially curative resection at a single center between March 1999 and May 2013 were included in the analyses (n = 813). All patient data were anonymized and all tumors were staged according to conventional tumor, node, metastasis (TNM5) classification, as per the Royal College of Pathologists guidelines16 and additional pathological data obtained from the pathology reports issued at the time of the resection. Patients were grouped into two cohorts and, in both cohorts, patient characteristics were collected in a prospective manner. In the test cohort (surgery from March 1999 to November 2007; n = 402) postoperative complication data were collected retrospectively from electronic records. In the validation cohort (surgery from January 2008 to May 2013; n = 411) postoperative complication data were prospectively collected from patient records following discharge. Due to the prospective method of data collection, the Clavien–Dindo classification of complications was also recorded for this validation cohort.
Preoperatively, all patients received thromboembolism prophylaxis and antibiotic prophylaxis as per local protocols. Blood samples were taken for routine laboratory analysis pre and postoperatively. The preoperative SIR was assessed using the modified Glasgow prognostic score17 (mGPS; electronic supplementary Table 1).
The postoperative SIR was assessed using the postoperative Glasgow prognostic score (poGPS; electronic supplementary Table 1). In essence, a postoperative CRP concentration below 150 mg/L, regardless of albumin concentration, scored 0, a CRP concentration ≥ 150 mg/L and albumin >25 g/L scored 1, and CRP ≥ 150 mg/L and albumin <25 g/L scored 2. The creation of this score was initially performed in the retrospective test cohort, and an attempt to subsequently validate this in the prospective validation cohort was then performed.
Postoperatively, all patients underwent daily clinical assessment. Clinicians were not blinded to these daily blood results and additional investigations and management were instigated at the surgical team’s discretion based on the relevant clinical findings.
Patients were assessed for both non-infective (ileus, acute coronary syndrome, acute myocardial infarction, pulmonary embolism and arrhythmias) and infective complications (wound, intra-abdominal abscess, anastomotic leak, urinary tract infection, and pneumonia). The criteria used to define these complications were the same as has been previously described18,19. In short, a wound infection included the presence of pus that discharged spontaneously or required drainage; an intra-abdominal abscess was confirmed by imaging and required either conservative therapy with antibiotics or drainage; an anastomotic leak was defined as a fistula to the bowel anastomosis that was confirmed radiologically or diagnosed at relaparotomy; and pneumonia was diagnosed as the presence of X-ray changes and fever that required antibiotic therapy and urinary tract infection as positive urine culture in the presence of symptoms that required antibiotic therapy. Patients were routinely followed up for 5 years following resection, as per national guidelines. The date and cause of death were cross-checked with the cancer registration system and the Registrar General (Scotland). The West of Scotland Research Ethics Committee approved this study.
Statistical Analysis
The comparison of categorical and continuous variables was performed using the Chi square test and Mann–Whitney U test, respectively. Univariate survival analysis was performed using Cox proportional hazards regression in order to calculate hazard ratios (HR) and 95% confidence intervals (95% CI). A two-sided p value of <0.05 was considered statistically significant. Statistical analysis was performed using SPSS version 22.0 for Windows (IBM Corporation, Armonk, NY, USA).
Results
In both the test (n = 402) and validation cohorts (n = 411), the majority of patients were aged >65 years, were male, had a colonic tumor, and had an elective operation. Comparison of the clinicopathological characteristics in both cohorts is shown in Table 1. There were significantly fewer emergency procedures (p < 0.05), more T1 tumors and less T3 tumors (p < 0.005), a greater overall complication rate (p < 0.001), and a greater infective complication rate (p < 0.001) in the validation cohort. Median follow-up of survivors in the test cohort was 116 months (range 76–180), and 31 months (range 10–71) in the validation cohort.
