Abstract
Background
To assess the correlations and functions of complement C1r/C1s, Uegf, Bmp1 domain-containing protein-1 (CDCP1) in identifying colorectal cancer (CRC) patients who are at high risk for metastasis.
Methods
Tumor specimens from 101 patients were analyzed by real-time polymerase chain reaction to detect CDCP1 expression. CDCP1 expression plasmids and shRNA were used to knock down CDCP1 expression in this study to investigate migratory and invasive abilities by Boyden chambers. The mRNA expression profiles in shCDCP1 transfectants were compared to those in control cells by conducting microarray analysis. Its downstream effectors were also invested in this study.
Results
CRC patients with a high CDCP1 expression had a statistically significant lower overall survival and disease-free survival compared to those exhibiting low CDCP1 expression. In vitro, knock-down CDCP1 expression significantly decreased migratory and invasive abilities in HCT116. Aberrant expression of CDCP1 increased cancer cell migration and invasion. By using integrated genomics, we identified ROCK1 (rho-associated, coiled-coil-containing protein kinase 1 pseudogene 1) as a downstream effector in CDCP1-mediated migration and as an invasion mediator. Clinically, ROCK1 and CDCP1 mRNA expression exhibited a strong positive correlation in CRC patient samples.
Conclusions
Our results implicated CDCP1 as a key regulator of CRC migration and invasion, and suggest that it is a useful prognostic factor for patients with CRC. Improved identification of a high-risk subset of early metastatic patients may guide indications of individualized treatment in clinical practice.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Cancer metastasis is the major cause of death in patients with colorectal cancer (CRC). Upon the diagnosis of metastases, only 10–15 % of patients live for more than 5 years.1 Therefore, a comprehensive analysis of gene expression in a primary tumor and its derived tumors can contribute to the identification of differential genes associated with the metastatic phenotype and the clarification of the molecular mechanism of cancer metastasis.2 Among these possible mechanisms, migration and invasion are the most critical events in cancer metastasis. Hence, determining the gene signatures potentially can be used to distinguish patients with high-risk recurrence or metastasis.
Complement C1r/C1s, Uegf, Bmp1 domain-containing protein-1 (CDCP1) is also known as transmembrane and is associated with Src kinase (TRASK) or substractive immunization Mt HEp3-associated 135 kDa protein (SIMA135). This 140 kDa type I transmembrane glycoprotein exhibits a large extracellular domain and a small intracellular region. The gene encoding CDCP1 was first cloned in 2001 by using cDNA chip hybridizations and is more preferentially expressed in colon cancer cells than healthy cells.3 , 4 It is widely expressed in most epithelial tissues and hematopoietic stem cells.5 , 6 Previous studies have reported that gene encoding CDCP1 is a substrate of Src family kinases when anchorage is lost.7 − 11 Because Src is strongly implicated in the development, maintenance, progression, and metastatic spread of human cancers, the proposal that CDCP1 constitutes a factor in cancer progression has received increasing support. Dysregulated CDCP1 expression has been associated with cancer of the lung, breast, colon, kidney, and endometium.7 , 12 − 17 However, the data have been descriptive and partially conflicting. Several studies have reported that CDCP1 expression increased in tumor tissues compared to that in healthy tissues.13 − 16 This has led to the suggestion that CDCP1 may promote cancer metastasis. In contrast, a high level of CDCP1 is a favorable sign of prognosis in endometrioid carcinoma.17 The various roles of CDCP1 in different tumors are crucial, but their exact mechanism has not yet been sufficiently explained.
At present, CDCP1 has been reported in patients with metastatic gastric, lung, prostate, and renal cell carcinomas as well as in various epithelial cell lines; however, its functional importance in colon cancer cells and its clinical relevance in CRC samples have not been determined. Therefore, in-depth research regarding the role of CDCP1 in CRC is warranted. In this study, we first investigated the association of CDCP1 expression in clinicopathologic parameters and prognosis in CRC patients, and focused on the downstream signaling pathway or pathways of CDCP1 in cell motility.
