Abstract
Purpose
Yeasts are widely used for the production of bioethanol from biomasses rich in sugar. The present study was aimed at isolating, screening, and characterizing fermentative wild yeast recovered from bio-waste and co-products of Ethiopian sugar factories for bioethanol production using sugarcane molasses as a substrate.
Method
The wild yeasts were identified according to their cellular morphology and D1/D2 and ITS1-5.8S-ITS2 rDNA sequencing. Analysis of ethanol and by-product concentration was done by HPLC equipped with a UV detector. Higher alcohols, acetaldehyde, and methanol were analyzed using GC-MS equipped with a flame ionization detector (FID).
Result
Seven strains (Meyerozyma caribbica MJTm3, Meyerozyma caribbica MJTPm4, Meyerozyma caribbica SHJF, Saccharomyces cerevisiae TA2, Wickerhamomyces anomalus MJTPm2, Wickerhamomyces anomalus 4m10, and Wickerhamomyces anomalus HCJ2F) were found tolerant to 18% (v/v) ethanol, whereas one strain Meyerozyma caribbica MJTm3 tolerated 20%. These strains also showed tolerance to 45°C, 50% of sugar, and pH 2–10. Meyerozyma caribbica MJTm3 produced 12.7% (v/v) of alcohol with an actual ethanol concentration of 26 g L−1, an ethanol yield of 47%, 78% of theoretical yield, and a productivity of 0.54 g L−1 h−1 from 30 °Brix of molasses at 48 h incubation under laboratory scale. Based on the one variable at a time optimization (OVAT), the optimal parameters for maximum bioethanol production were at initial pH 5.5, 35 °Brix, 30°C, 15% inoculum size, 150 rpm, 4 g L−1 di-ammonium phosphate supplement, and 48 h incubation. Under these optimum conditions, 14% (v/v) alcohol, 42 g L−1 actual ethanol concentration, 69% ethanol yield, 89% of theoretical yield, and productivity of 0.88 g L−1 h−1 were obtained.
Conclusion
These results indicated that M. caribbica MJTm3 should further be evaluated, optimized, and improved for industrial bioethanol production due to its fermentation potential.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Background
Industrialization and the continuous growth of the world population are the causes of the rapid utilization of fossil fuels as an energy source. This contributes to an increase in energy costs and the release of massive amounts of greenhouse gases that have adverse effects on the environment (Naik et al. 2010; Solomon 2010). Thus, the development of renewable alternative energy sources is important to minimize the risk of climate change. Biofuels, such as bioethanol, biodiesel, and biogas are major renewable alternative energy sources (Faaij 2006). Bioethanol is an environmentally important fuel that contributes to the reduction of greenhouse gases generated by the global consumption of fossil fuels (de Souza et al. 2018). Moreover, bioethanol has emerged as a potential renewable energy source since it is an environmentally friendly oxygenated fuel (Della-Bianca and Gombert 2013; Hansen et al. 2005).
Ethanol is commercially produced by fermentation of sugar, starch, hemicellulose, and lignocellulose materials. Sugarcane, sugar beet, and sweet sorghum are good sources of fermentable free sugars and are used as feedstock in ethanol production (Ensinas et al. 2009). In the United States, ethanol is produced from cornstarch, in Brazil from sugarcane, in India from cane molasses (Ghosh and Ghose 2003), and in Germany and France from rapeseed oil (Sagar and Kartha 2007). Molasses is a by-product of sugar industries that contains fermentable sugars, especially sucrose, glucose, and fructose (Dhaliwal et al. 2011) which makes it a more cost-effective feedstock than starchy or lignocellulosic materials (Sopandi and Wardah 2017). Molasses is a non-crystallizable residue that contains 60% sucrose that can be hydrolyzed into glucose and fructose by an invertase enzyme during the fermentation process (Dodić et al. 2009). Sugarcane molasses also contains minerals such as calcium 150–2000 mg L−1, potassium 300–12,000 mg L−1, and magnesium 80–3900 mg L−1 (Basso et al. 2011).
Bioconversion of fermentable sugars into fuel ethanol using Saccharomyces cerevisiae is widely used (Kosaric and Velikonja 1995). This is because of the tolerance of S. cerevisiae to a wide range of pH, and ethanol concentrations, its capability to grow in the concentrated fermentation broth, its resistance to inhibitor compounds and high osmotic pressure, and also its ethanologenic characteristics (Dien et al. 2003; Olsson and Hahn-Hägerdal 1993; Pozo et al. 2014). However, searching for an ideal yeast with robust fermentation characteristics (i.e., one that can withstand stress factors, has an excellent invertase activity, and ethanol productivity) is important for converting sucrose to ethanol and is ongoing (Jiménez and Benítez 1986). Several factors are associated with reduced bioethanol yields and low fermentation efficiencies of yeasts; these include quality and pretreatment methods of raw materials, microbial contaminations, and poor performance of yeast cells during ethanol production. During ethanol fermentation processes, yeasts are exposed to high temperatures that affect their fermentation performance (McMeekin et al. 2002; Phisalaphong et al. 2006).
In Ethiopia, Metehara and Fincha sugar factories have ethanol-producing plants that utilize sugarcane molasses as a substrate by diluting it into 12–15 °Brix. Currently, these factories are producing bioethanol with a yield below 7% (v/v) compared to other bioethanol-producing countries of the world that are producing yields of 12–14% (v/v). This indicates that the efficiency of these Ethiopian factories is 50% less efficient than those in other parts of the world. Therefore, the efficiency of the ethanologenic capacity of the distilleries needs to be improved using stress-tolerant and more efficient ethanol-producing wild yeasts that could be screened from the sugar factories’ vicinities. The present research study was aimed at exploring fermentative wild-type yeasts from bio-wastes and co-products of Metehara and Fincha sugar factories for efficient bioethanol production using sugarcane molasses as a substrate.
Results and discussion
Yeast isolation
In this study, wild yeasts were isolated using YPD medium from less commonly studied substrates, such as co-products and bio-wastes of sugar factories. A total of 120 isolates with different cultural characteristics were isolated. Previous studies have shown that yeast strains with excellent ethanol fermentation characteristics were isolated from leaves, flowers, sweet fruits, tree exudates, grain, roots, insects, dung, soil, water, plants, animals, local fermented foods, and beverages (Singh et al. 1998). This is due to their environmental adaptation over time and association with sugar-rich substrates. The colonies grown on the YPD agar medium were circular, smooth, raised, and diverse colors, e.g., creamy, pink, and white. Yeast isolates characterized as spherical, oval, and elongated cells with multipolar budding under a compound microscope were selected for further study (data not shown). Ninety isolates that displayed oval cell shape with multipolar budding were identified morphologically as potential Saccharomyces species.
