Abstract
Background
This study aimed to evaluate the impact of preoperative biliary drainage (PBD) on the long-term survival of patients with pancreatic ductal adenocarcinoma (PDAC) who underwent pancreaticoduodenectomy (PD).
Methods
A multicenter observational study was performed using a common database of patients with resected PDAC from seven high-volume surgical institutions in Japan.
Results
Of 932 patients who underwent PD for PDAC, 573 (62 %) underwent PBD, including 407 (44 %) who underwent endoscopic biliary drainage (EBD) and 166 (18 %) who underwent percutaneous transhepatic biliary drainage (PTBD). The patients who did not undergo PBD and those who underwent EBD had a significantly better overall survival than those who underwent PTBD, with median survival times of 25.7 months (P < 0.001), 22.3 months (P = 0.001), and 16.7 months, respectively. Multivariate analysis showed that seven clinicopathologic factors, including the use of PTBD but not EBD, were independently associated with poorer overall survival. Furthermore, patients who underwent PTBD more frequently experienced peritoneal recurrence (23 %) than those who underwent EBD (10 %; P < 0.001) and those who did not undergo PBD (11 %; P = 0.001). Multivariate analysis demonstrated that the independent risk factors for peritoneal recurrence included surgical margin status (P < 0.001) and use of PTBD (P = 0.004).
Conclusions
Use of PTBD, but not EBD, was associated with a poorer prognosis, with an increased rate of peritoneal recurrence among patients who underwent PD for PDAC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Preoperative biliary drainage (PBD) for pancreatic ductal adenocarcinoma (PDAC) may be clinically relevant for subsets of patients, such as those with acute cholangitis, those with severe liver dysfunction due to jaundice, and those scheduled for neoadjuvant therapy.1 Currently, accumulating data from recent multicenter randomized trials and systematic reviews demonstrate that PBD for distal biliary obstruction results in increased perioperative complications after pancreaticoduodenectomy (PD).2–4
However, a recent survey from the United States suggests that PBD has been increasing, and more than half of the patients in this survey who underwent PD for pancreatic cancer had undergone PBD despite consistent recommendations against its routine use.5 In addition, a percutaneous transhepatic approach has been used at a constant rate of about 15 % during the past two decades.5
Currently, little is known about the influence of PBD on the long-term survival of patients with PDAC who have undergone PD. A few investigators have reported that the use of PBD had no unfavorable impact on the long-term survival of patients with PDAC who underwent PD.6,7
A recent report demonstrated that patients who had undergone percutaneous transhepatic biliary drainage (PTBD) for hilar cholangiocarcinoma more frequently experienced peritoneal recurrence than those who underwent endoscopic biliary drainage (EBD) and that use of PTBD was the only independent factor predictive of peritoneal recurrence.8
On the basis of these findings, we hypothesized that PTBD for PDAC might affect long-term survival after surgical resection. We investigated the prognostic impact of PBD, including type, on the long-term survival of patients with PDAC who underwent PD in a large-scale, multicenter observational study. We also evaluated the influence of PBD on the peritoneal recurrence of PDAC after PD.
Methods
Study Design
This multicenter observational study investigated patients with resected PDAC who underwent PD. They were identified from a common database of patients registered by seven high-volume surgical institutions in Japan who had undergone resection for PDAC between 2001 and 2012. This study was performed after approval from the ethics review board of Hiroshima University Hospital, followed by approval from each institution.
Patient Selection
All patients who underwent R0 or R1 tumor resection and had a confirmed pathologic diagnosis of PDAC were collected into a database. Cases with advanced distal cholangiocarcinoma were excluded by careful assessment of the macro- and microscopic location of the lesion. Patients with invasive carcinoma associated with intraductal papillary mucinous neoplasms were not included. Patients with pancreatic anaplastic carcinoma and initially unresectable PDAC who had tumor resection after chemotherapy or chemoradiotherapy were excluded. Patients who underwent distal pancreatectomy, total pancreatectomy, and surgical biliary drainage before PD also were excluded.