Test Cohort
With regard to short-term outcomes, 87 patients (22%) developed an infective complication. Of the preoperative factors in this cohort, emergency presentation (p < 0.01) and raised preoperative CRP concentrations (p < 0.05) were associated with the development of infective complications. Moreover, rectal surgery (p < 0.01), exceeding the postoperative thresholds for CRP on days 3 and 4 (both p < 0.001) and albumin on days 3 and 4 (both p < 0.001), and the days 3 and 4 poGPS (both p < 0.001) were also associated with the development of infective complications (Table 2).
In patients undergoing surgery for colon cancer (n = 259), 46 (18%) developed an infective complication. In these patients, emergency presentation (p < 0.001), exceeding the postoperative days 3 and 4 thresholds of CRP (p < 0.001) and albumin (p < 0.005), and the days 3 and 4 poGPS (both p < 0.001) were associated with the development of infective complications (electronic supplementary Table 2).
When using the postoperative scoring system (poGPS) (Table 4), an incremental increase in the day 3 poGPS from 0 to 1 to 2 resulted in an increase in the infective complication rate from 10.3 to 31.0 to 56.6% (p < 0.001), and, using the day 4 poGPS, from 17.0 to 50.0 to 73.9% (p < 0.001).
Validation Cohort
In this cohort, findings similar to those in the test cohort were observed. With regard to short-term outcomes, 106 patients (26%) developed an infective complication. Male sex (p < 0.005), exceeding the postoperative thresholds for CRP on days 3 and 4 (both p < 0.001) and albumin on days 3 and 4 (both p < 0.005), and the days 3 and 4 poGPS (both p < 0.001) were also associated with the development of infective complications (Table 3).
When using the postoperative scoring system (Table 4), an incremental increase in the day 3 poGPS resulted in an increase in the infective complication rate from 14.4 to 28.9 to 41.9% (p < 0.001), and, using the day 4 poGPS, from 17.9 to 32.2 to 53.7% (p < 0.001). An increase in the day 3 poGPS score resulted in an increase in severity of Clavien–Dindo Scores (p < 0.001), as did an increase in the day 4 poGPS (p < 0.001).
Overall Cohort (n = 813)
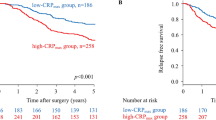
With regard to long-term outcomes, univariate survival analysis between the clinicopathological characteristics, the pre- and postoperative SIR, is shown in Table 5. There were 175 cancer deaths and 139 non-cancer deaths. Age (p < 0.001), TNM stage (p < 0.001), venous invasion (p < 0.005), margin involvement (p < 0.001), peritoneal involvement (p < 0.001), adjuvant therapy (p < 0.05), and mGPS (p < 0.001), as well as exceeding the postoperative days 3 and 4 threshold for CRP (both p < 0.05) and albumin (both p < 0.05), and the days 3 and 4 poGPS (p < 0.05), were associated with OS.
Furthermore, in patients undergoing resection of colon cancer (electronic supplementary Table 3), age (p < 0.01), emergency presentation (p < 0.05), TNM stage (p < 0.001), venous invasion (p < 0.05), margin involvement (p < 0.001), and adjuvant therapy (p < 0.05), as well as the mGPS (p < 0.001), exceeding the days 3 and 4 threshold for CRP (p < 0.05) and albumin (p < 0.001), and the days 3 and 4 poGPS (p < 0.005) were associated with overall survival.
Discussion
The results of the present study confirm that the development of postoperative complications, particularly infective complications, was associated with the postoperative SIR in patients undergoing surgery for colorectal cancer. Furthermore, an objective postoperative scoring approach (poGPS) was capable of stratifying the risk of developing postoperative infective complications, ranging from approximately 12% with a score of 0 to approximately 30% with a score of 1, and approximately 50% with a score of 2. These results have been demonstrated in a retrospective ‘test cohort’ and subsequently validated in the prospective ‘validation’ cohort. Moreover, the postoperative SIR, evidenced by the poGPS, was associated with survival in patients undergoing surgery for colorectal cancer.
