Abstract
Background
Cancer chemotherapy is still hampered by clinical failures due to multi-drug resistance (MDR) of tumor cells. In the present study, we have investigated the cytotoxicity of 20 methanol extracts from 10 medicinal plants against the sensitive leukemia CCRF-CEM cells. The most cytotoxic extracts were then further tested on a panel of 8 human cancer cell lines, including various MDR phenotypes.
Methods
The cytotoxicity of the 20 methanol extracts from 10 Cameroonian medicinal plants was determined using a resazurin reduction assay. Meanwhile, flow cytometry was used to measure cell cycle, apoptosis, mitochondrial membrane potential (MMP), and reactive oxygen species (ROS).
Results
In the preliminary assay using CCRF-CEM cells, 12 extracts from five plants displayed IC50 values below 80 μg/mL, namely Albizia adianthifolia, Alchornea cordifolia, Alchornea laxiflora, Pennisetum purpureum, and Spathodea campanulata. the four best extracts were from two plants: Albizia adianthifolia roots (AAR) and bark (AAB) as well as Alchornea cordifolia leaves (ACL) and bark (ACB) had respective IC50 values of 0.98 μg/mL, 1.45 μg/mL, 8.02 μg/mL and 12.57 μg/mL in CCRF-CEM cells. They were further tested in 8 other cell lines as well as in normal AML12 hepatocytes. IC50 values ranging from 2.71 μg/mL (towards glioblastoma U87MG.ΔEGFR cells) to 10.30 μg/mL (towards breast adenocarcinoma MDA-MB-231-BCRP cells) for AAB, from 3.43 μg/mL (towards U87MG cells) to 10.77 μg/mL (towards colon carcinoma HCT116 (p53−/−) cells) for AAR and from 0.11 μg/mL (towards CCRF-CEM cells) to 108 μg/mL (towards leukemia CEM/ADR5000 cells) for doxorubicin (as control drug) were obtained. ACL and ACB extracts displayed selective activities. AAR and ACL extracts induced apoptosis in CCRF-CEM cells, through caspases activation and loss of MMP, while apoptotic cell death was mediated by MMP diruption and increase ROS production for ACL.
Conclusion
Some of the tested plants namely Albizia adianthifolia, Alchornea cordifolia, Alchornea laxiflora, Pennisetum purpureum, Spathodea campanulata represent a potential source of novel anticancer drugs. Especially, Albizia adianthifolia and Alchornea cordifolia revealed considerable cytotoxic activities that could be exploited to develop phytomedicines to fight cancers including MDR phenotypes.
Similar content being viewed by others
Background
Malignant diseases are amongst the major causes of death worldwide with a growing burden and low survival rates in developing countries [1]. Clinically, chemotherapy is still hampered by treatment failures due to multidrug resistance (MDR) of cancer cells. Anticancer drug development should take into account the fact that cancer cells rapidly acquire resistance [2–5]. Natural ressources such as medicinal plants constitute an indeniable reservoir of antiproliferative compounds [6]. Hence, fighting cancers and mostly drug-resistant phenotypes with phytochemical represents a very promising alternative, especially regarding the diversity of plant’s secondary metabolites. In the past, several bioactive compounds belonging to several classes of secondary metabolites isolated from African plants showed considerable antiproliferative activity against MDR cancer cells. Some of these molecules include benzophenones (2,2′,5,6′-tetrahydroxybenzophenone, guttiferone E, isogarcinol and isoxanthochymo) [7], xanthones (xanthone V1, quinones: 2-acetylfuro-1,4-naphthoquinone) [8], flavonoids (gancaonin Q, 4-hydroxylonchocarpin, 6-prenylapigenin, 6,8-diprenyleriodictyol [9], 2′,4′-dihydroxy-3′,6′-dimethoxychalcone, 4′-hydroxy-2′,6′-dimethoxychalcone, cardamomin [10, 11], 8-hydroxycudraxanthone G, morusignin I and cudraxanthone I [12] and alkaloids (isotetrandrine [13], montrofoline, 1-hydroxy-4-methoxy-10-methylacridone, norevoxanthine, evoxanthine and 1,3-dimethoxy-10-methylacridone) [14]. Moreover, several African medicinal plants previously displayed good cytotoxicity towards drug-sensitive and drug-resistant cancer cell lines. These plants include Echinops giganteus, Xylopia aethiopica, Piper capense, Imperata cylindrica [15, 16], Beilschmiedia acuta, Clausena anisata, Fagara tessmannii, Newbouldia laevis, Polyscias fulva [17], Garcina lucida, Fagara heitzii, Hymenocardia lyrata [18], Gladiolus quartinianus, Vepris soyauxii and Anonidium mannii [19].
In our ongoing search of anticancer drugs from African medicinal plants, we undertook the present work to assess the cytotoxicity of 10 Cameroonian medicinal plants traditionally used to manage cancer or disease states bearing relevance to cancer or cancer-like symptoms, such as immune and skin disorders, inflammatory, infectious, parasitic and viral diseases [15]. The study was extended to the evaluation of the ability of extracts from two most active plants, Albizia adianthifolia and Alchornea cordifolia to alter the cell cycle distribution, caspases activity, mitochondrial membrane potential (MMP) and to increase reactive oxygen species (ROS) in leukemia CCRF-CEM cells.