Screening of yeast isolates for stress tolerance
The majority of the yeast isolates exhibited high tolerance to 10–14% (v/v) ethanol for 48 h (Fig. 1). Seventeen yeast isolates (CJm4c, TA2, MJTm7w, MJTm4c, HCJ2F, CJm3p, CJm1, CJm5c, MJTPm2c, MJTPm1p, MJTm3, MJTPm2, FBm1, SHJF, MJTPm4, MJTm5c, and 4M10) were highly tolerant to 16% (v/v) ethanol. These results were consistent with those of Thammasittirong et al. (2013), who found that yeast isolate UVNR56 tolerated ethanol concentrations up to 15% (v/v). High ethanol concentrations are responsible to induce cellular stress in the metabolism of the yeast and reducing the rate of growth and cell viability (Cray et al. 2013; Hallsworth et al. 2003) which consequently leads to low productivity (Stanley et al. 2010). Yeasts that withstand stress factors and remain viable during the fermentation process are required for industrial applications (Tofighi et al. 2014). In the present study, seven isolates (MJTm3, MJTPm2, MJTPm4, HCJ2F, SHJF, TA2 and 4m10) tolerated up to 18% (v/v) ethanol. Interestingly, isolate MJTm3 exhibited an exceptional tolerance to 20% (v/v). Our result agreed with the findings of Rahman et al. (2013) who isolated yeasts from grapes that showed growth in 20% ethanol. In comparison to our study, the study done by Rahman et al. (2013) and Tikka et al. (2013) reported lower ethanol tolerance (12%) by Saccharomyces species which could be attributed to the difference in the genetic makeup of the yeast isolates and their ecological origin.
In this study, seven yeast isolates (MJTm3, MJTPM4, SHJF, 4M10, TA2, HCJ2F, and MJTPm2) recovered from mill juice and sulfated hot juice tankers were found thermotolerant as they were able to grow at 45°C (Fig. 2). This result is in accordance with Nasir et al. (2017) who reported thermotolerant yeasts isolated from fruits were able to grow up to 44°C. It is also consistent with Techaparin et al. (2017), who reported that yeast isolates designated as KKU-TH33, KKU-TH43, KKU-TH199, KKU-LA1, and KKU-LA4 were tolerant to 45°C. In agreement with the current findings, thermotolerant yeast isolates (40–42°C) were also reported from Thailand, Lao PDR, Vietnam, and Bangladesh (Ramos et al. 2013; Techaparin et al. 2017). High temperature affects the metabolism of yeasts and promotes the formation of acetic acid, glycerol, trehalose, and succinic acid (Singer and Lindquist 1998). The use of thermotolerant yeasts during ethanol production is beneficial because they do not require overall costs necessary for thermal maintenance and distillation (Fonseca et al. 2008), additions of the exogenous enzyme (Abdel-Banat et al. 2010), and have less chance of bacterial contamination (Murata et al. 2015).
Investigation of osmotolerant properties of the yeast isolates was conducted at different glucose concentrations that ranged from 10 to 50% (w/v) (Fig. 3). Twenty-two yeast isolates (TA2, SHJF, MJTPm4, MJTPm3c, MJTPm2c, MJTPm2, MJTPm1p, MJTPm1, MJTm7w, MJTm6c, MJTm5w, MJTm5c, MJTm4c, MJTm31, MJTm3, HCJ2F, FBm1, CJm5c, CJm4c, CJm3p, CJm1, and 4m10) were highly tolerant to 10–40% (v/v) sugar concentration. The results showed that yeast isolates (MJTm3, MJTPM4, SHJF, 4M10, TA2, HCJ2F, and MJTPm2) were tolerant to high glucose concentrations (50%). Among these isolates, MJTm3 showed high tolerance with a viable cell density 2.2 × 107 cells/mL followed by TA2 (1.8 × 107 cells/mL), MJTPm2 (1.5 × 107 cells/mL), HCJ2F (1.4 × 107 cells/mL), 4m10 (1.3 × 107 cells/mL), MJTPm4 (1.2 × 107 cells/mL), and SHJF (8.0 × 106 cells/mL), respectively. Thirty percent of glucose concentration was found favorable for the growth of the yeasts. The increase of sugar concentration up to a certain level results in increased cell density and when it reaches above its optimum level, it results in the decline of the cell population. In the current study, fourteen isolates (TA2, SHJF, MJTPm4, MJTPm2c, MJTPm2, MJTPm1, MJTm6c, MJTm5c, MJTm4c, MJTm31, MJTm3, HCJ2F, FBm1, and 4m10) were highly tolerant to 3.5 pH. Seven yeast isolates (MJTm3, MJTPm2, MJTPm4, HCJ2F, SHJF, and 4m10) were able to grow at pH 2.0. Acidophilous yeasts are beneficial in preventing bacterial contamination during the ethanol fermentation process.
Carbon source assimilation test of stress-tolerant yeast
The results of the carbon assimilation capability of selected yeast isolates using different carbon sources are shown in Table 1. Results showed that majority of yeasts were grown on various carbon sources except for lactose, soluble starch, saccharate, galactonic acid, D-glucuronate, D-galacturonate, L-rhamnose, quinic acid, and methanol. The yeast isolates HCJ2F, MJTPm2, and 4m10 demonstrated delayed growth on soluble starch after 2 weeks of incubation. The inability to ferment pentose sugars by industrial yeast is an intractable challenge in bioethanol production. In the present study, yeast isolates MJTPm4, SHJF, HCJ2F, and 4m10 fermented xylose weakly. MJTm3, MJTPm4, MJTPm2, TA2, 4m10, SHJF, and HCJ2F were able to ferment glucose, galactose, fructose, sucrose, and raffinose (Table 2). MJTPm2, HCJ2F, and TA2 fermented galactose and maltose. HCJ2F, MJTm3, MJTPm4, SHJF, and TA2 fermented trehalose. Results of this study showed diversity in the sugar utilization profile of the yeast isolates.
Identification of the potential bioethanol-producing yeast
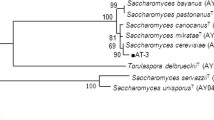
The selected yeast isolates were identified by analyzing the sequences of ITS1/ITS2 and D1/D2 domains. The obtained sequences were compared with sequences retrieved from the NCBI database and identified as Meyerozyma caribbica MJTm3, Meyerozyma caribbica MJTPm4, Meyerozyma caribbica SHJF, Saccharomyces cerevisiae TA2, Wickerhamomyces anomalus MJTPm2, Wickerhamomyces anomalus 4m10, and Wickerhamomyces anomalus HCJ2F. To confirm the phylogenetic position of each strain, several sequences of closely related species were retrieved from the NCBI database and phylogenetic trees were constructed using the MEGA software version 11.0 (Fig. 4). The accession numbers of the reference and type strains indicated in the tree were obtained from the NCBI nucleotide database (https://www.ncbi.nlm.nih.gov/nuccore).
Neighbor-joining based phylogenetic tree of Meyerozyma caribbica strain MJTm3 (OM329077) using maximum likely hood analysis of the combined inter-transcribed sequence (ITS) 1 and 2 regions using MEGA software version 11.0. The numbers provided on branches are frequencies with which a given branch appeared in 1000 bootstrap replications. Meyerozyma guilliermondii CBS 2030 and Meyerozyma caribbica CBS 9966 were used as a type strain as recognized by Schoch et al. (2020). Accession number type strain is in parentheses
Yeast propagation and fermentation dynamics
In this study, the dynamics of the stress-tolerant yeast strains populations showed an increasing trend up to the completion of the propagation of batches. Meyerozyma caribbica MJTm3 was shown to produce the highest cell density in contrast to S. cerevisiae TA2, M. caribbica MJTPm4, W. anomalus MJTPm2, M. caribbica SHJF, W. anomalus 4m10, and W. anomalus HCJ2F. Moreover, the cell density of M. caribbica MJTm3 continued to increase exponentially until the end of the propagation phase and reached a maximum count of 9.8 × 106 cells/mL. The cell density of M. caribbica MJTm3 was shown to increase by 42%, 46%, and 8% at 8 °Brix, 10 °Brix, and 12 °Brix, respectively (Fig. 5). The propagation of yeast cells is a prerequisite for achieving a maximum substrate conversion efficiency during ethanol fermentation (Nurgel et al. 2002).