Materials and Methods
Study Subjects and Surgical Specimens
Our study included 101 consecutive patients who underwent complete surgical resection for primary CRC in National Taiwan University (NTU) Hospital between December 2001 and July 2003. All the surgery was performed by attending surgeons subspecialized to deal with CRC, and their clinical and pathologic data were recorded. There was no patient selection bias in this study. Emergency operations for bowel obstruction or perforation and resection for recurrent diseases were excluded in this analysis. Our study was approved by the Institutional Review Board Committee of NTU Hospital
Real-Time Quantitative Polymerase Chain Reaction
Real-time reverse transcription–polymerase chain reaction (real-time RT-PCR) was achieved using FastStart Universal Probe master mix (Roche, Basel, Switzerland) and amplified using an ECO Sequence detector (Illumine, San Diego, CA, USA). The commercial primers specific for CDCP1 (Hs01080405_m1), ROCK1 (rho-associated, coiled-coil-containing protein kinase 1 pseudogene 1; Hs01127699_m1), and B2M (Hs99999907_m1) were designed by Applied Biosystems (TaqMan Gene Expression Assays); the results were modified using B2M. The experiment was performed 3 times in duplicate, at least. Relative quality expression was calculated using cancer tissue samples and healthy samples, which were used as the control group. The differences in values between groups were then measured as fold differences.
Transient Transfection and Established Stable Clone Cells
The shCDCP1 plasmid (National RNAi Core Facility) or CDCP1 construct vectors were transiently transfected into HCT116 and CaCo-2 cells by using lipofectamin 2000 transfection reagents (Invitrogen, Carlsbad, CA, USA). Briefly, 3 µg of plasmid DNA was mixed with 4.5 µg of transfection reagents. The transfection protocol was performed according to the manufacturer’s instructions (Invitrogen), and the results were verified using Western blot analysis. After 24 h of transfection, the cells were plated in fresh media with 10 % fetal calf serum and 1000 µg/mL G418 or 3 µg/mL puromycin. The resistant clones were selected and cultured.
In Vitro Migration and Invasion Assay
The transwells with pore sizes of 8 µm were coated with or without Matrigel (40 µg; Millipore, Billerica, MA, USA) in 24-well dishes (Nucleopore, Pleasanton, CA, USA). Approximately 3 × 105 cells in 100 µL of complete medium were placed in the upper chamber, and 1 mL of the same medium was placed in the lower chamber; the cells were incubated for 24 and 48 h; subsequently, the cells were fixed in methanol for 15 min and the cells in the upper chamber of the filters were removed using cotton-tipped swabs. The filters containing phosphate-buffered saline (PBS) were cleared and the cells were subsequently stained with 0.05 % crystal violet in PBS for 15 min. The cells in the lower chamber of the filters were observed and counted using a Leica Microsystems (Wetzlar, Germany) microscope system (type 090-135.001). Each clone was plated in duplicate in each experiment, and each experiment was repeated at least 3 times.
Statistical Analysis
An independent Student t test was used to compare the continuous variables between 2 groups, and a chi-square test was applied for the comparison of dichotomous variables. A Kaplan–Meier estimation and log rank test were used to compare the difference in the survival time between patient groups. The level of statistical significance was set at 0.05 for all tests. All statistical calculations were carried out by SPSS version 17.0 for Windows (IBM, Armonk, NY, USA).