Methods
Plant material and extraction
All medicinal plants parts used in the present study were collected in different regions of Cameroon in January 2014. These included leaves, bark and roots of Alchornea cordifolia, Alchornea laxiflora, Albizia adianthifolia and Spathodea campanulata, leaves and roots of Combretum hispidum and Laportea ovalifolia and the whole plant of Boerhavia diffusa, Eremomastax speciosa, Laportea aestuans and Pennisetum purpureum. The plants were identified at the National Herbarium (Yaoundé, Cameroon), where voucher specimens were deposited under the reference numbers shown in Table 1. The air-dried and powdered plant material was soaked in methanol for 48 h, at room temperature. The methanol extract was concentrated in vacuum under reduced pressure to give the crude extract. This extract was then conserved at 4 °C until further use.
Chemicals
Doxorubicin 98.0 % and vinblastine ≥ 96 % from Sigma-Aldrich (Munich, Germany) were provided by the University Pharmacy of the Johannes Gutenberg University (Mainz, Germany) and dissolved in phosphate buffer saline (PBS; Invitrogen, Eggenstein, Germany) at a concentration of 10 mM. Geneticin > 98 % (Sigma-Aldrich) was stored at a stock concentration of 72.18 mM.
Cell cultures
The cell lines used in the present study, their origins and their treatments were previously reported. They include drug-sensitive leukemia CCRF-CEM and multidrug-resistant P-glycoprotein-over-expressing subline CEM/ADR5000 cells [4, 20, 21], breast cancer MDA-MB-231-pcDNA3 cells and its resistant subline MDA-MB-231-BCRP clone 23 [22], colon cancer HCT116 (p53+/+) cells and its knockout clone HCT116 (p53−/−), glioblastoma U87MG cells and its resistant subline U87MG.ΔEGFR [7, 12, 16]. Leukemia CCRF-CEM and CEM/ADR5000 cells were cultured in RPMI 1640 medium (Invitrogen) supplemented with 10 % fetal calf serum in a humidified 5 % CO2 atmosphere at 37 °C. This medium was also used for the cytotoxicity test with these two cell lines. MDA-MB-231-BCRP as well as U87MG.ΔEGFR and HCT116 (p53−/−) were maintained in DMEM medium containing 10 % FBS (Invitrogen) and 1 % penicillin (100 U/mL)-streptomycin (100 μg/mL) (Invitrogen) and were continuously treated with 800 ng/mL and 400 μg/mL geneticin, respectively. The cytotoxicity of all carcinoma cells was performed in DMEM medium containing 10 % FBS (Invitrogen) and 1 % penicillin-streptomycin.
Resazurin reduction assay
The cytotoxicity of the tested samples was performed by resazurin reduction assay as previously described [7, 23]. The assay is based on reduction of the indicator dye, resazurin, to the highly fluorescent resorufin by viable cells. Non-viable cells rapidly lose the metabolic capacity to reduce resazurin and thus produce no fluorescent signal. Briefly, adherent cells were detached by treatment with 0.25 % trypsin/EDTA (Invitrogen) and an aliquot of 1x104 cells was placed in each well of a 96-well cell culture plate (Thermo Scientific, Germany) in a total volume of 200 μL. Cells were allowed to attach overnight and then were treated with different concentrations of the studied sample. For suspension cells, aliquots of 104 cells per well were seeded in 96-well-plates in a total volume of 100 μL. The studied sample was immediately added in varying concentrations in an additional 100 μL of culture medium to obtain a total volume of 200 μL/well. After 24 h or 48 h, 20 μL resazurin (Sigma-Aldrich, Germany) 0.01 % w/v in ddH2O was added to each well and the plates were incubated at 37 °C for 4 h. Fluorescence was measured on an Infinite M2000 Pro™ plate reader (Tecan, Germany) using an excitation wavelength of 544 nm and an emission wavelength of 590 nm. Each assay was done at least two times, with six replicates each. The viability was evaluated based on a comparison with untreated cells. IC50 values represent the sample’s concentrations required to inhibit 50 % of cell proliferation and were calculated from a calibration curve by linear regression using Microsoft Excel [8, 11]. The contribution of the extracts (at various tested concentrations) to the fluorescence has been determined, both in the absence and presence of resazurin, prior to any cell studies. In a preliminary study, all samples were tested against the sensitive CCRF-CEM cells at various concentrations ranging from 0.63 to 80 μg/mL (crude extracts) or 0.08 to 10 μg/mL (doxorubicin), and samples displaying IC50 values below 20 μg/mL were further investigated in 8 other tumor cell lines as well as in normal AML12 hepatocytes. Doxorubicin was used as positive control, while dimethylsulfoxide (DMSO) used to dissolve the samples was used as negative control. The highest concentration of DMSO was less than 0.4 %.
Flow cytometry for cell cycle analysis and detection of apoptotic cells
Extracts from Albizia adianthifolia roots (AAR) and Alchornea cordifolia leaves (ACL) that displayed the best cytotoxicity as well as doxorubicin were used to treat CCRF-CEM cells (1 × 106) at their IC50 values. The cell cycle was then analyzed after incubation for 24 h, 48 h and 72 h. All reagents, experimental conditions and apparatus were identical to those previously reported [12, 16]. Briefly, cell cycle analysis was performed by flow cytometry using Vybrant® DyeCycle™ (Invitrogen, Darmstadt, Germany). Cells were measured after Vybrant® DyeCycle™ Violet staining (30 min at 37 °C) on a LSR-Fortessa FACS analyzer (Becton-Dickinson, Heidelberg, Germany) using the violet laser. Vybrant® DyeCycle™ Violet stain was measured with 440 nm excitation. Cytographs were analyzed using FlowJo software (Celeza, Switzerland). All experiments were performed at least in triplicate.