In this study, the dynamics of alcohol production by the selected stress-tolerant yeast strains were examined using a molasses fermentation medium (Fig. 6a). The thermotolerant (≥ 40°C) and ethanol tolerant (>18% (v/v)) yeast strains were shown to produce ethanol in the range of 8.1–10.5, 10.9–12.7, and 10.7–12.6% (v/v) at 24, 48, and 72 h, respectively. The highest amount of alcohol 12.7% (v/v)) was produced by M. caribbica MJTm3 after 2 days of fermentation. The wild yeasts, M. caribbica MJTm3, W. anomalus HCJ2F, and W. anomalus 4m10 showed the best performance with an alcohol production of 12.7, 12.3, and 12.1% (v/v), respectively at 48 h. This confirms that the use of stress-tolerant yeasts promotes a rapid increase in the concentration of ethanol. The yeast strains showed bioethanol production in the range of 15–26 g L−1, and 44–78% of the theoretical yield. The amount of ethanol produced at 48 h by M. caribbica MJTm3 showed a significant difference (p<0.001) to S. cerevisiae TA2, M. caribbica MJTPm4, W. anomalus MJTPm2, M. caribbica SHJF, W. anomalus 4m10, and W. anomalus HCJ2F. M. caribbica MJTm3 demonstrate high alcohol content (12.7% (v/v)), ethanol concentration (26 g L−1), 78% of theoretical yield, ethanol productivity (0.54 g L−1 h−1), bioethanol yield (47%), and ethanol yield (0.50 g g−1 consumed sugar) at 48 h. The percentage of alcohol was negatively correlated with the molasses concentration (R2=0.90; p=0.052), cell density (R2=0.93; p=0.033), and residual sugar content (R2=0.86; p=0.071).
In the present study, W. anomalus 4m10, M. caribbica MJTm3, S. cerevisiae TA2, and M. caribbica MJTPm4 showed decreasing in ethanol yield as the fermentation time increased to 72 h. This is because the yeast cells entered the stationary phase due to the depletion of fermentable sugar during fermentation (Fig. 6d). This is in agreement with Nadir et al. (2009), where the highest ethanol concentration was obtained at 64 h and then declined by 10% after 72 h. During ethanol fermentation, yeasts produce a considerable amount of by-products, such as organic acids (i.e., acetic acid, propionic acid, and succinic acid), acetone, acetaldehyde, higher alcohols (i.e., n-butanol, 1-propanol, isopropanol), and methanol due to their exposure to stress factors (Mukhtar et al. 2010). Relatively high concentrations of acetone (2.5 g L−1) and methanol (2.4 g L−1) were detected in the final fermented products by W. anomalus HCJ2F and M. caribbica SHJF, respectively (Table 3). Compared to the current concentration, a higher amount of methanol (4.0 g L−1) was reported from sugarcane juice fermentation using S. cerevisiae under optimum conditions (Arshad et al. 2008).
The yeast strains showed a cell density in the range of 0.7–3.1 × 108, 0.7–2.4 × 108, and 1.0–1.8 × 108 cells/mL at 24, 48, and 72 h, respectively (Fig. 6c). Determining the yeast cell density is necessary to monitor ethanol production during fermentation processes (Ramos et al. 2013). Regardless of fermentation time, for majority of the yeast strains, cell counts declined except for strain W. anomalus 4m10, which exhibited better adaptability with the highest cell biomass of 2.0 × 106 cell/mL at 48 h. Furthermore, strain M. caribbica SHJF and W. anomalus HCJ2F showed a fast and significant (p < 0.005) drop in cell counts compared to S. cerevisiae TA2, M. caribbica MJTPm4, W. anomalus MJTPm2, W. anomalus 4m10, and M. caribbica MJTm3 at 48 h, indicating that persistence and growth of these yeast strains were less in the fermentation batches. The correlation analysis of the yeast cell viability with ethanol production (R2=0.93; p=0.033), Brix (R2=0.96; p=0.018), residual sugar (RS%) (R2=0.96; p=0.021), and fermentation time (R2=0.84; p=0.084) showed that these were highly negatively correlated.
The results showed that the RS% concentrations of the fermented substrate decreased inversely proportional to the increase in fermentation time (Fig. 6d). Wickerhamomyces anomalus HCJ2F, W. anomalus MJTPm2, and S. cerevisiae TA2 showed residual sugar content of 1.7%, 1.8%, and 1.8%, respectively, at 48 h. These isolates consumed the fermentable sugar by 97% at 48 h. The correlation analysis of the percentage of residual sugar with ethanol production (R2=0.86; p=0.071) and fermentation time (R2=0.68; p=0.177) was highly negatively correlated, but highly positively correlated with the cell viability (R2=0.96; p=0.021) and the Brix (R2=0.99; p=0.003). Three isolates (M. caribbica MJTPm4, S. cerevisiae TA2, and W. anomalus MJTPm2) showed the best performance in lowering the molasses Brix from 30 °Brix to 12.9, 12.6, and 12.4 °Brix, respectively at 48 h (Fig. 6b). The Brix was highly positively correlated with cell viability (R2=0.96; p=0.018) and RS (R2=0.99; p=0.003), but highly negatively correlated with ethanol production (R2=0.90; p=0.052) and fermentation time (R2=0.68; p=0.0175).
Optimization of fermentation parameters
Initially, the pH factor was optimized, and results showed that alcohol content enhanced as it approached alkaline conditions (Fig. 7d). The maximum alcohol content of 8.73% (v/v) was recorded at a pH value of 5.5 after 2 days of incubation. The optimized pH was maintained in the following optimization experiments. Other parameters, including molasses concentration, inoculum size, rpm, temperature, and supplement concentration, were optimized as described by Fadel et al. (2013).
Alcohol production under various working conditions of M. caribbica MJTm3 to assess the best conditions namely molasses concentration (35 °Brix) and di-ammonium phosphate concentration (4 g L−1, (w/v)) (a), temperature (30°C) and incubation period (48 h) (b), inoculum size (15%, (v/v)) and mixing rate (150 rpm) (c), and pH (5.5) (d)
Different concentrations of molasses (w/v) were used for determining its effect on the alcohol yield using the optimized pH of 5.5. The results showed that 35 °Brix was the most suitable sugar concentration for M.caribbica MJTm3 to produce an alcohol yield of 14.1% (v/v) at 48 h (Fig. 7a). Arshad et al. (2008) and Fadel et al. (2013) reported 9.8 and 7.7% (v/v) alcohol yields from 18 and 29 °Brix of sugarcane molasses, respectively, which are lower than the current value. Regarding supplement optimization, 4 g L−1 DAP concentration was found effectively supporting alcohol production of 15.3% (v/v) by M. caribbica MJTm3 at 48 h (Fig. 7a). Compared to our result, a lower alcohol yield of 8.4% (v/v) was produced by S. cerevisiae F-514 using 4 g L−1 of DAP supplement (Fadel et al. 2013).
