Abstract
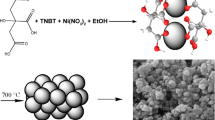
Nanosized powder materials made Zn-doped titanium dioxide with concentration of 0.1, 0.5, and 1.0 mol % Zn2+ have been synthesized from aqueous solutions of inorganic compounds of titanium modified with zinc(II) ions. The materials obtained have been studied by electron microscopy, thermogravimetric analysis, and powder X-ray diffraction. It has been found that if zinc ions introduced in titanium dioxide the stabilization of anatase modification takes place retaining size and morphology of particles. The materials synthesized have shown photocatalytic activity under ultraviolet and visible light irradiation. The highest photocatalytic activity has been found for material containing 0.1 mol % Zn2+ and calcined at 600°С.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Y. Chen, J. Y. Wang, W. Z. Li, and M. T. Ju, J. Mater. Eng. 44, 103 (2016). doi 10.11868/j.issn.1001-4381.2016.03.017
R. Fagan, D. E. McCormack, D. D. Dionysiou, and S. C. Pillai, Mater. Sci. Semicond. Process. 42, 2 (2016). doi 10.1016/j.mssp.2015.07.052
Z. R. Ismagilov, L. T. Tsikoza, N. V. Shikina, V. F. Zarytova, V. V. Zinoviev, and S. N. Zagrebelnyi, Russ. Chem. Rev. 78, 873 (2009). doi 10.1070/RC2009v078n09ABEH004082
M. Pérez-González, S. A. Tomás, J. Santoyo-Salazar, and M. Morales-Luna, Ceram. Int. 43, 8831 (2017). doi 10.1016/j.ceramint.2017.04.016
C. Wang, H. Liu, and Y. Qu, J. Nanomater. 2013, 319637 (2013). doi 10.1155/2013/319637
C. L. Bianchi, E. Colombo, S. Gatto, et al., J. Photochem. Photobiol., A 280, 27 (2014). doi 10.1016/j.jphotochem. 2014.02.002
A. Buthiyappan, A. R. Abdul Aziz, and W. M. A. van Daud, Rev. Chem. Eng. 32, 1 (2016). doi 10.1515/revce-2015-0034
H. Wang and C. You, J. Chem. Eng. 292, 199 (2016). doi 10.1016/j.cej.2016.02.017
S. H. Yoon, Appl. Surf. Sci. 423, 71 (2017). doi 10.1016/j.apsusc.2017.06.147
Y. Li, Y. Bian, Y. Zhang, and Z. Bian, Appl. Catal., B 206, 293 (2017). doi 10.1016/j.apcatb.2017.01.044
J. Li, X. Yang, and T. Ishigaki, J. Phys. Chem. B 110, 14611 (2006). doi 10.1021/jp0620421
L. G. Devi and R. Kavitha, Appl. Catal. B 140–141, 559 (2013). doi 10.1016/j.apcatb.2013.04.035
V. Etacheri, C. D. Valentin, J. Schneider, et al., J. Photochem. Photobiol., C 25, 1 (2015). doi 10.1016/j.jphotochemrev. 2015.08.003
M. Pelaez, N. T. Nolan, S. C. Pillai, et al., Appl. Catal. B: Environ. 125, 331 (2012). doi 10.1016/j.apcatb.2012.05.036
S. A. Bakar and C. Ribeiro, J. Photochem. Photobiol., C 27, 1 (2016). doi 10.1016/j.jphotochemrev.2016.05.001
T. Umebayashi, T. Yamaki, S. Yamamoto, et al., J. Appl. Phys. 93, 5156 (2003). doi 10.1063/1.1565693
M. Fronzi, A. Iwaszuk, A. Lucid, and M. Nolan, J. Phys.: Condens. Matter 28, 23 (2016). doi 10.1088/0953-8984/28/7/074006
M. Hamadanian, S. Karimzadeh, V. Jabbari, and D. Villagrán, Mater. Sci. Semicond. Proc. 41, 168 (2016). doi 10.1016/j.mssp.2015.06.085