Caspase-Glo 3/7, caspase-Glo 8 and caspase-Glo 9 assay
Caspase activity in CCRF-CEM cells treated with AAR and ACL was detected using Caspase-Glo 3/7, Caspase-Glo 8 and Caspase-Glo 9 Assay kits (Promega, Mannheim, Germany) as previously described [24].
Analysis of mitochondrial membrane potential (MMP)
CCRF-CEM cells were treated with AAR, ACL or vinblastine. The MMP was analyzed using 5,5′,6,6′-tetrachloro-1,1′,3,3′-tetraethylbenzimidazolylcarbocyanine iodide) (JC-1; Biomol, Hamburg, Germany) staining as previously described [19]. Cells were measured in a LSR-Fortessa FACS analyzer (Becton-Dickinson). The JC-1 signal was measured at an excitation of 561 nm (150 mW) and detected using a 586/15 nm band-pass filter. The signal was analyzed at 640 nm excitation (40 mW) and detected using a 730/45 nm bandpass filter. Cytographs were analyzed using FlowJo software (Celeza, Olten, Switzerland). All experiments were performed at least in triplicate.
Measurement of reactive oxygen species (ROS) by flow cytometry
The 2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA) (Sigma-Aldrich, Schnelldorf, Germany) was used for the detection of ROS in CCRF-CEM cells treated with AAR, ACL, DMSO (solvent control), or hydrogen peroxide (H2O2; positive control) as previously described [17, 25, 26]. Cells were measured in a FACS Calibur flow cytometer (Becton-Dickinson). Dichlorofluorescein (DCF) was measured at 488 nm excitation (25 mW) and detected using a 530/30 nm band-pass filter. Cytographs were analyzed using FlowJo software (Celeza). All experiments were performed at least in triplicate.
Statistical analysis
Statistical analysis of all data was performed using a Student’s t-test or Kruskal–Wallis test followed by Dunn’s post-hoc multiple comparison test (Graph-Pad Prism 5.01; GraphPad Software, Inc., CA, USA). P < 0.05 denoted significance in all cases.
Results
In the present investigation, the cytotoxicity of 20 methanol extracts from 10 plants was first determined at different concentrations in drug-sensitive CCRF-CEM leukemia cells. The results are summarized in Table 2. Twelve out of 20 (60 %) extracts displayed IC50 values below 80 μg/mL. These extracts were from Pennisetum purpureum, Spathodea campanulata bark, Spathodea campanulata roots, Alchornea laxiflora bark, Alchornea laxiflora leaves, Albizia adianthifolia leaves (AAL), Combretum hispidum leaves, Alchornea cordifolia roots (ACR), Alchornea cordifolia bark (ACB), Alchornea cordifolia leaves (ACL), Albizia adianthifolia bark (AAB) and Albizia adianthifolia roots (AAR). Extracts from Alchornea laxiflora roots, Boerhavia diffusa (whole plant), Combretum hispidum bark, Eremomastax speciosa (whole plant), Laportea aestuans (whole plant), Laportea ovalifolia leaves, Laportea ovalifolia roots, Spathodea campanulata leaves resulted in more than 50 % proliferation of CCRF-CEM cells at 80 μg/mL (Table 2). Four extracts from two plants including ACB (IC50 value of 12.57 μg/mL), ACL (IC50: 8.02 μg/mL), AAB (IC50: 1.45 μg/mL) and AAR (IC50: 0.98 μg/mL) as well as doxorubicin (IC50: 0.11 μg/mL) displayed IC50 values below 20 μg/mL in CCRF-CEM cells (Table 2). These extracts were further selected for IC50 determination towards a panel of sensitive and MDR cell lines. The results summarized in Table 2 revealed IC50 values ranging from 2.71 μg/mL (towards glioblastoma U87MG.ΔEGFR cells) to 10.30 μg/mL (towards breast adenocarcinoma MDA-MB-231-BCRP cells) for AAB, from 3.43 μg/mL (towards U87MG cells) to 10.77 μg/mL (towards resistant colon carcinoma HCT116 (p53−/−) cells) for AAR and from 0.11 μg/mL (towards CCRF-CEM cells) to 108 μg/mL (towards P-glycoprotein-over-expressing CEM/ADR5000 cells) for doxorubicin on the 8 other cancer cell lines studied. Extracts from Alchornea cordifolia, ACL and ACB displayed selective activities. However, ACL and ACB were also less toxic towards normal AML12 hepatocytes, with IC50 values above 80 μg/mL contrary to AAB (IC50: 29.18 μg/mL) and AAR (IC50: 29.14 μg/mL). It is worth noting that collateral sensitivity (or hypersensitivity: higher toxicity to resistant than to sensitive cells with a degree of resistance below 1) was observed in drug-resistant epidermal growth factor receptor-transfected U87MG.ΔEGFR cells to AAB (degree of resistance of 0.43-fold), to AAR (0.39-fold), to ACL (0.83-fold) and to ACB (<0.40-fold) compared to its sensitive counterpart U87MG cells. Importantly, if cross-resistance to the tested extracts were observed, the degrees of resistance were in all cases lower than that of the reference compound, doxorubicin (Table 3). AAR and ACL were the most active extracts from Albizia adiathifolia and Alchornea cordifolia respectively, and were subsequently used for mechanistic studies.