An extensive investigation was carried out to determine the effect of different temperatures on alcohol yield. Results of effect of different temperatures on alcohol yield showed that 30°C was found to be favorable for M. caribbica MJTm3 to produce 14.6% (v/v) of alcohol at 48 h (Fig. 7b). Optimization of the mixing rate of the orbital shaker was carried out by regulating its different speeds under the optimized pH of 5.5, 35 °Brix, 4 g L−1 DAP, and 30°C (Fig. 7c). The maximum alcohol content of 15.0% (v/v) was obtained at 150 rpm at 48 h fermentation. The effect of the inoculum size was studied under the previously optimized parameters of pH 5.5, 35 °Brix, 30°C, 150 rpm, and DAP 4 g L−1 (Fig. 7c). Maximum alcohol content of 14.6% (v/v) was found at an inoculum size of 15% (v/v). Previously, a 20% of inoculum size resulted in 9.8% (v/v) of alcohol content (Fadel et al. 2013), which is lower than our result. The incubation period (h) was optimized using all the indicated optimized parameters, i.e., pH 5.5, 35 °Brix, 30°C, 150 rpm, DAP 4 g L−1, and 15% inoculum size (Fig. 7b). With these optimized conditions, M. caribbica MJTm3 produced 15.4% (v/v) alcohol at 48 h.
To verify the reliability of the results from the one variable at a time (OVAT) experiments, batch fermentation under the optimum operating conditions of the corresponding parameters was carried out in a bioreactor with a working volume of 5 L. Meyerozyma caribbica MJTm3 yielded a maximal alcohol percentage of 14% (v/v), an ethanol concentration of 42 g L−1, an ethanol yield of 69%, and 89% of theoretical yield, and productivity of 0.88 g L−1 h−1 (Table 4). The ethanol concentration increased by approximately 38% compared to the unoptimized conditions. In addition, lower concentrations of acetone (0.4 g L−1), acetaldehyde (0.4 g L−1), methanol (0.3 g L−1), isopropanol (0.2 g L−1), 1-butanol (0.2 g L−1), and n-butanol (0.2 g L−1) were detected. In connection to this, other compounds, such as acetic acid, propionic acid, succinic acid, iso-butanol, and n-propanol, were undetected (Table 4). Arshad et al. (2008) reported the detection of higher amounts of methanol (4.0 g L−1) and aldehydes (22.6 g L−1) under optimum conditions from sugarcane juice fermented by S. cerevisiae.
Materials and methods
Samples collection
A total of 176 samples were collected from sugary substrates including mill juice, raw juice, mixed juice, sulfated hot juice, hot clear juice, clear juice, muddy juice, bagasse, fine bagasse, tandem B juice, and sugar cane molasses of Metehara and Fincha Sugar Factories for the isolation of wild fermentative yeasts. These sites are located at (851′ N 39° °52′ E and 8° 31′ N 39° 12′ E, respectively) and are characterized by a humid subtropical climate with average temperatures of 31°C. Samples were transported to Microbial Biotechnology Laboratory, Departments of Biological and Chemical Engineering, Addis Ababa Science and Technology University in an icebox. The samples were kept at 4°C for further investigation.
Wild yeasts isolation and characterization
For isolation of yeasts, 0.1 mL dilution of each sample was spread plated on pre-dried yeast extract peptone dextrose (YPD) agar medium (Pons et al. 1986) with a composition of (10 g L−1 yeast extract, 20 g L−1 peptone, 20 g L−1 dextrose, and 20 g L−1 agar) (Ramos et al. 2013) and supplemented with 100 mg L−1 chloramphenicol to suppress bacterial contaminants (Duarte et al. 2009) and incubated at 30°C for 72 h.
The growing colonies were distinguished based on their cultural characteristics (Boboye and Dayo-Owoyemi 2009; Boekhout and Kurtzman 1996). The colonies were further purified by repeated streaking on a YPD agar medium. Yeast isolates were identified based on their cellular morphology under a microscope and further identified to strain levels using molecular methods. Morphological characterization (i.e., shape, size, and budding) was performed according to the classical method as described in Van der Walt (1984). Isolates were designated according to the origin of the sample and their colony texture on the YPD plates. The pure yeast isolates were preserved using 20% (v/v) glycerol and stored at −80°C. A carbon assimilation test was conducted using the liquid medium method (Bhadra et al. 2008; Matos et al. 2021).
YPD broth was used for determining the sugar fermentation profile of the wild yeast isolates. Nine mL of the YPD broth and 1 mL of filter sterile 10% (w/v) sugars (glucose, galactose, maltose, sucrose, trehalose, lactose, raffinose, fructose, and xylose) were distributed into the fermentation tubes (Nasir et al. 2017), and inoculated with 0.1 mL of overnight active yeast cultures. Thereafter, sterile Durham tubes were inserted into the fermentation tubes and cotton was plugged and incubated at 30°C for 14 days at 150 revolutions per minute (rpm) evaluated every 24 h (Matos et al. 2021). Fermentation capabilities of the yeast isolates were evaluated by observing the formation of gas in the Durham tubes (Barnett et al. 1990). Finally, the potential fermentative yeasts were selected for further screening under stress conditions.
Screening of wild yeasts under stress conditions
The ethanol tolerance test was performed by inoculating each yeast isolate (1 × 106 cells/mL or OD 0.6) into YPD broth supplemented with varying concentrations of absolute ethanol (v/v) (10, 12, 14, 16, 18, and 20%) (Rahman et al. 2013; Techaparin et al. 2017). Subsequently, the cultures were incubated at 30°C for 24-72 h at 150 rpm. Enumeration of the viable cell population was done with a hemocytometer and methylene blue staining method (Alfenore et al. 2004). As an index of ethanol tolerance, based on the percentage of cell viability at 48 h, the yeast isolates were grouped into three categories as follows: highly tolerant (> 50% survival), moderately tolerant (25–50% survival), and slightly tolerant (25% survival) (Negi et al. 2013). Yeast isolates showing high tolerance at 16% (v/v) of ethanol were selected for further screening.
In order to assess the temperature tolerance activity of ethanol-tolerant yeasts, the yeasts were spread-plated on YPD agar and incubated at 37, 40, 42, 44, and 45°C for 72 h (Breisha 2010; Rahman et al. 2013). The growth of the yeast isolates was confirmed by directly observing their colony appearance on YPD agar plates. Yeast isolates capable of growing at 40°C or higher were selected for further screening. Osmotolerance and pH tolerance of the yeast isolates were evaluated quantitatively by transferring portions of active yeast cultures (1 × 106 cells/mL) into YPD broth adjusted to different pH values (i.e., 2, 3, 4, 5, 6, 7, 8, 9, and10) (Rahman et al. 2013) and glucose concentrations (i.e., 10, 20, 30, 40, 50, 55, and 60%) (Fakruddin et al. 2013) and incubated at 30 °C for 72 h at 150 rpm. Viable cell density and percentage of cell survival were determined as described by Negi et al. (2013). Yeasts grew at ≥ 16% (v/v) of ethanol concentration, ≥ 40°C, ≥ 2.0 pH, and ≥ 30% of glucose concentration were selected for molasses fermentation.