M. Anpo, Pure Appl. Chen. 72, 1787 (2000). doi 10.1351/pac200072091787
A. Fuerte, M. D. Hernéz-Alonso, A. J. Maira, et al., Chem. Commun., No. 24, 2718 (2001). doi 10.1039/b107314a
H. Yamashita, J. Synchrotr. Radiat. 8, 569 (2001).
S. N. Phattalung, S. Limpijumnong, and J. Yu, Appl. Catal., B 200, 1 (2017). doi 10.1016/j.apcatb.2016.06.054
L. G. Devi, B. N. Murthy, and S. G. Kumar, Mater. Sci. Eng. B 166, 1 (2010). doi 10.1016/j.mseb.2009.09.008
C. Chen, Z. Wang, S. Ruan, et al., Dyes Pigments 77, 204 (2008). doi 10.1016/j.dyepig.2007.05.003
Y. H. Lin, T. K. Tseng, and H. Chu, Appl. Catal., A 469, 221 (2014). doi 10.1016/j.apcata.2013.10.006
S. Chen, W. Zhao, W. Liu, and S. Zhang, Appl. Surf. Sci. 255, 2478 (2008).
J. N. Deng, B. Yu, and Z. Lou, Sens. Actuators, B 184, 21 (2013).
Y. Ku, Y. H. Huang, and Y. C. Chou, J. Mol. Catal., A 342–343, 18 (2011). doi 10.1016/j.molcata.2011.04.003
G. K. Prasad, P.V.R.K. Ramacharyulu, B. Singh, et al., J. Mol. Catal., A 349, 55 (2011). doi 10.1016/j.molcata. 2011.08.018
E. M. Bayan, T. G. Lupeiko, L. E. Pustovaya, and A. G. Fedorenko, Springer Proc. Phys. 175, 51 (2016). doi 10.1007/978-3-319-26324-3_4
E. M. Bayan, T. G. Lupeiko, L. E. Pustovaya, and A. G. Fedorenko, Nanotechnol. Russ. 12, 269 (2017). doi 10.1134/S199507801703003X
E. M. Bayan, T. G. Lupeiko, E. V. Kolupaeva, et al., Springer Proc. Phys. 193, 17 (2017). doi 10.1007/978-3-319-56062-5_2
V. Stengl, S. Bakardjieva, and N. Murafa, Mater. Chem. Phys. 114, 217 (2009). doi 10.1016/j.matchemphys.2008.09.025
L. Wang, X. Fu, Y. Han, et al., J. Nanomater. 2013, 321459 (2013). doi 10.1155/2013/321459
E. M. Bayan, T. G. Lupeiko, L. E. Pustovaya, A. A. Knyashchuk and A. G. Fedorenko, Russ. J. Phys. Chem. B 11, 600 (2017). doi 10.1134/S1990793117040042
S. Silvestr and E. L. Foletto, Ceram. Int. 43, 14057 (2017). doi 10.1016/j.ceramint.2017.07.140
B. Palanisamy, C. M. Babu, B. Sundaravel, et al., J. Hazard. Mater. 252, 233 (2013). doi 10.1016/j.jhazmat.2013.02.060
M. Crisan, N. Dragan, D. Crisan, et al., Ceram. Int. 42, 3088 (2016). doi 10.1016/j.ceramint.2015.10.097
N. Khatun, P. Rajput, D. Bhattacharya, et al., Ceram. Int. 43, 14128 (2017). doi 10.1016/j.ceramint.2017.07.153
Y. Jiang, Y. Sun, H. Liu, et al., Dyes Pigments 78, 77 (2008). doi 10.1016/j.dyepig.2007.10.009
Z. Liu, C. Liu, J. Ya, and E. Lei, Renew. Energy 36, 1177 (2011). doi 10.1016/j.renene.2010.09.019
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © E.M. Bayan, T.G. Lupeiko, L.E. Pustovaya, 2018, published in Khimicheskaya Fizika, 2018, Vol. 37, No. 10, pp. 36–42.
Rights and permissions
About this article
Cite this article
Bayan, E.M., Lupeiko, T.G. & Pustovaya, L.E. Synthesis and Photocatalytic Activity of Nanosized Powder of Zn-Doped Titanium Dioxide. Russ. J. Phys. Chem. B 12, 923–928 (2018). https://doi.org/10.1134/S1990793118050159
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990793118050159