Results
CDCP1 mRNA Expression Is Correlated with Advanced Tumor, Node, Metastasis Classification System (TNM) Stage and Poor Survival in CRC Patients
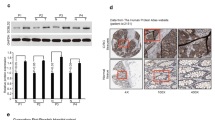
To examine the clinical relevance of CDCP1 in CRC, quantitative RT-PCR was performed to analyze the CDCP1 level in the tumors of patients. Analysis was conducted on 101 CRC patients. The distributions of demographic, clinical, and pathologic features are presented in Table 1. Our results showed that expression of CDCP1 level was highly associated with clinical staging (Fig. 1a, P = 0.009), node metastases (Fig. 1b, P < 0.001), and disease outcome. No statistically significant relationship was found between CDCP1 expression and age, sex, tumor depth, or differentiation. To clarify the correlation between CDCP1 expression and postoperative survival or recurrence-free survival of CRC patients, we first defined the cutoff values of the CDCP1 mRNA levels, which were calculated using receiver-operating characteristic analysis, according to the most accurate predictive probability for recurrence. Using these criteria, the patients were divided into high (fold change >0.3171) or low (fold change ≤0.3171) CDCP1 expression categories. Kaplan–Meier survival curves revealed that patients with low CDCP1 expression (n = 59) significantly higher survival and recurrence-free survival rates than those with high CDCP1 expression (n = 42; P < 0.001 and P = 0.002, respectively, Fig. 1c, d). The cumulative survival rate up to 60 months was 79.1 % in patients with low CDCP1 expression level. In contrast, it was only 38.7 % in those with high CDCP1 expression level. Furthermore, the multivariate analysis summarized in Supplementary Table 1 also revealed that CDCP1 expression level acts as a prognostic markers of CRC. Taken together, because the death of patients with CRC is largely due to metastasis, CDCP1 may play a role in modulating the invasiveness and metastasis.
Real-time quantitative RT-PCR of CDCP1 mRNA expression among 101 patients with CRC. Patient samples were grouped by a TNM stage (P = 0.009) and b lymph node involvement (P < 0.001). Analysis of survival (Kaplan–Meier) between high (fold change >0.3171) or low (fold change ≤0.3171) CDCP1 expression levels. Log rank tests showed statistical difference in c overall survival curves (P < 0.001) and d recurrence-free survival (P = 0.002)
CDCP1 Expression Induces CRC Cell Migration and Invasion
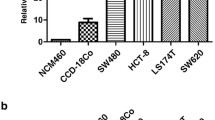
We screened a panel of CRC cell lines with invasion and migration phenotypes on the basis of the results of invasion and migration assays. We observed a positive correlation between endogenous CDCP1 expression and the number of migrating or invading cells. The advanced KM12SM cells with a higher invasive and migratory ability exhibited high CDCP1 protein expression levels compared to the KM12C and KM12sp cells (Fig. 2a). In addition, the cell lines revealed that HCT116, which was highly invasive and had migrated, exhibited high CDCP1 expression and CaCo-2 exhibited low CDCP1 expression, and a low migratory and invasive ability (Fig. 2b). The cell-based data were consistent with the clinical outcome of the treatment of CRC patients.
Correlation of CDCP1 expression and cell migration/invasion abilities in different CRC cell lines. Analysis of endogenous CDCP1 protein expression was immunoblotted in a KM12C serial cells and b other CRC cell lines. Migratory and invasive ability was measured using 8 µm pore transwells for HCT116 cells transiently transfected with shCDCP1 plasmids c and CaCo-2 cells transiently overexpressed with CDCP1 plasmids d. After 48 h of incubation, the migrated cells were stained using crystal violet and counted. e CDCP1 expression level (top) and migratory and invasive ability (bottom) of HCT116 stably transfectants were analyzed by immunoblot analysis and a Boyden chamber. f Photographs of spleens and livers from mice that had their spleen injected with either HCT116/Neo or HCT116/shCDCP1 cells; the livers and spleens were then collected when mice were moribund or after 6 weeks. * P < 0.05; ** P < 0.001
To further investigate whether CDCP1 is involved in CRC migration and invasion, we reduced endogenous CDCP1 expression by using shRNA in HCT116 cells and enhanced CDCP1 in CaCo-2 cells. In HCT116 cells, transient transfection with shCDCP1 significantly decreased their migratory and invasive capabilities (Fig. 2c). In CaCo-2 cells, transiently overexpressed CDCP1 demonstrated a higher migratory and invasive ability (Fig. 2d). In addition, we selected and established HCT116 CDCP1-silenced stable clones and observed that the migratory and invasive ability were significantly decreased (Fig. 2e). To exclude a proliferative effect of CDCP1, we compared the growth rates of HCT116 cells silenced CDCP1 expression. As shown in Supplementary Fig. 1a, the growth rates of these cells were the same, suggesting that the ability of silenced CDCP1 to decrease migratory and invasive cell numbers was not caused by an effect on cell growth ability. Subsequently, we examined the adhesion ability of CDCP1 expression in the HCT116 cells and demonstrated that the adhesion ability was not distinguishable in HCT116 cells, with or without CDCP1 knockdown (Supplementary Fig. 1B). However, a murine liver metastasis model revealed that the number of tumor nodules and the metastasis rate were not different between the control group and the shCDCP1 group in vivo (Fig. 2f; Supplementary Table 2).