This study appears to finally tie the SIR to outcomes following surgery for colorectal cancer. Specifically, the present study builds on work by McSorley et al. who, in a cohort of 377 patients, reported that the magnitude of the postoperative SIR was significantly associated with long-term outcomes, independent of complications and tumor stage15. These findings, along with the present study, would appear to suggest that the mechanisms behind the development of postoperative complications and poor long-term survival are linked by the SIR. Therefore, a plausible hypothesis is that the cancer itself elicits an SIR in a significant proportion of patients, while the added surgical injury produces an immunological hit in all patients that leads to homeostatic decompensation in some. The nature and consequences of that decompensation may be the same as seen in patients with untreated or metastatic disease, perhaps even stimulating the growth of residual cancer cells (micrometastatic disease). Moreover, it would appear that the SIR involves both a dimension of magnitude and duration, both of which affect the prognosis. Consistent with this hypothesis, it has recently been reported that serum and peritoneal fluid samples from patients undergoing colorectal cancer surgery who had postoperative peritoneal infection increased the in vitro invasiveness capacity of cancer cell lines, causing increased tumor dissemination and tumor cell survival20. If this were to prove to be the case, then it may be that colorectal cancer surgery should be practiced in such a way to minimize the postoperative SIR. This study lays the foundation for further work in this field.
In the present study, it was of interest that elevated poGPS scores were significantly associated with emergency presentation and the presence of a SIR preoperatively. It has now been established that patients who present as an emergency for surgery for colorectal cancer have poorer 5 year survival21–23. In the present study, when only patients who had elective surgery and an mGPS of 0 were examined, the poGPS stratified the postoperative infective complication rate on both day 3 (poGPS0 rate was 13.7%, poGPS1 rate was 32.8%, and poGPS2 rate was 50.7%; p < 0.001) and day 4 (poGPS0 rate was 17.3, poGPS1 rate was 43.1%, and poGPS2 rate was 71.4%; p < 0.001). In addition, the poGPS stratified the 5 year survival rates on day 3 (poGPS0 rate was 74%, poGPS1 rate was 67%, and poGPS2 rate was 60%; p = 0.039). Therefore, the results of the present study would indicate a role for a postoperative SIR scoring system in predicting both short- and long-term outcomes in patients undergoing surgery for colorectal cancer.
It was recently reported that the depletion of skeletal muscle mass following surgery for colorectal cancer was greater with older age, female sex, open surgery, and an elevated preoperative SIR, as evidenced by the neutrophil lymphocyte ratio24. Therefore, consistent with the present results, it may be hypothesized that this was related to a greater postoperative SIR. Given the above, it would be of interest to examine whether approaches to minimize the poGPS, other than laparoscopic surgery, such as perioperative steroids25, would reduce the loss of skeletal muscle mass following surgery for colorectal cancer.
This study has several potential clinical benefits. In those with a low score, it may provide the clinician with reassurance regarding the development of infective complications, and allow prompt discharge, particularly in an enhanced recovery setting. In contrast, in those with a high score, it may provide an early warning to the clinician and prompt reassessment and management of the patient. In addition, by enabling objective comparison of the impact of different surgical approaches and techniques on the magnitude of the postoperative SIR following surgery, it may be possible to identify individuals or techniques that minimize the poGPS score26. Finally, by acting as a therapeutic target, the use of postoperative anti-inflammatory agents has the potential to improve short-term outcomes. The use of anti-inflammatory agents in the postoperative period, particularly following colorectal surgery, continues to be a subject of intense debate, with studies reporting conflicting outcomes27–29. Cautious use in the postoperative period may provide benefit to patients with an exaggerated postoperative SIR, but more work is required to test this.
The magnitude of surgical injury in different colorectal procedures may be different, e.g. for colonic resections and rectal resections. This may also vary across surgical centers, with differences in patient cohorts and operative expertise. Therefore, the present results require external validation. However, given the simplicity of the measurement of postoperative SIR developed, such validation can be readily tested.