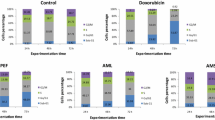
IC50 values of AAR and ACL extracts as well as doxorubicin were used to treat CCRF-CEM cells for 6 h, and the cycle distribution was analyzed. The results are depicted in Fig. 1. Dose-dependent and significant modifications of the cell cycle phases were observed. Both AAR and ACL induced cell cycle arrest in the G0/G1 phase. After treatment with these two extracts, CCRF-CEM cells underwent apoptosis with a dose-dependent increase in the sub-G0/G1 phase. The percentages of cells in the sub-G0/G1 phase varied from 32.14 % (in 24 h) to 57.99 % (72 h) and from 31.69 % (24 h) to 59.67 % (72 h), respectively, for AAR and ACL treatments, while doxorubicin increased apoptosis in a range of 6.02 % (24 h) to 51.87 % (72 h). The highest percentage of sub-G0/G1 phase in non-treated cells was only 6.42 % after 72 h. After treating CCRF-CEM cells for 6 h at 2-fold IC50, AAR induced 4.35-fold, 2.02-fold and 1.52-fold increase of caspase 3/7, caspase 9 and caspase 8 activities, respectively, whereas no changes were observed upon ACL treatment (Fig. 2). AAR also induced significant MMP loss in a range of 35.5 % (1/2-fold IC50 treatment) to 87.6 % (2-fold IC50) (Fig. 3). ACL caused up 41.7 % MMP loss at 1/2-fold IC50 treatment and complete rupture of the membrane (99.7 %) at 2-fold IC50 (Fig. 3). A 48.6 % loss of MMP at 2-fold IC50 of vinblastine was previously reported under similar experimental conditions in CCRF-CEM cells [12]. AAR did not induce ROS generation in CCRF-CEM cells contrary to ACL (Fig. 4). Dose-dependant increase in ROS production was also observed upon treatment of cells with ACL in a range of 0.73 % (1/2-fold IC50 treatment) to 33.6 % (2-fold IC50).
Cell cycle distribution of CCRF-CEM leukemia cells treated with extracts from Albizia adianthifolia roots (AAR), Alchornea cordifolia leaves (ACL) or doxorubicin (control drug) at different time. AAR and ACL were tested at 0.98 μg/mL and 8.02 μg/mL respectively, while doxorubicin was tested at 0.11 μg/mL corresponding to their IC50 values. At each experimental time (24 h, 48 h, 72 h), data with different superscript letters are significantly different (P < 0.05)
Enzymatic activity of caspase 3/7, caspase 8 and caspase 9 after 6 h treatment of CCRF-CEM cells with AAR and ACL extracts. The activity of caspase is expressed as percentage % relative to untreated cells. IC50: 0.98 μg/mL and 8.02 μg/mL for AAR and ACL extracts, respectively. Data with different superscript letters are significantly different (P < 0.05)
Effect of AAR and ACL extracts on the mitochondrial membrane potential in CCRF-CEM cells. C: control; AAR was tested at 24 h at 0.49 μg/mL (AAR1), 0.98 μg/mL (AAR2), and 1.96 μg/mL (AAR3), while ACL was tested at 4.01 μg/mL (ACL1), 8.02 μg/mL (ACL2), and 16.04 μg/mL (ACL3) corresponding to 1/2-fold, IC50 and 2-fold IC50 values. Data for the positive control, vinblastine under similar experimental conditions were previously reported [7]. Loss of MMP (Q1), intact cells (Q2), ruptured cell membrane (Q3 and Q4)
ROS generation in CCRF-CEM cells after treatment with AAR and ACL extracts for 24 h. C: control; AAR was tested at 24 h at 0.49 μg/mL (AAR1), 0.98 μg/mL (AAR2), and 1.96 μg/mL (AAR3), while ACL was tested at 4.01 μg/mL (ACL1), 8.02 μg/mL (ACL2), and 16.04 μg/mL (ACL3) corresponding to 1/2-fold, IC50 and 2-fold IC50 values
Discussion
The development of resistance by malignant cells remains a serious issue in cancer chemotherapy. Cancer cells rapidly develop chemoresistance, mainly due to the presence of adenosine triphosphate-binding cassette (ABC) transporters [2–4], such as the breast cancer resistance protein (BCRP/ABCG2) and P-glycoprotein (P-gp/MDR1/ABCB1) [2] as well as the oncogene epidermal growth factor receptor (EGFR) [3, 4, 27] and the deletion or inactivation of tumor suppressor gene p53 [5]. Hence, identifying the mechanisms of resistance to different drugs is necessary, in order to efficiently prevent and overcome drug resistance. In this study, multi-factorial drug-resistant cancer cell lines such as leukemia CEM/ADR5000 cells over-expressing P-glycoprotein, breast adenocarcinoma MDA-MB-231-BCRP clone 23 expressing BCRP, p53 knockout HCT116 (p53−/−) colon cancer cells and EGFR-transfected U87MG.ΔEGFR glioblastoma cells [4, 7, 12, 20–22, 28] were used to determine to assess the cytotoxicity the selected plant extracts. According to the US NCI plant screening program, botanicals with IC50 values below of 20 μg/mL following incubation between 48 and 72 h [29] have been recognized as potential cytotoxic substances. In preliminary assays using the sensitive leukemia CCRF-CEM cells, AAB, AAR, ACL and ACB (Table 2) displayed IC50 values below 20 μg/mL and were therefore selected for further assays against MDR phenotypes of other cell lines. Interestingly, AAB and AAR also displayed IC50 values below or around 10 μg/mL and could therefore be considered as potential source for novel anti-cancer drugs. Most importantly, the degree of resistance of cells lines to AAB and AAR were in all cases lower than that of doxorubicin, highlighting their potential to combat MDR phenotypes. Though the IC50 values recorded with ACL and ACB were all above 20 μg/mL, the cytotoxicity of these two samples on malignant cells can still be considered interesting, as they were much less toxic on normal AML12 hepatocytes, highlighting their good selectivity. It is also worth to note that the two best extracts, AAB and AAR were slightly toxic to normal AML12 hepatocytes (IC50 values of 29.18 μg/mL and 29.14 μg/mL respectively for AAB and AAR). However, their high cytotoxicity towards cancer cells also suggests that they might be safely used in cancer chemotherapy. However, further evidence of the clinical efficacy of these extracts will be needed, as many phytochemicals are poorly bioavailable and they may be metabolized to more or less potent compounds by gut bacterial metabolism. MMP loss and increased ROS have been reported as a mode of apoptosis induction of plant extracts [29]. Hence, the ability of AAR and ACL to cause MMP breakdown in CCRF-CEM cells fits to this theory. The mode of action of AAR also includes the activation of caspases. Initiator caspases 9 (2.02-fold) and effector caspases 3/7 (4.35-fold) (Fig. 2) were significantly activated [29]. In addition to MMP alterations, ACL-induced apoptosis also include ROS production (Fig. 4).