Molecular identification of the stress-tolerant yeast isolates
Extraction of genomic DNA of yeast isolates was done using the Ultraclean Microbial DNA Isolation Kit (MoBio Laboratories Inc., Solana Beach, USA). The D1/D2 region of the large subunit (LSU) and internal transcribed spacer ITS1-5.8S-ITS2 rDNA region (ITS1/ITS2) was amplified using primer pairs LR5 and LROR (Vilgalys and Hester 1990) and V9G and LS266 (De Hoog and van den Ende 1998; Masclaux et al. 1995), respectively. PCR amplification was done using an initial denaturing at 95°C for 5 min; 35 cycles under the following conditions: 95°C for 45 s, 55°C for 45 s, 72°C for 60 s; and final extensions at 72°C for 4 min. The quality of the PCR products was analyzed by agarose gel electrophoresis (1.5% agarose) and sequenced using the Sanger sequencer (Applied Biosystem, HITACHI, 3730XL DNA Analyzer) according to the manufacturer’s recommendations at Westerdijk Fungal Biodiversity Institute, Netherlands. The sequences were compared pairwise to those previously deposited sequences from the genebank database using the BLAST search program (https://blast.ncbi.nlm.nih.gov/Blast.cgi) and then aligned with the multiple alignment program CLUSTAL W (Fell et al. 2000). The neighbor-joining method was applied to construct a phylogenetic tree based on the ITS1/ITS2 and D1/D2 rDNA sequences using MEGA software version 11.0. The evolutionary distances were computed using the maximum composite likelihood method (López et al. 2010).
Bioethanol production from molasses
Pretreatment of molasses
Raw molasses was collected from Fincha Sugar Factory and pretreated with 99.8% sulfuric acid (H2SO4) to remove unwanted particles, dirt, and retarding microbial contaminants (De Vasconcelos et al. 2004b; Malik 2016; Rahman et al. 2013). The desired Brix was obtained by diluting raw molasses with distilled water. For yeast cell propagation, molasses concentration was adjusted to 8, 10, and 12 °Brix, whereas for ethanol fermentation, was diluted to 30 °Brix. Before adjusting pH, the medium was supplemented with 5 g L−1 di-ammonium phosphate (DAP) and homogenized using a magnetic stirrer. Finally, pH was adjusted to 4.0–4.6 using H2SO4 and autoclaved at 121°C for 15 min. The Treated medium was standing overnight to sediment undesirable constituents, i.e., sludge, ash contents, and other particulates (Arshad et al. 2008).
Inoculum preparation and propagation
Yeast isolates (24–48 h old cultures) were inoculated into 10 mL of YPD broth in test tubes at pH 4.6 and incubated at 30°C and 150 rpm for 24 h (Flayeh 2017). Yeast cells were harvested by centrifuging at 5000 g for 2 min at 4°C. Harvested cells were washed by resuspending the pellet in sterile distilled water and recentrifuged. For propagation of the inoculum, cell biomass was re-suspended in 10 mL of sterile distilled water (Boboye and Dayo-Owoyemi 2009).
Propagation of the inoculum was initiated in 250 mL Erlenmeyer flasks that contain 100 mL of molasses diluted to 8 °Brix supplemented with 5 g L−1 of DAP (Prescott et al. 2002). The pH of the fermentation medium was adjusted to 4.6 using H2SO4 and autoclaved at 121°C for 15 min (De Vasconcelos et al. 2004a). After cooling, 0.1 mL of aliquots of active yeast cultures were added into flasks and placed in a rotary incubator at 30°C for 24 h with vigorous shaking (150 rpm) to obtain homogeneous suspensions. At the end of the propagation phase, samples were collected and subjected to measurement of Brix, pH, and cell viability using a refractometer, pH meter, and hemocytometer, respectively.
Propagated yeast cultures were transferred to the second stage of propagation in Erlenmeyer flasks of 500 mL containing 100 mL of 10 °Brix fresh molasses medium supplemented with 5 g L−1 of DAP and sterilized at 121°C for 15 min and incubated at 30°C for 24 h at 150 rpm. Then after, the Brix, pH, and viable cells of the propagated cultures were measured and counted. Propagated yeast cultures were transferred into a third propagation flask containing 100 mL of sterilized 12 °Brix of molasses for further yeast cell propagation. Finally, fermented broth samples were collected to determine the viable cells, residual sugar (RS%), ethanol content, and Brix. The yeast cultures with ≥ 3.0 × 108 cells/mL, RS% below 3.0% and ethanol content 3–4% (v/v) were taken for molasses fermentation (Mukhtar et al. 2010).
Fermentation
Batch fermentation system was used to test the bioethanol production efficiency of the selected stress tolerant wild yeasts. Fermentation was initiated in 2 L Erlenmeyer flasks containing 1 L of 30 °Brix of molasses fermentation medium (MFM). Then MFM was inoculated with the propagated yeast culture at an initial yeast cell density of (3.13–9.80 × 108 cells/mL). The fermentation process was performed at pH 4.30, and 30°C with continuous orbital shaking at 150 rpm for 72 h. Portions of the fermented samples were collected aseptically at 24 h intervals from 24 to 72 h of fermentation. The fermentation was considered complete when the Brix level was stable (Duarte et al. 2009).
Analytical methods
The Brix of fermented samples was measured using a refractometer (Atago densimeter model 2312; Atago Co. Ltd., Tokyo, Japan). Yeast cell counts were determined using a compound microscope (Labomed, USA, 100×) using a hemocytometer (Ebeler 1997). Yeast cell viability was tested using 1% methylene blue (Alfenore et al. 2004). Residual sugar was determined using the 3, 5-dinitrosalicylic acid method (DNS) (Miller 1959). The alcohol content of the fermented sample was measured with an Ebulliometer (Latif and Rajoka 2001). Analyses of the ethanol, acetic acid, propionic acid, succinic acid, acetone, and glycerol concentration were done by high-performance liquid chromatography (HPLC) (1200 Series Agilent HPLC, Germany) equipped with a UV detector (Duarte et al. 2009). Content of the higher alcohols (n-butanol, 1-butanol, iso-butanol, 1-propanol, and isopropanol), acetaldehyde, and methanol was analyzed using a Gas chromatography-mass spectrophotometer (Shimadzu GC-17A version 3.0) equipped with a flame ionization detector (FID) (Shen et al. 2003). Calculation of the bioethanol yield (YE/S) and percent of theoretical yield were done according to Hamouda et al. (2015) and Laopaiboon et al. (2009).
Optimization of fermentation process parameters
A batch fermentation system was used to optimize the process parameters, including inoculum size (10, 15, 20, 25, 30, and 35%), pH (3.5, 4.0, 4.5, 5.0, 5.5, 6.0, and 6.5), temperature (25, 30, 35, and 40°C), agitation (50, 80, 110, 130, 150, 180, and 210 rpm), DAP supplement (1, 2, 3, 4, and 5 g L−1) and molasses concentration (15, 20, 25, 30, 35, and 40 °Brix) by applying the one variable at a time method (OVAT) (De Vasconcelos et al. 2004b; Fadel et al. 2013). Accordingly, raw molasses was diluted with distilled water to the desired Brix (8, 10, and 12 °Brix) and then yeast cells were allowed to propagate until the Brix concentration dropped by half. After obtaining the required cell number and residual sugar < 2%, batch fermentation was conducted for ethanol production. Samples were withdrawn at intervals of 24 h for 3 days and analyzed for alcohol content using Ebulliometer (Latif and Rajoka 2001).