Coiled-Coil-Containing Protein Kinase 1 Pseudogene 1 is Essential for CDCP1-Mediated CRC Cell Invasion
To predict the possible downstream target gene of CDCP1-mediated migration and invasion in CRC, a high-throughput microarray analysis was performed to compare the difference of gene expression in HCT116 parent and its CDCP1-silencing transfectants (Fig. 3a; Supplementary Table 3). This revealed the ROCK1 proteins as potential CDCP1 targets. After further examining the effects of CDCP1 on potential target expression, only ROCK1 mRNA level was significantly decreased in stable or transient knockdown of CDCP1 in HCT116 cells (Fig. 3b, c). On the basis of the regulatory role of CDCP1 in ROCK1 expression, we propose that CDCP1-mediated ROCK1 expression and subsequent ROCK1 target changes might contribute to the CDCP1-driven migration and invasion in CRC. In CaCo-2 cells, cotransfected shROCK1 and CDCP1 expressive plasmid restored the migratory and invasive ability of the cells, which did not occur in the control vector group (Fig. 3d). In contrast, overexpressed ROCK1 in shCDCP1 stable transfectants induced its migratory and invasive ability (Fig. 3e).
ROCK1 as a downstream effector of the CDCP1 knockdown expression. a Microarray analysis was performed in HCT116/Neo and HCT116/shCDCP1 stable clones. Decreasing ROCK1 expression was noted in HCT116 cells stably b and transiently c transfected with shCDCP1 construct. d CaCo-2 cells were seeded and transiently cotransfected with shCDCP1 and ROCK1 plasmids for 48 h. e The stable transfectants of HCT116 cells were seeded and transfected with overexpressed ROCK1 plasmid for 48 h. Cells lyses were collected and analyzed by Western blot analysis (top). Transfected cells were seeded into 8 µm pore transwells and stained using crystal violet in 48 h of incubation (bottom)
ROCK1 mRNA Expression is Correlated with TNM Stage in Patients with CRC and is Associated with CDCP1 Expression
We demonstrated that in CRC patient those who were at high TNM stages exhibited high ROCK1 mRNA expression (P = 0.035) (Fig. 4a). Moreover, a statistically significant positive correlation between ROCK1 and CDCP1 mRNA expression was observed (γ = 0.2177, P = 0.001) (Fig. 4b). These data demonstrated that the ROCK1 and CDCP1 prognostic factors in CRC and were highly correlated with their mRNA expression and advanced clinical features.
Clinical association between ROCK1 mRNA expression and recurrence in a subset of patients. a Correlation between ROCK1 expression and TNM stages among 68 patients with CRC (P = 0.035). b Expression of CDCP1 and ROCK1 mRNA in 45 tumor samples. The association between CDCP1 and ROCK1 was significant (γ = 0.2177, P = 0.001)
Discussion
Pathologic staging is the convincible prognostic factor in most cancers, such as CRC, lung cancer, and breast cancer. However, its prognostic reliability is still uncertain in stage II and III CRC patients.18 For CRC patients with stage II disease, the 5 years survival rate is between 60 and 80 %, and for CRC patients at stage III, the 5 years survival rate is between 30 and 60 %.19 This difference in the 5 years survival rate of patients at the same stage might be caused by various factors, such as chemotherapy resistance, early recurrence, or reduced survival, that cannot be predicted using the common staging criteria alone. Therefore, determining the related molecular markers to identify patients at risk of recurrence is critical.