To the best of our knowledge, the cytotoxicty of Albizia adiathifolia and Alchornea cordifolia towards the cell line panel tested in this study is being reported for the first time. Triterpenoid saponins such as adianthifoliosides A, B, and D isolated from Albizia adianthifolia exhibited cytotoxic effects towards Jurkat leukemia cells [30]. The presence of these compounds as well as other cytotoxic constituents such as prosapogenins [31] and aurantiamide acetate [32] found in Albizia adianthifolia could explain the antiproliferative effects of this plant.
Conclusion
Twelve extracts from 5 medicinal plants (Albizia adianthifolia, Alchornea cordifolia, Alchornea laxiflora, Pennisetum purpureum, and Spathodea campanulata) displayed cytotoxicity against CCRF-CEM leukemia cells. They may represent a source for the development of novel anticancer drugs. Furthermore, Albizia adianthifolia and Alchornea cordifolia further displayed considerable cytotoxicity against MDR phenotypes in a panel of 8 other cancer cell lines. They may therefore be exploited to develop phytomedicine to fight cancers with various MDR phenotypes. AAR and AAB were the most cytotoxic extracts and the mechanism of AAR-induction apoptosis in CCRF-CEM cells included caspases activation and MMP loss. The mode of apotosis induction by ACL extract included MMP disruption and increased ROS generation in CCRF-CEM cells. The cytotoxicty of the two best plants, Albizia adiathifolia and Alchornea cordifolia towards the cell line panel tested in this study is being reported for the first time. Their purification will further be performed to identify their active constituents.
Abbreviations
AAB, Albizia adianthifolia bark; AAL, Albizia adianthifolia leaves; AAR, Albizia adianthifolia roots; ABC, adenosine triphosphate-binding cassette; ACB, Alchornea cordifolia bark; ACL, Alchornea cordifolia leaves; ACR, Alchornea cordifolia roots; BCRP, breast cancer resistance protein; DCF, dichlorofluorescein; DCFH-DA, 2′,7′-Dichlorodihydrofluorescein diacetate; DMSO, dimethylsufoxide; EGFR, epidermal growth factor receptor; IC50, inhibitory concentration 50 %; JC-1, 5,5′,6,6′-tetrachloro-1,1′,3,3′-tetraethylbenzimidazolylcarbocyanine iodide; MDR, multi-drug resistant; MMP, mitochondrial membrane potential; PBS, phosphate buffer saline; ROS, reactive oxygen species
References
Vorobiof DA, Abratt R. The cancer burden in Africa. S Afr Med J. 2007;97:937–9.
Shen B, Li D, Dong P, Gao S. Expression of ABC transporters is an unfavorable prognostic factor in laryngeal squamous cell carcinoma. Ann Otol Rhinol Laryngol. 2011;120:820–7.
Biedler JL, Spengler BA. Reverse transformation of multidrug-resistant cells. Cancer Metastasis Rev. 1994;13:191–207.
Efferth T, Sauerbrey A, Olbrich A, Gebhart E, Rauch P, Weber HO, et al. Molecular modes of action of artesunate in tumor cell lines. Mol Pharmacol. 2003;64:382–94.
El-Deiry WS. Role of oncogenes in resistance and killing by cancer therapeutic agents. Curr Opin Oncol. 1997;9:79–87.
Kuete V, Efferth T. Pharmacogenomics of Cameroonian traditional herbal medicine for cancer therapy. J Ethnopharmacol. 2011;137:752–66.
Kuete V, Tchakam PD, Wiench B, Ngameni B, Wabo HK, Tala MF, et al. Cytotoxicity and modes of action of four naturally occuring benzophenones: 2,2′,5,6′-tetrahydroxybenzophenone, guttiferone E, isogarcinol and isoxanthochymol. Phytomedicine. 2013;20:528–36.
Kuete V, Wabo HK, Eyong KO, Feussi MT, Wiench B, Krusche B, et al. Anticancer activities of six selected natural compounds of some Cameroonian medicinal plants. PLoS One. 2011;6(8):e21762.
Kuete V, Ngameni B, Wiench B, Krusche B, Horwedel C, Ngadjui BT, et al. Cytotoxicity and mode of action of four naturally occuring flavonoids from the genus Dorstenia: gancaonin Q, 4-hydroxylonchocarpin, 6-prenylapigenin, and 6,8-diprenyleriodictyol. Planta Med. 2011;77:1984–9.