Fermentation under optimum conditions
Batch fermentation was conducted under optimal fermentation conditions obtained from one variable at a time optimization step. After sterilization, the broth was cooled and inoculated with 15% (v/v) inoculum size having ≈9.0 × 108 cells/mL yeast suspension into a bioreactor (ECMA-C20604RS, Taiwan) with a working volume of 5 L containing 35 °Brix. The fermentation process was anaerobic and operated at pH 5.5, the temperature of 30°C, and agitation at 150 rpm. Upon completion of the fermentation, samples were collected after 48 h of fermentation. The fermented samples were subjected to ethanol % (v/v), ethanol concentration (g L−1), ethanol yield (%), and byproduct analysis following the previously described analytical protocols.
Data analysis
The data obtained from the ethanol fermentation experiments were analyzed using one-way ANOVA analysis, and Duncan’s multiple range test was used for means separation. The significant difference in the variables was considered at p < 0.05 using SPSS version 26. The correlation of fermentation process parameters was determined using the Pearson correlation method. All of the experiments were done in triplicate. The results are expressed as mean ± standard deviation.
Conclusion
In the present study, seven yeast strains, specifically M. caribbica MJTm3, M. caribbica MJTPm4, W. anomalus MJTPm2, M. caribbica SHJF, W. anomalus 4m10, W. anomalus HCJ2F, and S. cerevisiae TA2 were found to be multi-stress tolerant. Moreover, the non-conventional strains particularly M. caribbica MJTPm4, M. caribbica SHJF, W. anomalus MJTPm2, W. anomalus 4m10, and W. anomalus HCJ2F were capable of fermenting both hexose and pentose sugars. M. caribbica MJTm3 showed the highest alcohol-producing capacity. Under laboratory scale, M. caribbica MJTm3 yielded a maximum alcohol production (12.7% (v/v)), ethanol concentration (26 g L−1), productivity (0.54 g L−1 h−1), ethanol yield (0.50 g g−1), and low byproduct concentration at 30 °Brix of molasses, 30°C, 15% inoculum size, 150 rpm, 4 g L−1 DAP, 5.5 pH, and 48 h fermentation. In contrast to unoptimized conditions, the ethanol concentration was increased approximately by 38% after optimization. The candidate yeast strain (M. caribbica MJTm3) was found a stress-tolerant isolate and producing maximum alcohol of 14% (v/v) with a concentration of 42 g L−1 under the optimized fermentation conditions and has the potential to be used for bioethanol production from high Brix sugarcane molasses. The molecular mechanisms such as the heat shock response, oxidative stress defense, and cellular signaling by which yeast cells acquire tolerance to various stress conditions, should be examined.
Availability of data and materials
The data and materials are available according to request.
References
Abdel-Banat BM, Hoshida H, Ano A, Nonklang S, Akada R (2010) High-temperature fermentation: how can processes for ethanol production at high temperatures become superior to the traditional process using mesophilic yeast? Appl Microbiol Biotechnol 85(4):861–867. https://doi.org/10.1007/s00253-009-2248-5.
Alfenore S, Cameleyre X, Benbadis L, Bideaux C, Uribelarrea J-L, Goma G, Molina-Jouve C, Guillouet S (2004) Aeration strategy: a need for very high ethanol performance in Saccharomyces cerevisiae fed-batch process. Appl Microbiol Biotechnol 63(5):537–542. https://doi.org/10.1007/s00253-003-1393-5.
Arshad M, Khan Z, Shah F, Rajoka M (2008) Optimization of process variables for minimization of byproduct formation during fermentation of blackstrap molasses to ethanol at industrial scale. Lett Appl Microbiol 47(5):410–414. https://doi.org/10.1111/j.1472-765X.2008.02446.x
Barnett JA, Payne RW, Yarrow D (1990) Yeasts: characteristics and identification.
Basso LC, Basso TO, Rocha SN (2011) Ethanol production in Brazil: the industrial process and its impact on yeast fermentation. Biofuel Prod-Recent Dev Prosp 1530:85–100
Bhadra B, Rao RS, Singh PK, Sarkar PK, Shivaji S (2008) Yeasts and yeast-like fungi associated with tree bark: diversity and identification of yeasts producing extracellular endoxylanases. Curr Microbiol 56(5):489–494. https://doi.org/10.1007/s00284-008-9108-x.
Boboye B, Dayo-Owoyemi I (2009) Evaluation of dough sensory properties impacted by yeasts isolated from cassava. J Appl Sci 9(4):771–776
Boekhout T, Kurtzman CP (1996) Principles and methods used in yeast classification, and an overview of currently accepted yeast genera. Nonconvent Yeasts Biotechnol, 1–81. https://doi.org/10.1007/978-3-642-79856-6_1.
Breisha GZ (2010) Production of 16% ethanol from 35% sucrose. Biomass Bioenergy 34(8):1243–1249. https://doi.org/10.1016/j.biombioe.2010.03.017
Cray JA, Russell JT, Timson DJ, Singhal RS, Hallsworth JE (2013) A universal measure of chaotropicity and kosmotropicity. Environ Microbiol 15(1):287–296. https://doi.org/10.1111/1462-2920.12018
De Hoog G, van den Ende AG (1998) Molecular diagnostics of clinical strains of filamentous Basidiomycetes: Molekulare Diagnostik klinischer Stämme filamentöser Basidiomyzeten. Mycoses 41(5–6):183–189. https://doi.org/10.1111/j.1439-0507.1998.tb00321.x.