CDCP1 mRNA and protein expressions have been observed in various human cancers, including colon, lung, and breast cancers.4 , 13 , 20 CDCP1 was defined as a protein functionally involved in metastasis and was recently demonstrated to promote the invasion and peritoneal dissemination of gastric carcinoma.7 , 21 In addition, in highly metastatic pancreatic cell lines, phospho-CDCP1 levels are increased and regulate invasion, metastasis, and extracellular matrix degradation.22 Our data suggest that CDCP1-silenced cells decreased migration and invasion, and CDCP1-expressed cells increased migration and invasion (Fig. 2). We consider that CDCP1 is crucial in the progression and metastasis of colon cancer. Although CDCP1 is up-regulated in CRC, this does not imply that it is a factor in oncogenesis because differential gene expression could be only an effect and not a cause of the carcinogenic process.4
According to a previous report, the high expression of CDCP1 cells formed more lung metastases than did the low expression of CDCP1 cells in lung cancer–transplanted nude mice and melanoma-transplanted NOD/SCID mice.14 , 23 However, our animal xenograft studies did not reveal a difference of liver metastasis between HCT116/Neo and HCT116/shCDCP1. It could be explained that CRC metastases involve coexpression of many target genes, and similar studies have showed that simultaneous overexpression of multiple metastasis genes is often required to achieve high metastatic capabilities.24 − 26
To investigate the possible underlying molecular mechanism, a high-throughput microarray was used to detect a molecular fingerprint of genes and thereby identify changes that may promote the shCDCP1-mediated reduction of the migratory and invasive abilities of cancer cells (Fig. 3). One of the most strongly up-regulated genes was ROCK1, which belongs to the serine/threonine kinases family and is a crucial downstream effector of the small GTP-binding protein RhoA.27 The Rho/ROCK pathway was first identified in cardiovascular diseases including angina, hypertension, cardiac hypertrophy, ischemia–reperfusion injury, and chronic heart failure.28 − 34 Moreover, the ROCK family was determined to be implicated in various physiologic functions and associated with cancer progression in the actin cytoskeletal assembly, such as motility, migration, invasion, and cell adhesion.35 , 36 In our studies, silencing ROCK1 reduced the migratory and invasive abilities in CDCP1 expression cells; furthermore, a recent study indicated that blocking the Rho pathway reduced transendothelial migration in prostate cancer.37 To our knowledge our study is the first to demonstrate that ROCK1, as a downstream effector of CDCP1, acts as a positive regulator of CRC cell migration and invasion. Thus, the establishment of the CDCP1-ROCK1 relationship may provide a novel target for therapeutic intervention. Additional in-depth investigations are required to reveal the mechanism involved.
Taken together with our previous results, we expect to provide profound insights into the molecular regulation mechanisms of CDCP1, which leads to cancer progression and serves as a reference for identifying possible therapy targets of downstream genes for designing improved therapeutic strategies and embodiments. Our findings provide the rationale to include CDCP1 inhibitors against CRC in the future.
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics. CA Cancer J Clin. 2009;59:225–49.
Cheung ST, Chen X, Guan XY, et al. Identify metastasis-associated genes in hepatocellular carcinoma through clonality delineation for multinodular tumor. Cancer Res. 2002;62:4711–21.
Uekita T, Sakai R. Roles of CUB domain-containing protein 1 signaling in cancer invasion and metastasis. Cancer Sci. 2011;102:1943–8.
Scherl-Mostageer M, Sommergruber W, Abseher R, Hauptmann R, Ambros P, Schweifer N. Identification of a novel gene, CDCP1, overexpressed in human colorectal cancer. Oncogene. 2001;20:4402–8.