Kuete V, Nkuete AHL, Mbaveng AT, Wiench B, Wabo HK, Tane P, et al. Cytotoxicity and modes of action of 4′-hydroxy-2′,6′-dimethoxychalcone and other flavonoids toward drug-sensitive and multidrug-resistant cancer cell lines. Phytomedicine. 2014;21:1651–7.
Dzoyem JP, Nkuete AH, Kuete V, Tala MF, Wabo HK, Guru SK, et al. Cytotoxicity and antimicrobial activity of the methanol extract and compounds from Polygonum limbatum. Planta Med. 2012;78:787–92.
Kuete V, Sandjo L, Nantchouang Ouete J, Fouotsa H, Wiench B, Efferth T. Cytotoxicity and modes of action of three naturally occuring xanthones (8-hydroxycudraxanthone G, morusignin I and cudraxanthone I) against sensitive and multidrug-resistant cancer cell lines. Phytomedicine. 2013;21:315–22.
Kuete V, Sandjo LP, Mbaveng AT, Zeino M, Efferth T. Cytotoxicity of compounds from Xylopia aethiopica towards multi-factorial drug-resistant cancer cells. Phytomedicine. 2015;22:1247–54.
Kuete V, Fouotsa H, Mbaveng AT, Wiench B, Nkengfack AE, Efferth T. Cytotoxicity of a naturally occurring furoquinoline alkaloid and four acridone alkaloids towards multi-factorial drug-resistant cancer cells. Phytomedicine. 2015;22:946–51.
Kuete V, Krusche B, Youns M, Voukeng I, Fankam AG, Tankeo S, et al. Cytotoxicity of some Cameroonian spices and selected medicinal plant extracts. J Ethnopharmacol. 2011;134:803–12.
Kuete V, Sandjo LP, Wiench B, Efferth T. Cytotoxicity and modes of action of four Cameroonian dietary spices ethno-medically used to treat Cancers: Echinops giganteus, Xylopia aethiopica, Imperata cylindrica and Piper capense. J Ethnopharmacol. 2013;149:245–53.
Kuete V, Tankeo SB, Saeed ME, Wiench B, Tane P, Efferth T. Cytotoxicity and modes of action of five Cameroonian medicinal plants against multi-factorial drug resistance of tumor cells. J Ethnopharmacol. 2014;153:207–19.
Dzoyem J, Guru S, Pieme C, Kuete V, Sharma A, Khan I, et al. Cytotoxic and antimicrobial activity of selected Cameroonian edible plants. BMC Complement Altern Med. 2013;13:78.
Kuete V, Fankam AG, Wiench B, Efferth T. Cytotoxicity and modes of action of the methanol extracts of six Cameroonian medicinal plants against multidrug-mesistant tumor cells. Evid Based Complement Alternat Med. 2013;2013:285903.
Kimmig A, Gekeler V, Neumann M, Frese G, Handgretinger R, Kardos G, et al. Susceptibility of multidrug-resistant human leukemia cell lines to human interleukin 2-activated killer cells. Cancer Res. 1990;50:6793–9.
Gillet J, Efferth T, Steinbach D, Hamels J, de Longueville F, Bertholet V, et al. Microarray-based detection of multidrug resistance in human tumor cells by expression profiling of ATP-binding cassette transporter genes. Cancer Res. 2004;64:8987–93.
Doyle LA, Yang W, Abruzzo LV, Krogmann T, Gao Y, Rishi AK, et al. A multidrug resistance transporter from human MCF-7 breast cancer cells. Proc Natl Acad Sci U S A. 1998;95:15665–70.
O’Brien J, Wilson I, Orton T, Pognan F. Investigation of the Alamar Blue (resazurin) fluorescent dye for the assessment of mammalian cell cytotoxicity. Eur J Biochem. 2000;267:5421–6.
Kuete V, Sandjo LP, Djeussi DE, Zeino M, Kwamou GM, Ngadjui B, et al. Cytotoxic flavonoids and isoflavonoids from Erythrina sigmoidea towards multi-factorial drug resistant cancer cells. Invest New Drugs. 2014;32:1053–62.
Bass DA, Parce JW, Dechatelet LR, Szejda P, Seeds MC, Thomas M. Flow cytometric studies of oxidative product formation by neutrophils: a graded response to membrane stimulation. J Immunol. 1983;130:1910–7.
Cossarizza A, Ferraresi R, Troiano L, Roat E, Gibellini L, Bertoncelli L, et al. Simultaneous analysis of reactive oxygen species and reduced glutathione content in living cells by polychromatic flow cytometry. Nat Protoc. 2009;4:1790–7.
Efferth T, Sauerbrey A, Halatsch ME, Ross DD, Gebhart E. Molecular modes of action of cephalotaxine and homoharringtonine from the coniferous tree Cephalotaxus hainanensis in human tumor cell lines. Naunyn Schmiedebergs Arch Pharmacol. 2003;367:56–67.
Kuete V, Mbaveng AT, Zeino M, Fozing CD, Ngameni B, Kapche GD, et al. Cytotoxicity of three naturally occurring flavonoid derived compounds (artocarpesin, cycloartocarpesin and isobavachalcone) towards multi-factorial drug-resistant cancer cells. Phytomedicine. 2015;22:1096–102.
Kuete V, Efferth T. African flora has the potential to fight multidrug resistance of cancer. BioMed Res Int. 2015;2015:914813.