In the present study, thresholds for CRP and albumin were examined using receiver operating curve analysis and postoperative infective complications as an endpoint. On day 3, these were 153 mg/L and 26 g/L, respectively, and 125 mg/L and 27 g/L, respectively, on day 4. These were similar to those established from previous meta-analysis (CRP > 150 mg/L and albumin <25 g/L) and therefore the latter thresholds were used in the analysis. A limitation of the present study was that there are intrinsic and extrinsic factors not accounted for that may potentially affect the relationship between the postoperative SIR and long- and short-term outcomes in patients undergoing surgery for colorectal cancer. For example, comorbidities, the quality and type of anesthesia/surgery, blood loss, and blood transfusion may all affect this relationship. Nevertheless, the poGPS provides an objective framework against which such factors are to be investigated.
Conclusions
The magnitude of the postoperative SIR, as evidenced by the poGPS, was associated with an incremental increase in the postoperative infective complication rates and a reduction in survival. Elevated systemic inflammation, whether prior to or following surgery, is associated with poor outcome in patients with colorectal cancer.
References
Cancer Research UK. Key facts. 2014. http://www.cancerresearchuk.org/cancer-info/cancerstats/keyfacts/bowel-cancer/.
Oliphant R, Nicholson GA, Horgan PG, Molloy RG, McMillan DC, Morrison DS. Contribution of surgical specialization to improved colorectal cancer survival. Br J Surg. 2013;100(10):1388–95.
Abramovitch R, Marikovsky M, Meir G, Neeman M. Stimulation of tumour growth by wound-derived growth factors. Br J Cancer. 1999;79(9-10):1392–8.
Mynster T, Christensen IJ, Moesgaard F, Nielsen HJ. Effects of the combination of blood transfusion and postoperative infectious complications on prognosis after surgery for colorectal cancer. Danish RANX05 Colorectal Cancer Study Group. Br J Surg. 2000;87(11):1553–62.
Baum M, Demicheli R, Hrushesky W, Retsky M. Does surgery unfavourably perturb the “natural history” of early breast cancer by accelerating the appearance of distant metastases? Eur J Cancer. 2005;41(4):508–15.
McArdle CS, McMillan DC, Hole DJ. Impact of anastomotic leakage on long-term survival of patients undergoing curative resection for colorectal cancer. Br J Surg. 2005;92(9):1150–4.
Retsky MW, Demicheli R, Hrushesky WJ, Baum M, Gukas ID. Dormancy and surgery-driven escape from dormancy help explain some clinical features of breast cancer. APMIS. 2008;116(7-8):730–41.
Brown SR, Mathew R, Keding A, Marshall HC, Brown JM, Jayne DG. The impact of postoperative complications on long-term quality of life after curative colorectal cancer surgery. Ann Surg. 2014;259(5):916–23.
Mirnezami A, Mirnezami R, Chandrakumaran K, Sasapu K, Sagar P, Finan P. Increased local recurrence and reduced survival from colorectal cancer following anastomotic leak: systematic review and meta-analysis. Ann Surg. 2011;253(5):890–9.
Richards CH, Platt JJ, Anderson JH, McKee RF, Horgan PG, McMillan DC. The impact of perioperative risk, tumor pathology and surgical complications on disease recurrence following potentially curative resection of colorectal cancer. Ann Surg. 2011;254(1):83–9.
Pucher PH, Aggarwal R, Qurashi M, Darzi A. Meta-analysis of the effect of postoperative in-hospital morbidity on long-term patient survival. Br J Surg. 2014;101(12):1499–508.
Artinyan A, Orcutt ST, Anaya DA, Richardson P, Chen GJ, Berger DH. Infectious postoperative complications decrease long-term survival in patients undergoing curative surgery for colorectal cancer: a study of 12,075 patients. Ann Surg. 2015;261(3):497–505.
Singh PP, Zeng IS, Srinivasa S, Lemanu DP, Connolly AB, Hill AG. Systematic review and meta-analysis of use of serum C-reactive protein levels to predict anastomotic leak after colorectal surgery. Br J Surg. 2014;101(4):339–46.
McDermott FD, Heeney A, Kelly ME, Steele RJ, Carlson GL, Winter DC. Systematic review of preoperative, intraoperative and postoperative risk factors for colorectal anastomotic leaks. Br J Surg. 2015;102(5):462–79.