Wortmann A, He Y, Deryugina EI, Quigley JP, Hooper JD. The cell surface glycoprotein CDCP1 in cancer—insights, opportuniites, and challenges. IUBMB Life. 2009;61:723–30.
Bühring HJ, Kuçi S, Conze T, et al. CDCP1 identifies a broad spectrum of normal and malignant stem/progenitor cell subsets of hematopoietic and nonhematopoietic origin. Stem Cells. 2004;22:334–43.
Hooper JD, Zijlstra A, Aimes RT, et al. Subtractive immunization using highly metastatic human tumor cells identifies SIMA135/CDCP1, a 135 kDa cell surface phosphorylated glycoprotein antigen. Oncogene. 2003;22:1783–94.
Brown TA, Yang TM, Zaitsevskaia T, et al. Adhesion or plasmin regulates tyrosine phosphorylation of a novel membrane glycoprotein p80/gp140/CUB domain-containing protein 1 in epithelia. J Biol Chem. 2004;279:14772–83.
Bhatt AS, Erdjument-Bromage H, Tempst P, Craik CS, Moasser MM. Adhesion signaling by a novel mitotic substrate of src kinases. Oncogene. 2005;24:5333–43.
Spassov DS, Baehner FL, Wong CH, McDonough S, Moasser MM. The transmembrane src substrate trask is an epithelial protein that signals during anchorage deprivation. Am J Pathol. 2009;174:1756–65.
Spassov DS, Ahuja D, Wong CH, and Moasser MM. The structural features of trask that mediate its anti-adhesive functions. PLoS One. 2011;6:e19154.
Wong CH, Baehner FL, Spassov DS, et al. Phosphorylation of the src epithelial substrate trask is tightly regulated in normal epithelia but widespread in many human epithelial cancers. Clin Cancer Res. 2009;15:2311–22.
Ikeda JI, Oda T, Inoue M, et al. Expression of CUB domain containing protein (CDCP1) is correlated with prognosis and survival of patients with adenocarcinoma of lung. Cancer Sci. 2009;100:429–33.
Uekita T, Jia L, Narisawa-Saito M, Yokota J, Sakai R. CUB domain-containing protein 1 is a novel regulator of anoikis resistance in lung adenocarcinoma. Mol Cell Biol. 2007;27:7649–60.
Perry SE, Robinson P, Melcher A, et al. Expression of the CUB domain containing protein 1 (CDCP1) gene in colorectal tumour cells. FEBS Lett. 2007;581:1137–42.
Awakura Y, Nakamura E, Takahashi T, et al. Microarray-based identification of CUB-domain containing protein 1 as a potential prognostic marker in conventional renal cell carcinoma. J Cancer Res Clin Oncol. 2008;134:1363–9.
Mamat S, Ikeda JI, Enomoto T, et al. Prognostic significance of CUB domain containing protein expression in endometrioid adenocarcinoma. Oncol Rep. 2010;23:1221–7.
Lin BR, Huang MT, Chen ST, et al. Prognostic significance of TWEAK expression in colorectal cancer and effect of its inhibition on invasion. Ann Surg Oncol. 2012;19:S385–94.
Marsters SA, Sheridan JP, Pitti RM, Brush J, Goddard A, Ashkenazi A. Identification of a ligand for the death-domain-containing receptor Apo3. Curr Biol. 1998;8:525–8.
Bühring HJ, Kuçi S, Conze T, et al. CDCP1 identifies a broad spectrum of normal and malignant stem/progenitor cell subsets of hematopoietic and nonhematopoietic origin. J Biol Chem. 2011;286:42303–15.
Uekita T, Tanaka M, Takigahira M, et al. CUB domain–containing protein 1 regulates peritoneal dissemination of gastric scirrhous carcinoma. Am J Pathol. 2008;172:1729–39.
Miyazawa Y, Uekita T, Hiraoka N, et al. CUB domain–containing protein 1, a prognostic factor for human pancreatic cancers, promotes cell migration and extracellular matrix degradation. Cancer Res. 2010;70:5136–46.