Preoperative Biliary Drainage
Patients with PDAC who had distal biliary obstruction during the study period routinely underwent PBD. The PBD was performed at endoscopic retrograde cholangiopancreatography (ERCP) by placement of a plastic stent, endoscopic nasobiliary stent, or metallic stent according to institutional policy. In case of a need for additional drainage or a failed ERCP attempt, PTBD was used to achieve biliary drainage.
Main Outcome Measures
Of the patients who underwent PD for PDAC, those who underwent PBD were identified and stratified into two groups according to the type of drainage they received: the group that had PTBD and the group that had EBD.
The primary objective of this study was to evaluate the impact of PBD on the overall survival of patients with PDAC who underwent PD. The secondary objective was to investigate the influence of PBD on peritoneal recurrence of resected PDAC in this cohort. These analyses were performed with three groups of patients: those who underwent PTBD, those who underwent EBD, and those who did not undergo PBD.
Definition
The final stage of PDAC was assessed pathologically according to the tumor-node-metastasis (TNM) classification by the International Union Against Cancer (UICC), 7th edition. Overall survival time was measured from the time of surgery until death or the last follow-up visit. Peritoneal metastasis was diagnosed on computed tomography, and the diagnosis was confirmed by cytologic examination if possible.
Statistical Analysis
Data are expressed as median (range), and the median value was determined to be the cutoff value of continuous variables. Clinicopathologic factors were compared between the groups using the Chi square test or Fisher’s exact test. Survival curves were constructed with the Kaplan–Meier method, and differences in survival curves were compared by univariate log-rank (Mantel–Cox) analysis. Factors found to be significant in the univariate analysis were subjected to multivariate analysis with a Cox proportional hazards model. Variables with a P value lower than 0.05 were entered into a logistic regression model to determine independent risk factors. The independent risk factors were expressed as odds ratios with their 95 % confidence intervals. Differences were considered significant at a P value lower than 0.05. Statistical analysis was performed with JMP statistical software version 5.1 (SAS Institute, Cary, NC, USA).
Results
Patient Demographics
The study included 932 patients with PDAC who underwent PD. Of the 932 patients with PDAC who underwent PD, 573 (62 %) underwent PBD. Of these, 166 patients (18 %) underwent PTBD. The remaining 407 patients (44 %) underwent EBD using one of three stent types: a plastic stent in 302 patients (32 %), a nasobiliary stent in 83 patients (9 %), and a metallic stent in 22 patients (2 %).
Whereas PBD was indicated at a constant rate, from 63 % in 2001–2006 to 61 % in 2007–2012 (P = 0.513), the rate of EBD increased from 31 % in 2001–2006 to 49 % in 2007–2012 (P < 0.001).
Table 1 shows a comparison of patient demographics and clinicopathologic factors. The patients who underwent PTBD, those who underwent EBD, and those who did not undergo PBD differed significantly in terms of age (P = 0.042), operative time (P = 0.026), amount of blood loss (P < 0.001), use of blood transfusion (P < 0.001), rate of lymph node metastasis (P = 0.009), surgical margin status (P = 0.012), UICC pT factor (P < 0.001), and UICC stage (P < 0.001).
Postoperative Complications and Mortality
A significant difference was found in the incidence of wound infection among the patients who underwent PTBD, those who underwent EBD, and those who did not undergo PBD (P = 0.039). No significant differences were found in the incidence of intraabdominal abscess (P = 0.952), clinically relevant postoperative pancreatic fistula (P = 0.498), severe complications (P = 0.117), 30-day mortality (P = 0.169), or in-hospital mortality (P = 0.139) among the patients who underwent PTBD, those who underwent EBD, and those who did not undergo PBD (Table 1).