Haddad M, Laurens V, Lacaille-Dubois MA. Induction of apoptosis in a leukemia cell line by triterpene saponins from Albizia adianthifolia. Bioorg Med Chem. 2004;12:4725–34.
Haddad M, Khan IA, Lacaille-Dubois MA. Two new prosapogenins from Albizia adianthifolia. Pharmazie. 2002;57:705–8.
Tamokou Jde D, Simo Mpetga DJ, Keilah Lunga P, Tene M, Tane P, Kuiate JR. Antioxidant and antimicrobial activities of ethyl acetate extract, fractions and compounds from stem bark of Albizia adianthifolia (Mimosoideae). BMC Complement Altern Med. 2012;12:99.
Watt J, Breyer-Brandwyk M. The medicinal and poisonous plants of Southern and Easthern Africa. 2nd ed. London: Livingstone; 1962.
Van-Wyk B, Gerick N. People’s plants: a guide to useful plants of Southern Africa. Pretoria: Briza publications; 2000.
Haddad M, Miyamoto T, Laurens V, Lacaille-Dubois MA. Two new biologically active triterpenoidal saponins acylated with salicylic acid from Albizia adianthifolia. J Nat Prod. 2003;66:372–7.
Tchinda CF, Voukeng KI, Beng VP, Kuete V. Antibacterial activities of the methanol extracts of Albizia adianthifolia, Alchornea laxiflora, Laportea ovalifolia and three other Cameroonian plants against multi-drug resistant Gram-negative bacteria. Saudi J Biol Sci. 2016. doi:10.1016/j.sjbs.2016.01.033.
Beppe GJ, Dongmo AB, Foyet HS, Tsabang N, Olteanu Z, Cioanca O, et al. Memory-enhancing activities of the aqueous extract of Albizia adianthifolia leaves in the 6-hydroxydopamine-lesion rodent model of Parkinson’s disease. BMC Complement Altern Med. 2014;14:142.
Ogungbamila FO, Samuelsson G. Smooth muscle relaxing flavonoids from Alchornea cordifolia. Acta Pharm Nord. 1990;2:421–2.
Adeneye AA, Oreagba AI, Ishola IO, Kalejaiye HA. Evaluation of the anti-arthritic activity of the hydroethanolic leaf extract of Alchornea cordifolia in rats. Afr J Tradit Complement Altern Med. 2014;11:402–10.
Manga HM, Brkic D, Marie DE, Quetin-Leclercq J. In vivo anti-inflammatory activity of Alchornea cordifolia (Schumach. & Thonn.) Mull. Arg. (Euphorbiaceae). J Ethnopharmacol. 2004;92:209–14.
Okeke IN, Ogundaini AO, Ogungbamila FO, Lamikanra A. Antimicrobial spectrum of Alchornea cordifolia leaf extract. Phytother Res. 1999;13:67–9.
Mambe FT, Voukeng KI, Beng VP, Kuete V. Antibacterial activities of methanol extracts of Alchornea cordifolia and four other Cameroonian plants against MDR phenotypes. J Taibah Univ Med Sci. 2016;11:121–7.
Tona L, Kambu K, Ngimbi N, Mesia K, Penge O, Lusakibanza M, et al. Antiamoebic and spasmolytic activities of extracts from some antidiarrhoeal traditional preparations used in Kinshasa, Congo. Phytomedicine. 2000;7:31–8.
Osadebe PO, Okoye FB. Anti-inflammatory effects of crude methanolic extract and fractions of Alchornea cordifolia leaves. J Ethnopharmacol. 2003;89:19–24.
Ogundipe OO, Moody JO, Houghton PJ, Odelola HA. Bioactive chemical constituents from Alchornea laxiflora (benth) pax and hoffman. J Ethnopharmacol. 2001;74:275–80.
Oladunmoye M, Kehinde F. Ethnobotanical survey of medicinal plants used in treating viral infections among Yoruba tribe of South Western Nigeria. Afr J Microbiol Res. 2011;5:2991–3004.
Akinpelu DA, Abioye EO, Aiyegoro OA, Akinpelu OF, Okoh AI. Evaluation of antibacterial and antifungal properties of Alchornea laxiflora (Benth.) Pax. & Hoffman. Evid Based Complement Alternat Med. 2015;2015:684839.
Farombi EO, Ogundipe OO, Samuel Uhunwangho E, Adeyanju MA, Olarenwaju MJ. Antioxidant properties of extracts from Alchornea laxiflora (Benth) Pax and Hoffman. Phytother Res. 2003;17:713–6.
Kouakou S, Nguessan I, Kablan B. Activité antioxydante et antiélastasique de trois plantes à usage antiasthmatique en médecine traditionnelle. J Sci Pharm Biol. 2009;12:6–12.
Ahmed-Belkacem A, Macalou S, Borrelli F, Capasso R, Fattorusso E, Taglialatela-Scafati O, et al. Nonprenylated rotenoids, a new class of potent breast cancer resistance protein inhibitors. J Med Chem. 2007;50:1933–8.
Olaleye MT, Akinmoladun AC, Ogunboye AA, Akindahunsi AA. Antioxidant activity and hepatoprotective property of leaf extracts of Boerhaavia diffusa Linn against acetaminophen-induced liver damage in rats. Food Chem Toxicol. 2010;48(8–9):2200–5.
Wagh S, Vidhale N. Antimicrobial efficacy of Boerhaavia diffusa against some human pathogenic bacteria and fungi. Biosci Biotechnol Res Asia. 2010;7:267–72.
