Abstract
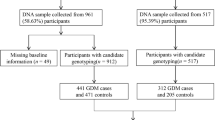
Aims: This study was conducted to investigate the relationship between a genetic polymorphism and the expression of melatonin receptor 1B (MTNR1B) in the placenta of Han Chinese women with gestational diabetes mellitus (GDM). Methods: In this study, 215 patients with GDM and 243 healthy controls were genotyped using direct sequencing for the MTNR1B single-nucleotide polymorphism rs10830963. The expression of MTNR1B in placenta was detected by immunohistochemistry and Western blotting. The association of rs10830963 with the expression of MTNR1B, plasma glucose, and insulin levels as well as blood lipid levels was investigated. Results: The genotype and allele frequencies of rs10830963 were significantly different between women with GDM and controls (P <.05). Fasting blood glucose, fasting insulin, and homeostasis model assessment for insulin resistance in women with GDM with the GG and GC genotypes were significantly higher than those with the CC genotype (P <.05). The expression level of MTNR1B in placenta was significantly higher in the GDM group than in the control group (P <.05). The expression of MTNR1B was significantly higher in all participants with the GG and GC genotypes (1.31 [0.74]) than in pregnant women with the CC genotype (0.92 [0.52], P <.05). Conclusions: The genetic polymorphism rs10830963 in MTNR1B and its protein expression levels in placenta are associated with an increased risk of developing GDM. Furthermore, rs10830963 may tag a molecular mechanism leading to insulin resistance in Han Chinese women with GDM.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Buchanan TA, Xiang AH, Page KA. Gestational diabetes melli-tus: risks and management during and after pregnancy. Nat Rev Endocrinol. 2012;8(11):639–649.
Ruchat SM, Houde AA, Voisin G, et al. Gestational diabetes mellitus epigenetically affects genes predominantly involved in metabolic diseases. Epigenetics. 2013;8(9):935–943.
Reece EA. The fetal and maternal consequences of gestational diabetes mellitus. J Matern Fetal Neonatal Med. 2010;23(3): 199–203.
Mao H, Li Q, Gao S. Meta-analysis of the relationship between common type 2 diabetes risk gene variants with gestational diabetes mellitus. PLoS One, 2012;7(9):e45882.
Huopio H, Cederberg H, Vangipurapu J, et al. Association of risk variants for type 2 diabetes and hyperglycemia with gestational diabetes. Eur J Endocrinol. 2013;169(3):291–297.
Tanev D, Robeva R, Andonova S, et al. Melatonin receptor 1B polymorphisms in women with systemic lupus erythematosus. Acta Reumatol Port. 2016;41(1):62–67.
Thomas S, Amelie B, Ehm A, et al. G-allele of intronic rs10830963 in MTNR1B confers increased risk of impaired fasting glycemia and type 2 diabetes through an impaired glucose-stimulated insulin release. Diabetes. 2009;58(6):1450–1456.
Chambers JC, Zhang W, Zabaneh D, et al. Common genetic variation near melatonin receptor MTNR1B contributes to raised plasma glucose and increased risk of type 2 diabetes among Indian Asians and European Caucasians. Diabetes. 2009;58(11): 2703–2708.
Zheng C, Man CD, Cobelli C, et al. A common variant in the melatonin receptor 1B gene (MTNR1B) is associated with increased risk of impaired fasting glucose (IFG) in obese youth. Obesity. 2015;23:1022–1029.
Kwak SH, Jang HC, Park KS. Finding genetic risk factors of gestational diabetes. Genomics Inform. 2012;10(4):239–243.
Ronn T, Wen J, Yang Z, et al. A common variant in MTNR1B, encoding melatonin receptor 1B, is associated with type 2 diabetes and fasting plasma glucose in Han Chinese individuals. Diabetologia. 2009;52(5):830–833.
De Luis DA, Izaola O, Primo D, Aller R. Association of the rs10830963 polymorphism in melatonin receptor type 1B (MTNR1B) with metabolic response after weight loss secondary to a hypocaloric diet based in Mediterranean style. Clin Nutr. 2017;pii:S0261-5614(17)30298–4.
Sparso T, Bonnefond A, Andersson E, et al. G-allele of intronic rs10830963 in MTNR1B confers increased risk of impaired fasting glycemia and type 2 diabetes through an impaired glucose-stimulated insulin release: studies involving 19,605 Europeans. Diabetes. 2009;58(6):1450–1456.
Okatani Y, Okamoto K, Hayashi K, Wakatsuki A, Tamura S, Sagara Y. Maternal-fetal transfer of melatonin in pregnant woman near term. J Pineal Res. 1998;25(3):129–134.
Hua W, Ling L, Mei Z, et al. Melatonin alleviates lipopolysaccharide-induced placental cellular stress response in mice. J Pineal Res. 2011;50(4):418–426.