Overall Survival
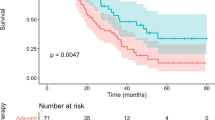
The median follow-up period for all the patients in the current study was 18.2 months, and the median follow-up period was 15.6 months for the patients who underwent PTBD, 17.3 months for the patients who underwent EBD, and 19.7 months for the patients who did not undergo PBD. Figure 1 shows the Kaplan–Meier curves for overall survival among the patients with PDAC stratified by type of PBD. Overall survival differed significantly among the patients who underwent PTBD, those who underwent EBD, and those who did not undergo PBD (P < 0.001). The patients who did not undergo PBD (n = 359) and those who underwent EBD (n = 407) had a significantly better overall survival than those who underwent PTBD (n = 166), with median survival times (MSTs) of 25.7 months (P < 0.001), 22.3 months (P = 0.001), and 16.7 months, respectively.
Kaplan–Meier curves for overall survival (OS) among patients with pancreatic ductal adenocarcinoma (PDAC) stratified by the type of preoperative biliary drainage (PBD). The patients who did not undergo PBD (n = 359) and those who underwent endoscopic biliary drainage (EBD) (n = 407) had a significantly better OS than those who underwent percutaneous transhepatic biliary drainage (PTBD) (n = 166), with respective median survival times (MSTs) of 25.7 months (P < 0.001), 22.3 months (P = 0.001), and 16.7 months. Overall survival did not differ significantly between the patients who did not undergo PBD and those who underwent EBD (P = 0.123)
The overall 5-year survival rate was 9.8 % for the patients who underwent PTBD, 23.1 % for the patients who underwent EBD, and 26.1 % for the patients who did not undergo PBD. Overall survival did not differ significantly between the patients who did not undergo PBD and those who underwent EBD (P = 0.123).
Table 2 shows the uni- and multivariate survival analyses of the prognostic factors for patients with PDAC who underwent PD. The univariate analysis showed that older age (P = 0.022), type of PBD (P < 0.001), type of PD (P = 0.013), longer operative time (P = 0.001), increased blood loss (P < 0.001), use of blood transfusion (P < 0.001), no completion of adjuvant chemotherapy (P < 0.001), positive lymph node metastasis (P < 0.001), positive surgical margin status (P < 0.001), advanced UICC pT factor (P < 0.001), advanced UICC stage (P < 0.001), and presence of severe complications (P = 0.001) were significantly associated with worse overall survival.
The aforementioned factors, except for UICC stage, were entered into a multivariate analysis with a Cox proportional hazard model. The multivariate analysis showed that increased blood loss (P = 0.005), use of blood transfusion (P < 0.001), positive lymph node metastasis (P < 0.001), advanced UICC pT factor (P = 0.034), positive surgical margin status (P = 0.029), no completion of adjuvant chemotherapy (P < 0.001), and the use of PTBD (P = 0.029) remained independently associated with worse overall survival.
Risk Factors for Peritoneal Recurrence
Figure 2 compares the incidence of peritoneal metastasis as a primary site of recurrence in patients with PDAC who underwent PD by the type of PBD. The incidence of peritoneal recurrence differed significantly among the patients who underwent PTBD, those who underwent EBD, and those who did not undergo PBD (P < 0.001).
Incidence of peritoneal metastasis (PR) by type of preoperative biliary drainage (PBD) among patients with pancreatic ductal adenocarcinoma (PDAC). The patients who underwent percutaneous transhepatic biliary drainage (PTBD) (22 %) more often experienced PR than those who underwent endoscopic biliary drainage (EBD) (10 %; P < 0.001) and those who did not undergo PBD (11 %; P = 0.001). The incidence of PR did not differ significantly between the patients who did not undergo PBD (11 %; P = 0.001) and those who underwent EBD (P = 0.657)
The incidence of peritoneal recurrence among the patients who underwent EBD (10 %; P < 0.001) and who did not undergo PBD (11 %; P = 0.001) was significantly lower than among the patients who underwent PTBD (23 %). The incidence of peritoneal recurrence did not differ significantly between the patients who underwent EBD and those who did not undergo PBD (P = 0.657).
