Abstract
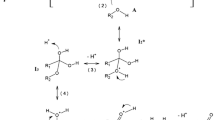
The kinetic parameters of the oxidation of a series of polyunsaturated fatty acids (PUFAs), linoleic and linolenic acids and ethyl esters thereof are determined by measuring the rate R OX of the process in chlorobenzene at 310 K. The selective replacement of H atoms by deuterium atoms results in a dramatic decrease in R OX (the H/D kinetic isotope effect, KIE). Furthermore, the addition of a deuterated PUFA to its nondeuterated analogue decreases R OX for the latter. It looks as if the deuterated PUFA partially protects nondeuterated one from oxidation. The information on the KIE obtained under conditions of initiated liquid-phase oxidation is reported in the current work for the first time; all the numerous previous reports have dealt with enzymatic oxidation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
N. Erdemoglu, S. Kusmenoglu, and M. Vural, Eur. J. Lipid Sci. Technol. 106, 160 (2004).
M. Buchgraber, F. Ulberth, H. Emons, et al., Eur. J. Lipid Sci. Technol. 106, 621 (2004).
K. Aitzetmuller, B. Matthaus, and H. Friedrich, Eur. J. Lipid Sci. Technol. 105, 92 (2003).
T. T. Reed, Free Rad. Biol. Med. 51, 1302 (2011).
G. Spiteller, Free Rad. Biol. Med. 41, 362 (2006).
G. Paradies, G. Petrosillo, V. Paradies, et al., Free Rad. Biol. Med. 48, 1286 (2010).
M. Inoue, E. F. Sato, M. Nishikawa, et al., Redox Rep. 9, 237 (2004).
J. Parry, L. Su, M. Luther, et al., J. Agric. Food Chem. 53, 566 (2005).
N. A. Porter, Acc. Chem. Res. 19, 262 (1986).
J. P. Cosgrove, D. F. Church, and W. A. Pryor, Lipids 22, 299 (1987).
V. A. Roginsky, Mol. Biol. 24, 1582 (1990).
A. D. French, A.-M. Kelterer, G. P. Johnson, et al., J. Mol. Struct. 556, 303 (2000).
S. Korcek, J. H. B. Chenier, J. A. Howard, et al., Can. J. Chem. 50, 2285 (1972).
L. R. C. Barclay and K. U. Ingold, J. Am. Chem. Soc. 103, 6478 (1981).
L. R. C. Barclay, S. J. Locke, and J. M. MacNeil, Can. J. Chem. 61, 1288 (1983).
L. R. C. Barclay, S. J. Locke, J. M. MacNeil, et al., Can. J. Chem. 63, 2633 (1985).
N. A. Porter and D. G. Wujek, J. Am. Chem. Soc. 106, 2626 (1984).
V. A. Roginsky, Kinet. Katal. 31, 546 (1990).
V. Roginsky, Chem. Phys. Lipids 163, 127 (2010).
S. Scheiner, Biochim. Biophys. Acta 1458, 28 (2000).
A. Kohen, Prog. React. Kinet. Mech. 28, 119 (2003).
M. H. Glickman, J. S. Wiseman, and J. P. Klinman, J. Am. Chem. Soc. 116, 793 (1994).
S. Peng and W. A. van der Donk, J. Am. Chem. Soc. 125, 8988 (2003).
D. L. Luthria and H. Sprecher, Biochim. Biophys. Acta 1213, 1 (1994).
K. W. Rickert and J. P. Klinman, Biochemistry 38, 12218 (1999).
C. Jacquot, A. T. Wecksler, C. M. McGinley, et al., Biochemistry 47, 7295 (2008).
M. J. Knapp and J. P. Klinman, Biochemistry 42, 11466 (2003).
A. Kohen and J. P. Klinman, Acc. Chem. Res. 31, 397 (1998).
C. Jacquot, S. Peng, and W. A. van der Donk, Bioorg. Med. Chem. Lett. 18, 5959 (2008).
M. J. Knapp and J. P. Klinman, Eur. J. Biochem. 269, 3113 (2002).
S. Hill, K. Hirano, V. V. Shmanai, et al., Free Rad. Biol. Med. 50, 130 (2010).
E. Bascetta, F. D. Gunstone, and J. C. Walton, J. Chem. Soc., Perkin Trans. II, No. 5, 603 (1983).
J. A. Howard and K. U. Ingold, Can. J. Chem. 40, 1851 (1962).
J. A. Howard and K. U. Ingold, Can. J. Chem. 41, 1744 (1963).
I. Sajenko, V. Pilepic, and C. J. Brala, J. Phys. Chem. A 114, 3423 (2010).
A. Ouchi, S. Nagaoka, and K. Mukai, J. Phys. Chem. B 114, 6601 (2010).
R. H. Bisby and A. W. Parker, Arch. Biochem. Biophys. 317, 170 (1995).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.A. Roginskii, 2015, published in Khimicheskaya Fizika, 2015, Vol. 34, No. 5, pp. 18–22.
Rights and permissions
About this article
Cite this article
Roginskii, V.A. Kinetic isotope effect in the oxidation of unsaturated fatty acids. Russ. J. Phys. Chem. B 9, 352–356 (2015). https://doi.org/10.1134/S1990793115030094
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990793115030094