Lanoix D, Beghdadi H, Lafond J, Vaillancourt C. Human placental trophoblasts synthesize melatonin and express its receptors. J Pineal Res. 2008;45(1):50–60.
Knabl J, Hiden U, Huttenbrenner R, et al. GDM alters expression of placental estrogen receptor a in a cell type and gender-specific manner. Reprod Sci. 2015;22(12):1488–1495.
Lanoix D, Gurin P, Vaillancourt C. Placental melatonin production and melatonin receptor expression are altered in pre-eclampsia: new insights into the role of this hormone in pregnancy. JPineal Res. 2012;53(4):417–425.
Li C, Qiao B, Zhan Y, et al. Association between genetic variations in MTNR1A and MTNR1B genes and gestational diabetes mellitus in Han Chinese women. Gynecol Obstet Invest. 2013,76(4):221–227.
Costes S, Boss M, Thomas AP, et al. Activation of melatonin signaling promotes beta-cell survival and function. Mol Endocrinol. 2015;29(5):682–692.
Mulder H. Melatonin signalling and type 2 diabetes risk: too little, too much or just right?. Diabetologia. 2017;60(5):826–829.
Vlassi M, Gazouli M, Paltoglou G, et al. The rs10830963 variant of melatonin receptor MTNR1B is associated with increased risk for gestational diabetes mellitus in a Greek population. Hormones (Athens), 2012;11(1):70–76.
Ren J, Xiang AH, Trigo E, et al. Genetic variation in MTNR1B is associated with gestational diabetes mellitus and contributes only to the absolute level of beta cell compensation in Mexican Americans. Diabetologia, 2014;57(7):1391–1399.
Takeuchi F, Katsuya T, Chakrewarthy S, et al. Common variants at the GCK, GCKR, G6PC2-ABCB11 and MTNR1B loci are associated with fasting glucose in two Asian populations. Diabetologia. 2010;53(2):299–308.
Tam CH, Ho JS, Wang Y, et al. Common polymorphisms in MTNR1B, G6PC2 and GCK are associated with increased fasting plasma glucose and impaired beta-cell function in Chinese subjects. PLoS One. 2010;5(7):e11428.
Wang H, Liu L, Zhao J, et al. Large scale meta-analyses offasting plasma glucose raising variants in GCK, GCKR, MTNR1B and G6PC2 and their impacts on type 2 diabetes mellitus risk. PLoS One. 2013;8(6):e67665.
Zhan Y, Li C, Gao Q, Chen J, Yu S, Liu SG. Association between the rs4753426 polymorphism in MTNR1B with fasting plasma glucose level and pancreatic β-cell function in gestational diabetes mellitus. Genet Mol Res. 2015;14(3):8778–8785.
Elmar P, Ina S, Ivonne B, et al. Melatonin and type 2 diabetes—a possible link?. J Pineal Res. 2007;42(4):350–358.
Peschke E, Frese T, Chankiewitz E, et al. Diabetic Goto Kakizaki rats as well as type 2 diabetic patients show a decreased diurnal serum melatonin level and an increased pancreatic melatonin-receptor status. J Pineal Res. 2006;40(2):135–143.
Li MV, Chang B, Imamura M, Poungvarin N, Chan L. Glucose-dependent transcriptional regulation by an evolutionarily conserved glucose-sensing module. Diabetes. 2006;55(5): 1179–1189.
Stumpf I, Bazwinsky I, Peschke E. Modulation of the cGMP signaling pathway by melatonin in pancreatic beta-cells. Journal of Pineal Research. 2010;46(2):140–147
Qiu XS, Tang NL, Yeung HY, et al. Melatonin receptor 1B (MTNR1B) gene polymorphism is associated with the occurrence of adolescent idiopathic scoliosis. Spine. 2007;32(16):1748–1753.
Peschke E, Muhlbauer E. New evidence for a role of melatonin in glucose regulation. Best Pract Res Clin Endocrinol Metab. 2010;24(5):829–841.
Lyssenko V, Nagorny CL, Erdos MR, et al. A common variant in the melatonin receptor gene (MTNR1B) is associated with increased risk of future type 2 diabetes and impaired early insulin secretion. Nat Genet. 2009;41(1):82–88.
Bonnefond A, Clement N, Fawcett K, et al. Rare MTNR1B variants impairing melatonin receptor 1B function contribute to type 2 diabetes. Nat Genet. 2012;44(3):297–301.
Acknowledgments
The authors thank all participants, whose participation made this work possible, and gratefully acknowledge the support of the clinicians and researchers who contributed to this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, C., Zhou, Y., Qiao, B. et al. Association Between a Melatonin Receptor IB Genetic Polymorphism and Its Protein Expression in Gestational Diabetes Mellitus. Reprod. Sci. 26, 1382–1388 (2019). https://doi.org/10.1177/1933719118765983
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719118765983