Table 3 shows the results of the uni- and multivariate analyses of risk factors for peritoneal metastasis as a primary site of recurrence in patients with PDAC who underwent PD. The univariate analysis showed that type of PBD (P < 0.001), increased blood loss (P = 0.020), use of blood transfusion (P = 0.005), positive lymph node metastasis (P = 0.034), and positive surgical margin status (P < 0.001) were significantly associated with peritoneal metastasis as a primary site of recurrence. These factors were entered into a multivariate analysis with a logistic regression model. The multivariate analysis showed that positive surgical margin status (P < 0.001) and use of PTBD (P = 0.004), but not EBD (P = 0.355), remained independently associated with peritoneal metastasis as a primary site of recurrence.
Discussion
Various prior reports have evaluated prognostic factors for patients who underwent PD for PDAC. These factors include blood loss,9 blood transfusion,10 tumor size,11 lymph node metastasis,12–14 lymph node ratio,15,16 surgical margin status,17 vascular invasion,18 adjuvant chemotherapy,19 and host inflammatory response.20,21
In the current study, a multivariate analysis of unfavorable prognostic factors showed that blood loss, blood transfusion, lymph node metastasis, advanced UICC pT factor, positive surgical margin status, no completion of adjuvant chemotherapy, and use of PTBD were independently associated with overall survival. Most factors, except for use of PTBD, are in line with the previous reports.
Currently, there are few studies regarding the prognostic impact of PBD on overall survival of patients with PDAC. The study of di Mola et al.7 investigated 59 patients who underwent PD for PDAC and demonstrated that the presence of jaundice, PBD, and postoperative complications had no unfavorable impact on overall survival. Smith et al.6 investigated the influence of PBD on 155 patients who underwent PD for PDAC, which included 130 patients who underwent PBD and 25 patients who did not. These authors demonstrated that PBD had no adverse impact on overall survival. However, these studies were single-center investigations with small samples.
One multicenter randomized clinical trial investigated 185 patients with periampullary malignancy, including 77 patients with PDAC, to clarify the impact of PBD on overall survival.1 It demonstrated that PBD followed by surgical resection does not impair the long-term overall survival of patients with obstructive jaundice compared with surgery alone. This trial also showed that PBD does not confer a survival benefit. However, because it is well accepted that survival after PD for periampullary malignancy is closely related to the histopathologic origin and biologic behavior of the underlying disease, the fact that PDAC was present in significantly more patients in the early surgery group (52 %) than in the PBD group (32 %; P = 0.004) might have affected these results.
Very few studies have focused on the influence of the route of PBD on overall survival after PD for PDAC. Only one study6 investigated the survival difference between patients who required EBD (n = 118) and those who required PTBD (n = 12). This study found no significant difference in overall survival between the two groups, although the number of patients who underwent PTBD was too small for a comparative survival analysis.
Despite recommendations to avoid routine PBD, a survey from the United States demonstrated that the use of PBD for patients with pancreatic cancer has been increasing.5 The survey showed that the use of PBD increased from 30 % in 1992–1995 to 60 % in 2004–2007. The increased rate of PBD was reported to be driven by a rise in the use of EBD, whereas PTBD was used at a constant rate of 12–17 % during the past two decades.5 Our data from seven high-volume surgical institutions in Japan during a decade demonstrated that PBD for PDAC was used at a constant rate of about 60 % from 2001 to 2012, whereas the use of EBD increased from 31 % in 2001–2006 to 49 % in 2007–2012. The increase in the use of EBD denotes the same tendency in both studies.
Several prior reports have evaluated the risk factors of peritoneal recurrence for patients who underwent PD for PDAC. Takahashi et al.22 demonstrated that the presence of perineural invasion was the single independent variable significantly associated with an increased risk of peritoneal recurrence. Hata et al.23 showed that the postoperative CA19-9 level was associated with positive surgical margins and hepatic or peritoneal recurrence.