Burkill H. The useful plants of west tropical Africa. Kew: Edinburgh Royal Botanic Gardens; 1985.
Adjanohoun J, Aboubakar N, Dramane K, Ebot M, Ekpere J, Enow-Orock E, et al., editors. Traditional medicine and pharmacopoeia: contribution to ethnobotanical and floristic studies in Cameroon. Lagos: OUA/STRC; 1996.
Jiofack T, Ayissi I, Fokunang C, Guedje N, Kemeuze V. Ethnobotany and phytomedicine of the upper Nyong valley forest in Cameroon. Afr J Pharm Pharm. 2009;3:144–50.
Schmelzer G, Gurib-Fakim A. Plant resources of tropical Africa, Medicinal plants, vol. 11 (2). Wageningen: PROTA Foundation-CTA; 2013.
Oben J, Assi S, Agbor G, Musoro D. Effect of Eremomastax speciosa on experimantal diarrhoea. Afr J Trad Complement Altern Med. 2006;3:95–100.
Kuete V, Voukeng IK, Tsobou R, Mbaveng AT, Wiench B, Beng VP, et al. Cytotoxicity of Elaoephorbia drupifera and other Cameroonian medicinal plants against drug sensitive and multidrug resistant cancer cells. BMC Complement Altern Med. 2013;13:250.
Okokon J, Antia B, Udoh A, Akpan M. Antianaemic and antimicrobial activity of Eremomastax speciosa. J Pharmacol Toxicol. 2007;2:196–9.
Essiet U, Edet N, Bala D. Phytochemical and physiochemical analysis of the leaves of Laportea aestuans (Linn) Chew and Laportea aestuans (Schumach) Chew (male and female). Asian J Plant Sci Res. 2011;1:35–42.
Oloyede G, Oyelola M. Chrysen-2-ol derivative from west indian wood nettle Laportea aestuans (l.) chew inhibits oxidation and microbial growth in vitro. EXCLI J. 2013;12:894–906.
Okereke S, Elekwa I, Nmaju A. Gas chromatographic fid, hypoglycemic and hypolipidemic effects of leaves of Laportea aestuans in alloxan induced diabetes in male albino rats. IOSR J Environ Sci Toxicol Food Technol. 2014;42:42–6.
Oloyede GK, Ayanbadejo OE. Phytochemical, toxicity, antimicrobial and antioxidant screening of extracts obtained from Laportea aestuans (Gaud). J Med Sci. 2014;14:51–9.
Focho D, WT N, Fonge B. Medicinal plants of Aguambu - Bamumbu in the Lebialem highlands, southwest province of Cameroon. Afr J Pharm Pharmacol. 2009;3:1–13.
Momo CE, Oben JE, Tazoo D, Dongo E. Antidiabetic and hypolipidaemic effects of a methanol/methylene-chloride extract of Laportea ovalifolia (Urticaceae), measured in rats with alloxan-induced diabetes. Ann Trop Med Parasitol. 2006;100:69–74.
Tazoo D, Krohn K, Hussain H, Kouam S, Dongo E. Laportoside A and laportomide A: a new cerebroside and a new ceramide from leaves of Laportea ovalifolia. Z Naturforsch. 2007;62b:1208–12.
Njoku O, Okorie I, EC O, Okafor J. Investigation on the phytochemical and antimicrobial properties of Pennisetum purpureum. J Med Arom Plant Sci. 2004;26:311–4.
Zahid Z, Aniruddha P, Sagar D, Subur K, Rana Z. Comparative phytochemical screening of flowers and bark of Spathodea campanulata. Int J Appl Biol Pharmaceut Technol. 2011;2:233–5.
Sowjanya P, Hapsana P, Kiran B, Vagdevi G, Srinivasa B. Pharmacognostical and physicochemical standardization of leaves of Spathodea campanulata P. Beauv. J Pharmacogn Phytochem. 2013;2:189–92.
Rajesh K, Harsha R, Mohammed G, Hareesh A, Thammanna G, Dinesha R, et al. Antimicrobial activity of ethanol extract of leaf and flower of Spathodea campanulata P. Beauv. Res J Pharmaceut Biol Chem Sci. 2010;3:691–8.
Acknowledgments
Authors acknowledge the Cameroon National Herbarium (Yaoundé) for the plant identification. VK is very grateful to the Alexander von Humboldt Foundation for the 2015–2018 Linkage program and 18 months’ fellowship in Germany through the “Georg Foster Research Fellowship for Experienced Researcher” program.
Funding
This work was funded by Alexander von Humboldt Foundation through the 2015–2018 Linkage program to VK and TE.
Availability of data and materials
The datasets supporting the conclusions of this article are presented in this main paper. Plant materials used in this study have been identified at the Cameroon National Herbarium where voucher specimens are deposited.
Authors’ contributions
VK, CFT and FTM carried out the study; VK wrote the manuscript; VPB and TE supervised the work; VK and TE designed the experiments, TE provided the facilities for the study. All authors read the manuscript and approved the final version.
Competing interests
The authors declare that they have no competing interests.
Consent for publication
Not applicable.
Ethics approval and consent to participate
Not applicable.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Kuete, V., Tchinda, C.F., Mambe, F.T. et al. Cytotoxicity of methanol extracts of 10 Cameroonian medicinal plants towards multi-factorial drug-resistant cancer cell lines. BMC Complement Altern Med 16, 267 (2016). https://doi.org/10.1186/s12906-016-1253-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12906-016-1253-3