Liu H, Ong SE, Badu-Nkansah K, Schindler J, White FM, Hynes RO. CUB-domain-containing protein 1 (CDCP1) activates Src to promote melanoma metastasis. Proc Natl Acad Sci U S A. 2011;108:1379–84.
Gao W, Chen L, Ma Z, et al. Isolation and phenotypic characterization of colorectal cancer stem cells with organ-specific metastatic potential. Gastroenterology. 2013;145:636–46.
Gupta GP, Nguyen DX, Chiang AC, et al. Mediators of vascular remodelling co-opted for sequential steps in lung metastasis. Nature. 2007;446:765–70.
Ding Q, Chang CJ, Xie X, et al. APOBEC3G promotes liver metastasis in an orthotopic mouse model of colorectal cancer and predicts human hepatic metastasis. J Clin Invest. 2011;121:4526–36.
Satoh K, Fukumoto Y, Shimokawa H. Rho-kinase: important new therapeutic target in cardiovascular diseases. Am J Physiol Heart Circ Physiol. 2011;301:H287–96.
Shimokawa H, Rashid M. Development of Rho-kinase inhibitors for cardiovascular medicine. Trends Pharmacol Sci. 2007;28:296–302.
Lin G, Craig GP, Zhang L, et al. Acute inhibition of Rho-kinase improves cardiac contractile function in streptozotocin-diabetic rats. Cardiovasc Res. 2007;75:51–8.
Kobayashi N, Horinaka S, Mita S, et al. Critical role of Rho-kinase pathway for cardiac performance and remodeling in failing rat hearts. Cardiovasc Res. 2002;55:757–67.
Bao W, Hu E, Tao L, et al. Inhibition of Rho-kinase protects the heart against ischemia/reperfusion injury. Cardiovasc Res. 2004;61:548–58.
Wolfrum S, Dendorfer A, Rikitake Y, et al. Inhibition of Rho-kinase leads to rapid activation of phosphatidylinositol 3-kinase/protein kinase Akt and cardiovascular protection. Arterioscler Thromb Vasc Biol. 2004;24:1842–7.
Noma K, Oyama N, Liao JK. Physiological role of ROCKs in the cardiovascular system. Am J Physiol Cell Physiol. 2006;290:C661–8.
Surma M, Wei L, Shi J. Rho kinase as a therapeutic target in cardiovascular disease. Future Cardiol. 2011;7:657–71.
Hall A. Rho GTPases and the actin cytoskeleton. Science. 1998;279:509–14.
Wan X, Cheng Q, Peng R, et al. ROCK1, a novel target of miR-145, promotes glioma cell invasion. Mol Med Rep. 2014;9:1877–82.
Brown M, Roulson JA, Hart CA, Tawadros T, Clarke NW, Arachidonic acid induction of Rho-mediated transendothelial migration in prostate cancer. Br J Cancer. 2014;110:2099–108.
Acknowledgment
This work was supported by Grants from National Science Council, Taiwan (NSC 99-2314-B-002-083-MY3), National Taiwan University Hospital (NTUH.100-S1508), and Chang Gung Medical Research Foundation, Taiwan (CMRPF6A0043, CMRPF6C0061 and CMRPF6C0062). We thank Dr. Zee-Fen Chang (Institute of Biochemistry and Molecular Biology, College of Medicine and National Yang-Ming University, Taipei, Taiwan, R.O.C.) for providing the ROCK1 plasmid and antibody.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Equal contribution
Yue-Ju Li and Cheng-Chi Chang contributed equally to this article
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chou, CT., Li, YJ., Chang, CC. et al. Prognostic Significance of CDCP1 Expression in Colorectal Cancer and Effect of Its Inhibition on Invasion and Migration. Ann Surg Oncol 22, 4335–4343 (2015). https://doi.org/10.1245/s10434-015-4505-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-015-4505-4