In the current study, the multivariate analysis of risk factors for peritoneal recurrence showed that surgical margin status and use of PTBD remained independently associated with peritoneal recurrence after PD for PDAC.
Concerning PTBD use and peritoneal recurrence, Chapman et al.24 reported in 1989 the finding of pancreatic cancer seeding along the PTBD. They reviewed 18 patients with this type of complication, including 7 patients with pancreatic cancer, and 3 of the 18 patients (all 3 had a cholangiocarcinoma) experienced diffuse peritoneal seeding believed to be secondary to the transperitoneal catheter.
Moreover, in a recent report, patients who underwent PTBD for hilar cholangiocarcinoma more frequently experienced peritoneal recurrence than those who underwent EBD, and use of PTBD was the only independent predictive factor for peritoneal recurrence.8 The investigators concluded that EBD might confer a better prognosis than PTBD due to prevention of peritoneal seeding, and it is recommended as the initial procedure for PBD in patients with hilar cholangiocarcinoma. The poorer prognosis of the patients who underwent PTBD in the current study is more likely associated with a poorer biologic behavior of the primary tumor. However, no previous report has addressed the influence of PBD, including its route, on peritoneal recurrence in patients with resected PDAC.
Several limitations need to be considered when the results of the current study are interpreted. First, this was a retrospective observational study. A prospective validation study to confirm the possible risks of PTBD is necessary. Second, because we have no data concerning jaundice, including the level of serum total bilirubin and liver function before PBD, the influence of jaundice on long-term overall survival could not be evaluated.
Finally, we have no data regarding the rate of failed endoscopic attempts for the patients who underwent PTBD or the preoperative duration of PBD. Therefore, the survival data may have a lead time bias. However, this study was both the largest and the first multicenter study of patients with PDAC to elucidate the prognostic influence of PBD on long-term survival.
In conclusion, the use of PTBD, but not EBD, was associated with a poorer prognosis, with an increased rate of peritoneal recurrence in patients who underwent PD for PDAC.
References
Eshuis WJ, van der Gaag NA, Rauws EA, et al. Therapeutic delay and survival after surgery for cancer of the pancreatic head with or without preoperative biliary drainage. Ann Surg. 2010;252:840–9.
Sewnath ME, Karsten TM, Prins MH, Rauws EJ, Obertop H, Gouma DJ. A meta-analysis on the efficacy of preoperative biliary drainage for tumors causing obstructive jaundice. Ann Surg. 2002;236:17–27.
van der Gaag NA, Rauws EA, van Eijck CH, et al. Preoperative biliary drainage for cancer of the head of the pancreas. N Engl J Med. 2010;362:129–37.
Iacono C, Ruzzenente A, Campagnaro T, Bortolasi L, Valdegamberi A, Guglielmi A. Role of preoperative biliary drainage in jaundiced patients who are candidates for pancreatoduodenectomy or hepatic resection: highlights and drawbacks. Ann Surg. 2013;257:191–204.
Jinkins LJ, Parmar AD, Han Y, et al. Current trends in preoperative biliary stenting in patients with pancreatic cancer. Surgery. 2013;154:179–89.
Smith RA, Dajani K, Dodd S, et al. Preoperative resolution of jaundice following biliary stenting predicts more favourable early survival in resected pancreatic ductal adenocarcinoma. Ann Surg Oncol.2008;15:3138–46.
di Mola FF, Tavano F, Rago RR, et al. Influence of preoperative biliary drainage on surgical outcome after pancreaticoduodenectomy: single-centre experience. Langenbeck’s Arch Surg. 2014;399:649–57.
Hirano S, Tanaka E, Tsuchikawa T, et al. Oncological benefit of preoperative endoscopic biliary drainage in patients with hilar cholangiocarcinoma. J Hepatobiliary Pancreat Sci. 2014;21:533–40.
Nagai S, Fujii T, Kodera Y, et al. Impact of operative blood loss on survival in invasive ductal adenocarcinoma of the pancreas. Pancreas. 2011;40:3–9.