McSorley ST, Watt DG, Horgan PG, McMillan DC. Postoperative systemic inflammatory response, complication severity, and survival following surgery for colorectal cancer. Ann Surg Oncol. 2016;23(9):2832–40.
The Royal College of Pathologists. Dataset for colorectal cancer histopathology reports (3rd edition). 2014. https://www.rcpath.org/resourceLibrary/dataset-for-colorectal-cancer-histopathology-reports–3rd-edition-.html.
McMillan DC. The systemic inflammation-based Glasgow prognostic score: a decade of experience in patients with cancer. Cancer Treat Rev. 2013;39(5):534–40.
Platt JJ, Ramanathan ML, Crosbie RA, et al. C-reactive protein as a predictor of postoperative infective complications after curative resection in patients with colorectal cancer. Ann Surg Oncol. 2012;19(13):4168–77.
Ramanathan ML, Mackay G, Platt J, Horgan PG, McMillan DC. Impact of day 2 C-reactive protein on day 3 and 4 thresholds associated with infective complications following curative surgery for colorectal cancer. World J Surg. 2013;37(11):2705–10.
Salvans S, Mayol X, Alonso S, et al. Postoperative peritoneal infection enhances migration and invasion capacities of tumor cells in vitro: an insight into the association between anastomotic leak and recurrence after surgery for colorectal cancer. Ann Surg. 2014;260(5):939–44.
McArdle CS, Hole DJ. Emergency presentation of colorectal cancer is associated with poor 5-year survival. Br J Surg. 2004;91(5):605–9.
Oliphant R, Horgan PG, Morrison DS, McMillan DC, West of Scotland Colorectal Cancer Managed Clinical Network. Validation of a modified clinical risk score to predict cancer-specific survival for stage II colon cancer. Cancer Med. 2015;4(1):84–9.
Oliphant R, Mansouri D, Nicholson GA, et al. Emergency presentation of node-negative colorectal cancer treated with curative surgery is associated with poorer short and longer-term survival. Int J Colorectal Dis. 2014;29(5):591–8.
Malietzis G, Currie AC, Johns N, et al. Skeletal muscle changes after elective colorectal cancer resection: a longitudinal study. Ann Surg Oncol. 2016;23(8):2539–47.
McSorley ST, Horgan PG, McMillan DC. The impact of preoperative corticosteroids on the systemic inflammatory response and postoperative complications following surgery for gastrointestinal cancer: a systematic review and meta-analysis. Crit Rev Oncol Hematol. 2016;101:139–50.
Watt DG, Horgan PG, McMillan DC. Routine clinical markers of the magnitude of the systemic inflammatory response after elective operation: a systematic review. Surgery. 2015;157(2):362–80.
Klein M, Gogenur I, Rosenberg J. Postoperative use of non-steroidal anti-inflammatory drugs in patients with anastomotic leakage requiring reoperation after colorectal resection: cohort study based on prospective data. BMJ. 2012;345:e6166.
Bhangu A, Singh P, Fitzgerald JE, Slesser A, Tekkis P. Postoperative nonsteroidal anti-inflammatory drugs and risk of anastomotic leak: meta-analysis of clinical and experimental studies. World J Surg. 2014;38(9):2247–57.
STARSurg Collaborative. Impact of postoperative non-steroidal anti-inflammatory drugs on adverse events after gastrointestinal surgery. Br J Surg. 2014;101(11):1413–23.
Funding
No sources of financial support were obtained for the writing of this paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
David G. Watt, Stephen T. McSorley, James H. Park, Paul G. Horgan, and Donald C. McMillan declare no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Watt, D.G., McSorley, S.T., Park, J.H. et al. A Postoperative Systemic Inflammation Score Predicts Short- and Long-Term Outcomes in Patients Undergoing Surgery for Colorectal Cancer. Ann Surg Oncol 24, 1100–1109 (2017). https://doi.org/10.1245/s10434-016-5659-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-016-5659-4