de Souza JP, do Prado CD, Eleutherio EC, Bonatto D, Malavazi I, da Cunha AF (2018) Improvement of Brazilian bioethanol production–Challenges and perspectives on the identification and genetic modification of new strains of Saccharomyces cerevisiae yeasts isolated during ethanol process. Fungal Biol 122(6):583–591. https://doi.org/10.1016/j.funbio.2017.12.006
De Vasconcelos J, Lopes C, De Franca F (2004a) Continuous ethanol production using yeast immobilized on sugar-cane stalks. Braz J Chem Eng 21(3):357–365. https://doi.org/10.1590/S0104-66322004000300002
De Vasconcelos J, Lopes C, De Franca F (2004b) Continuous ethanol production using yeast immobilized on sugar-cane stalks. Braz J Chem Eng 21:357–365. https://doi.org/10.1590/S0104-66322004000300002
Della-Bianca BE, Gombert AK (2013) Stress tolerance and growth physiology of yeast strains from the Brazilian fuel ethanol industry. Antonie Van Leeuwenhoek 104(6):1083–1095. https://doi.org/10.1007/s10482-013-0030-2
Dhaliwal SS, Oberoi HS, Sandhu SK, Nanda D, Kumar D, Uppal SK (2011) Enhanced ethanol production from sugarcane juice by galactose adaptation of a newly isolated thermotolerant strain of Pichia kudriavzevii. Bioresour Technol 102(10):5968–5975. https://doi.org/10.1016/j.biortech.2011.02.015
Dien B, Cotta M, Jeffries T (2003) Bacteria engineered for fuel ethanol production: current status. Appl Microbiol Biotechnol 63(3):258–266. https://doi.org/10.1007/s00253-003-1444-y
Dodić S, Popov S, Dodić J, Ranković J, Zavargo Z, Mučibabić RJ (2009) Bioethanol production from thick juice as intermediate of sugar beet processing. Biomass Bioenergy 33(5):822–827. https://doi.org/10.1016/j.biombioe.2009.01.002
Duarte WF, Dias DR, de Melo Pereira GV, Gervásio IM, Schwan RF (2009) Indigenous and inoculated yeast fermentation of gabiroba (Campomanesia pubescens) pulp for fruit wine production. J Ind Microbiol Biotechnol 36(4):557–569. https://doi.org/10.1007/s10295-009-0526-y
Ebeler SE (1997) Phytochemicals and Wine Flavor. In: Johns T, Romeo JT (eds) Functionality of Food Phytochemicals. Springer, US, pp 155–178. https://doi.org/10.1007/978-1-4615-5919-1_7
Ensinas A, Modesto M, Nebra S, Serra L (2009) Reduction of irreversibility generation in sugar and ethanol production from sugarcane. Energy 34(5):680–688. https://doi.org/10.1016/j.energy.2008.06.001
Faaij A (2006) Modern biomass conversion technologies. Mitig Adapt Strat Glob Chang 11(2):343–375. https://doi.org/10.1007/s11027-005-9004-7
Fadel M, Keera AA, Mouafi FE, Kahil T (2013) High level ethanol from sugar cane molasses by a new thermotolerant Saccharomyces cerevisiae strain in industrial scale. Biotechnol Res Int. https://doi.org/10.1155/2013/253286
Fakruddin M, Islam MA, Quayum MA, Ahmed MM, Chowdhury N (2013) Characterization of stress tolerant high potential ethanol producing yeast from agro-industrial waste. Am J Biosci 1(2):24–34. https://doi.org/10.11648/j.ajbio.20130102.11
Fell JW, Boekhout T, Fonseca A, Scorzetti G, Statzell-Tallman A (2000) Biodiversity and systematics of basidiomycetous yeasts as determined by large-subunit rDNA D1/D2 domain sequence analysis. Int J Syst Evol Microbiol 50(3):1351–1371. https://doi.org/10.1099/00207713-50-3-1351
Flayeh HM (2017) Optimization of Process Parameters for Bioethanol Production from Low grade Iraqi Dates. Al-Nahrain J Eng Sci 20(3):647–656.
Fonseca GG, Heinzle E, Wittmann C, Gombert AK (2008) The yeast Kluyveromyces marxianus and its biotechnological potential. Appl Microbiol Biotechnol 79(3):339–354. https://doi.org/10.1007/s00253-008-1458-6
Ghosh P, Ghose TK (2003) Bioethanol in India: recent past and emerging future. In: Biotechnology in India II, pp 1–27. https://doi.org/10.1007/3-540-36466-8_1
Hallsworth JE, Heim S, Timmis KN (2003) Chaotropic solutes cause water stress in Pseudomonas putida. Environ Microbiol 5(12):1270–1280. https://doi.org/10.1111/j.1462-2920.2003.00478.x
Hamouda HI, Nassar HN, Madian HR, Amr SSA, El-Gendy NS (2015) Response surface optimization of bioethanol production from sugarcane molasses by Pichia veronae strain HSC-22. Biotechnol Res Int 2015. https://doi.org/10.1155/2015/905792
Hansen AC, Zhang Q, Lyne PW (2005) Ethanol–diesel fuel blends––a review. Bioresour Technol 96(3):277–285. https://doi.org/10.1016/j.biortech.2004.04.007
Jiménez J, Benítez T (1986) Characterization of wine yeasts for ethanol production. Appl Microbiol Biotechnol 25(2):150–154. https://doi.org/10.1007/BF00938939
Kosaric N, Velikonja J (1995) Liquid and gaseous fuels from biotechnology: challenge and opportunities. FEMS Microbiol Rev 16(2–3):111–142. https://doi.org/10.1111/j.1574-6976.1995.tb00161.x
Laopaiboon L, Nuanpeng S, Srinophakun P, Klanrit P, Laopaiboon P (2009) Ethanol production from sweet sorghum juice using very high gravity technology: effects of carbon and nitrogen supplementations. Bioresour Technol 100(18):4176–4182. https://doi.org/10.1016/j.biortech.2009.03.046
Latif F, Rajoka MI (2001) Production of ethanol and xylitol from corn cobs by yeasts. Bioresour Technol 77(1):57–63. https://doi.org/10.1016/S0960-8524(00)00134-6
López CVG, García M d CC, Fernández FGA, Bustos CS, Chisti Y, Sevilla JMF (2010) Protein measurements of microalgal and cyanobacterial biomass. Bioresour Technol 101(19):7587–7591. https://doi.org/10.1016/j.biortech.2010.04.077
Malik, H. (2016). Utilization of agro-industrial wastes for the biomass production of baker‟ s yeast M. Sc. Thesis, Punjab Agricultural University, Punjab.
Masclaux F, Guého E, De Hoog G, Christen R (1995) Phylogenetic relationships of human-pathogenic Cladosporium (Xylohypha) species inferred from partial LS rRNA sequences. J Med Vet Mycol 33(5):327–338. https://doi.org/10.1080/02681219580000651
Matos ITSR, de Souza VA, Giovana do Rosário DÂ, Astolfi-Filho S, Vital MJS (2021) Yeasts with Fermentative Potential Associated with Fruits of Camu-Camu (Myrciaria Dubia, Kunth) from North of Brazilian Amazon. https://doi.org/10.1155/2021/9929059
McMeekin T, Olley J, Ratkowsky D, Ross T (2002) Predictive microbiology: towards the interface and beyond. Int J Food Microbiol 73(2-3):395–407. https://doi.org/10.1016/S0168-1605(01)00663-8
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31(3):426–428.
Mukhtar K, Asgher M, Afghan S, Hussain K, Zia-Ul-Hussnain S (2010) Comparative study on two commercial strains of Saccharomyces cerevisiae for optimum ethanol production on industrial scale. J Biomed Biotechnol 2010. https://doi.org/10.1155/2010/419586.