Cameron JL, Crist DW, Sitzmann JV, et al. Factors influencing survival after pancreaticoduodenectomy for pancreatic cancer. Am J Surg. 1991;161:120–4; discussion 124–5.
Mu DQ, Peng SY, Wang GF. Risk factors influencing recurrence following resection of pancreatic head cancer. World J Gastroenterol. 2004;10:906–9.
Schnelldorfer T, Ware AL, Sarr MG, et al. Long-term survival after pancreatoduodenectomy for pancreatic adenocarcinoma: is cure possible? Ann Surg. 2008;247:456–62.
Doi R, Kami K, Ito D, et al. Prognostic implication of para-aortic lymph node metastasis in resectable pancreatic cancer. World J Surg. 2007;31:147–54.
Cordera F, Arciero CA, Li T, Watson JC, Hoffman JP. Significance of common hepatic artery lymph node metastases during pancreaticoduodenectomy for pancreatic head adenocarcinoma. Ann Surg Oncol. 2007;14:2330–6.
Sierzega M, Popiela T, Kulig J, Nowak K. The ratio of metastatic/resected lymph nodes is an independent prognostic factor in patients with node-positive pancreatic head cancer. Pancreas. 2006;33:240–5.
Pawlik TM, Gleisner AL, Cameron JL, et al. Prognostic relevance of lymph node ratio following pancreaticoduodenectomy for pancreatic cancer. Surgery. 2007;141:610–18.
Rau BM, Moritz K, Schuschan S, Alsfasser G, Prall F, Klar E. R1 resection in pancreatic cancer has significant impact on long-term outcome in standardized pathology modified for routine use. Surgery. 2012;152(3 Suppl 1):S103–11.
Fukuda S, Oussoultzoglou E, Bachellier P, et al. Significance of the depth of portal vein wall invasion after curative resection for pancreatic adenocarcinoma. Arch Surg. 2007;142:172–9; discussion 180.
Valle JW, Palmer D, Jackson R, et al. Optimal duration and timing of adjuvant chemotherapy after definitive surgery for ductal adenocarcinoma of the pancreas: ongoing lessons from the ESPAC-3 study. J Clin Oncol. 2014;32:504–12.
La Torre M, Nigri G, Cavallini M, Mercantini P, Ziparo V, Ramacciato G. The glasgow prognostic score as a predictor of survival in patients with potentially resectable pancreatic adenocarcinoma. Ann Surg Oncol. 2012;19:2917–23.
Szkandera J, Stotz M, Absenger G, et al. Validation of C-reactive protein levels as a prognostic indicator for survival in a large cohort of pancreatic cancer patients. Br J Cancer. 2014;110:183–8.
Takahashi H, Ohigashi H, Ishikawa O, et al. Perineural invasion and lymph node involvement as indicators of surgical outcome and pattern of recurrence in the setting of preoperative gemcitabine-based chemoradiation therapy for resectable pancreatic cancer. Ann Surg. 2012;255:95–102.
Hata S, Sakamoto Y, Yamamoto Y, et al. Prognostic impact of postoperative serum CA 19-9 levels in patients with resectable pancreatic cancer. Ann Surg Oncol. 2012;19:636–41.
Chapman WC, Sharp KW, Weaver F, Sawyers JL. Tumor seeding from percutaneous biliary catheters. Ann Surg. 1989;209:708–13; discussion 713–705.
Disclosure
There are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
For the Multicenter Study Group of Pancreatobiliary Surgery (MSG-PBS)
Rights and permissions
About this article
Cite this article
Uemura, K., Murakami, Y., Satoi, S. et al. Impact of Preoperative Biliary Drainage on Long-Term Survival in Resected Pancreatic Ductal Adenocarcinoma: A Multicenter Observational Study. Ann Surg Oncol 22 (Suppl 3), 1238–1246 (2015). https://doi.org/10.1245/s10434-015-4618-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-015-4618-9