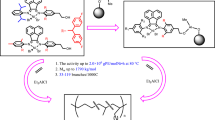
Abstract
The Late transition metal catalysts based on Ni(II) and Co(II) were synthesized and their structure and activity in polymerization of ethylene were compared. Methylaluminoxane (MAO) was used as a co-catalyst. To discover the optimum polymerization conditions, the effect of polymerization temperature, monomer pressure, [Al]: [Ni] molar ratio and time of polymerization were studied. Activity of the catalysts was promoted by increasing of the monomer pressure. The viscosity average molecular weights M v of the synthesized polymers using 1,2-bis(2,4,6-trimethyl phenyl imino) acenaphthene Nickel(II) dibromide were increased with increasing of the monomer pressure from 1 up to 6 bar which studied. Explicitly, the ortho-substituent has a significant effect on the catalyst behavior. Melting point and crystallinity of the obtained polyethylene using 1,2-bis(2,4,6-trimethyl phenyl imino) acenaphthene Nickel(II) dibromide catalyst were increased with enhancing monomer pressure. The optimum and stable structures were computed and some factors related to the activity were studied. Catalyst 1,2-bis(2,4,6-trimethyl phenyl imino) acenaphthene Nickel(II) dibromide had the highest activity with the highest quantities of dipole moment (18.29 Debye), charge of Mullikan on metal atom (1.48) and Sum of electronic and thermal Energies (–7906.52 e.u.).
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
T. J. Pullukat and R. E. Patterson, in Handbook of Transitional Metal Polymerization Catalysts (Wiley, New Jersey, 2010).
T. E. Nowlin, R. I. Mink, and Y. V. Kissin, in Handbook of Transitional Metal Polymerization Catalysts (Wiley, New Jersey, 2010).
L. Wu and S. E. Wanke, in Handbook of Transitional Metal Polymerization Catalysts (Wiley, New Jersey, 2010).
M. P. McDaniel, in Handbook of Transitional Metal Polymerization Catalysts (Wiley, New Jersey, 2010).
L. K. Johnston, C. M. Killian, and M. Brookhart, J. Am. Chem. Soc. 117 (23), 6414 (1995).
W. Kaminsky, J. Polym. Sci., Part A: Polym. Chem. 42, 3911 (2004).
W. E. Piers and T. Chivers, Chem. Soc. Rev. 26, 345 (1997).
Sh. Matsui and T. Fujita, Catal. Today 66, 63 (2001).
J. Huang, B. Lian, Y. Qian, and W. Zhou, Macromolecules 35, 4871 (2002).
G. H. Zohuri, S. Damavandi, S. Ahmadjo, R. Sandaroos, and A. M. Shamekhi, Polyolefins J. 1, 25 (2014).
S. N. Sauca and J. M. Asua, Chem. Eng. J. 168, 1319 (2011).
L. Wang and Q. Wu, Eur. Polym. J. 43, 3970 (2007).
B. R. James and L. D. Markham, J. Catal. 27, 442 (1972).
G. W. Son, K. B. Bijal, D. W. Park, Ch. S. Ha, and I. Kim, Catal. Today 111, 412 (2006).
S. Collins, in Handbook of Transitional Metal Polymerization Catalysts (Wiley, New Jersey, 2010).
H. Pourtaghi-Zahed and G. H. Zohuri, J. Polym. Res. 19, 9996 (2012).
J. Merna, Z. Hostalek, J. Peleska, and J. Roda, Polym. J. 50, 5016 (2009).
J. P. L. Santos, M. Castier, and P. A. Melo, Polym. J. 48, 5152 (2007).
M. Delferro and T. J. Marks, Chem. Rev. 111 (3), 2474 (2011).
S. Damavandi, N. Samadieh, S. Ahmadjo, Z. Etemadinia, and G. H. Zohuri, Eur. Polym. J. 64, 118 (2015).
G. J. P. Britovsek, S. P. D. Baugh, O. Hoarau, V. C. Gibson, D. F. Wass, A. J. P. White, and D. J. Williams, Inorg. Chim. Acta. 345, 279 (2003).
I. Kim, B. H. Han, Y. S. Ha, Ch. S. Ha, and D. W. Park, Catal. Today} 93, 281 (2004).
L. K. Johnson, S. Mecking, and M. Brookhart, J. Am. Chem. Soc. 118, 267 (1996).
H. Zou, F. M. Zhu, Q. Wu, J. Y. Ai and A. Lin, J. Polym. Sci., Part A: Polym. Chem. 43, 1325 (2005).
H. Zou, Sh. Hu, H. Huang, F. Zhu, and Q. Wu, Eur. Polym. J. 43, 3882 (2007).
M. Jeon, Ch. J. Han, and S. Y. Kim, Macromol. Res. 14(3), 306 (2006).
J. Long, H. Gao, F. Liu, K. Song, L. Zhang, F. Zhu, Q. Wu, and H. Hu, Inorg. Chim. Acta. 362, 3035 (2009).
H. Liu, W. Sun, and W. Zhang, WO Patent No. 2012122854 A1 (2012).
L. Li, M. Jeon, and S. Y. Kim, J. Mol. Catal. A: Chem. 303, 110 (2009).
S. A. Sangokoya, EP Patent No. 0463555 B1 (1996).
J. Brandrup and E. H. Immergut, Polymer Handbook (Wiley, New York, 1989).
L. Wang and J. Sun, Inorg. Chim. Acta. 361, 1843 (2008).
K. P. Bryliakov, E. P. Talsi, and M. Bochmann, Organometallics 23, 149 (2004).
G. H. Zohuri, S. Ahmadjo, S. Damavandi, and R. Sandaroos, Polym. Bull. 66, 1051 (2011).
E. N. Jacobson and R. Breinbauer, Science 287, 437 (2000).
G. H. Zohuri, S. Damavandi, S. Ahmadjo, R. Sandaroos, and M. A. Shamekhi, Polyolefins J. 1, 25 (2014).
L. C. Simona, C. P. Williams, J. B. P. Soares, and R. F. de Souza, J. Mol. Catal. A: Chem. 165, 55 (2001).
H. Arabi, M. S. Beheshti, M. Yousefi, N. Ghasemi Hamedani, and M. Ghafelebashi, Polym. Bull. 70, 2765 (2013).
Zh. Guan and Ch. S. Popeney, Top. Organomet. Chem. 24, 179 (2009).
M. Vatankhah-Varnoosfaderani, S. Pourmahdian, and F. Afshar-Taromi, Iran. Polym. J. 20(11), 897 (2011).
S. P. Netalkar, S. Budagumpi, H. H. Abdallah, P. P. Netalkar, and K. Revankar, J. Mol. Struct. 1075, 559 (2014).
G. H. Zohuri, R. Sandaroos, A. Mohammadi, S. M. Seyedi, and S. Damavandi, Catal. Lett. 140, 160 (2010).
N. Bahri-Laleh, M. Nekoomanesh-Haghighi, and S. A. Mirmohammadi, J. Organomet. Chem. 719, 74 (2012).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Khoshsefat, M., Beheshti, N., Zohuri, G.H. et al. Practical and theoretical study on the α-substituent effect on α-diimine Nickel(II) and Cobalt(II)-based catalysts for polymerization of ethylene. Polym. Sci. Ser. B 58, 487–494 (2016). https://doi.org/10.1134/S1560090416050067
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1560090416050067