Murata Y, Danjarean H, Fujimoto K, Kosugi A, Arai T, Ibrahim WA, Suliman O, Hashim R, Mori Y (2015) Ethanol fermentation by the thermotolerant yeast, Kluyveromyces marxianus TISTR5925, of extracted sap from old oil palm trunk. Aims Energ 3(2):201–213. https://doi.org/10.3934/energy.2015.2.201
Nadir N, Mel M, Karim M, Yunus R (2009) Comparison of sweet sorghum and cassava for ethanol production by using Saccharomyces cerevisiae. J Appl Sci 9(17):3068–3073. https://doi.org/10.3923/jas.2009.3068.3073
Naik SN, Goud VV, Rout PK, Dalai AK (2010) Production of first and second generation biofuels: a comprehensive review. Renew Sustain Energy Rev 14(2):578–597. https://doi.org/10.1016/j.rser.2009.10.003
Nasir A, Rahman SS, Hossain MM, Choudhury N (2017) Isolation of Saccharomyces cerevisiae from pineapple and orange and study of metal’s effectiveness on ethanol production. Eur J Microbiol Immunol 7(1):76–91. https://doi.org/10.1556/1886.2016.00035
Negi B, Sharma P, Kashyap S, Seth S, Dey G (2013) Screening of yeast strains for vinification of fruits from cold desert regions of North West India. Int Food Res J 20(2):975
Nurgel C, Erten H, Canbaş A, Cabaroğlu T, Selli S (2002) Influence of Saccharomyces cerevisiae strains on fermentation and flavor compounds of white wines made from cv. Emir grown in Central Anatolia, Turkey. J Ind Microbiol Biotechnol 29(1):28–33. https://doi.org/10.1038/sj.jim.7000258
Olsson L, Hahn-Hägerdal B (1993) Fermentative performance of bacteria and yeasts in lignocellulose hydrolysates. Process Biochem 28(4):249–257. https://doi.org/10.1016/0032-9592(93)80041-E
Phisalaphong M, Srirattana N, Tanthapanichakoon W (2006) Mathematical modeling to investigate temperature effect on kinetic parameters of ethanol fermentation. Biochem Eng J 28(1):36–43. https://doi.org/10.1016/j.bej.2005.08.039
Pons M-N, Rajab A, Engasser J-M (1986) Influence of acetate on growth kinetics and production control of Saccharomyces cerevisiae on glucose and ethanol. Appl Microbiol Biotechnol 24(3):193–198. https://doi.org/10.1007/BF00261536
Pozo MI, Lievens B, Jacquemyn H (2014) Impact of microorganisms on nectar chemistry, pollinator attraction and plant fitness. In: Nectar: production, chemical composition and benefits to animals and plants. New York: Nova Science Publishers, pp 1–40
Prescott L, Harley J, Klein D (2002) Microbiology, 5th edn. McGraw–Hill compenies, Chapeter, 41, New York, pp 964–976
Rahman SS, Hossain MM, Choudhury N (2013) Effect of various parameters on the growth and ethanol production by yeasts isolated from natural sources. Bangladesh J Microbiol 30(1-2):49–54. https://doi.org/10.3329/bjm.v30i1-2.28453
Ramos CL, Duarte WF, Freire AL, Dias DR, Eleutherio ECA, Schwan RF (2013) Evaluation of stress tolerance and fermentative behavior of indigenous Saccharomyces cerevisiae. Braz J Microbiol 44(3):935–944. https://doi.org/10.1590/S1517-83822013005000051
Sagar AD, Kartha S (2007) Bioenergy and sustainable development? Annu Rev Env Resour 32:131–167. https://doi.org/10.1146/annurev.energy.32.062706.132042
Schoch CL, Ciufo S, Domrachev M, Hotton CL, Kannan S, Khovanskaya R, Leipe D, Mcveigh R, O’Neill K, Robbertse B (2020) NCBI Taxonomy: a comprehensive update on curation, resources and tools. Database 2020. https://doi.org/10.1093/database/baaa062.
Shen H-Y, Moonjai N, Verstrepen K, Delvaux F (2003) Impact of attachment immobilization on yeast physiology and fermentation performance. J Am Soc Brewing Chem 61(2):79–87. https://doi.org/10.1094/ASBCJ-61-0079
Singer MA, Lindquist S (1998) Thermotolerance in Saccharomyces cerevisiae: the Yin and Yang of trehalose. Trends Biotechnol 16(11):460–468. https://doi.org/10.1016/S0167-7799(98)01251-7
Singh D, Nigam P, Banat I, Marchant R, McHale A (1998) Ethanol production at elevated temperatures and alcohol concentrations: Part II–Use of Kluyveromyces marxianus IMB3. World J Microbiol Biotechnol 14(6):823–834. https://doi.org/10.1023/A:1008802704374
Solomon BD (2010) Biofuels and sustainability. Ann N Y Acad Sci 1185(1):119–134. https://doi.org/10.1111/j.1749-6632.2009.05279.x.
Sopandi T, Wardah A (2017) Improving ethanol production by co-culturing of Saccharomyces cerevisiae with Candida tropicalis from rice husk hydrolysate media. Afr J Microbiol Res 11(3):65–74. https://doi.org/10.5897/AJMR2016.8375.
Stanley D, Bandara A, Fraser S, Chambers P, Stanley GA (2010) The ethanol stress response and ethanol tolerance of Saccharomyces cerevisiae. J Appl Microbiol 109(1):13–24. https://doi.org/10.1111/j.1365-2672.2009.04657.x
Techaparin A, Thanonkeo P, Klanrit P (2017) High-temperature ethanol production using thermotolerant yeast newly isolated from Greater Mekong Subregion. Braz J Microbiol 48(3):461–475. https://doi.org/10.1016/j.bjm.2017.01.006
Thammasittirong SN-R, Thirasaktana T, Thammasittirong A, Srisodsuk M (2013) Improvement of ethanol production by ethanol-tolerant Saccharomyces cerevisiae UVNR56. SpringerPlus 2(1):1–5. https://doi.org/10.1186/2193-1801-2-583
Tikka C, Osuru HP, Atluri N, Raghavulu PCV (2013) Isolation and characterization of ethanol tolerant yeast strains. Bioinformation 9(8):421. https://doi.org/10.6026/97320630009421.
Tofighi A, Assadi MM, Asadirad MHA, Karizi SZ (2014) Bio-ethanol production by a novel autochthonous thermo-tolerant yeast isolated from wastewater. J Environ Health Sci Eng 12(1):1–6. https://doi.org/10.1186/2052-336X-12-107.
Van der Walt J (1984) Methods for the isolation, maintenance, classification and identification of yeasts. In: The yeasts, a taxonomic study, pp 45–105
Vilgalys R, Hester M (1990) Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J Bacteriol 172(8):4238–4246. https://doi.org/10.1128/jb.172.8.4238-4246.1990
Acknowledgements
We are grateful to the Fermentation Laboratory, Department of Biotechnology, Addis Ababa Science and Technology University, Ethiopia for supporting the equipment and facilities for this research. The authors are thankful to the Westerdijk Fungal Biodiversity Institute (WI), the Netherlands for the molecular identification of the yeast isolates.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
Estifanos Hawaz, Mesfin Tafesse, Anteneh Tesfaye, Dereje Beyene, Solomon Kiros, Alene Admas, Ayantu Degefe, Sissy Degu, and Diriba Muleta did the proposal write-up and sample collection. Estifanos Hawaz, did fermentation dynamics data analysis, and manuscript preparation. Estifanos Hawaz, Teun Boekhout, and Bart Theelen did molecular identification of the yeast isolates. Estifanos Hawaz and Bart Theelen did sequence data analysis. Diriba Muleta, Anteneh Tesfaya, Solomon Kiros, Marzieth Groenewald, and Teun Boekhout did manuscript edition. Marzieth Groenewald did yeast culture collection and preservation. The author(s) read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors have read and agreed on the final version of the manuscript for submission.
Competing interests
The authors declare that they have no competing interests regarding the publication of this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hawaz, E., Tafesse, M., Tesfaye, A. et al. Isolation and characterization of bioethanol producing wild yeasts from bio-wastes and co-products of sugar factories. Ann Microbiol 72, 39 (2022). https://doi.org/10.1186/s13213-022-01695-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13213-022-01695-3