Abstract
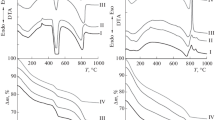
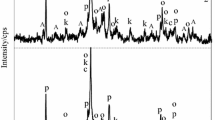
The effect of a medium’s acidity on the composition of the solid phase formed in aqueous calcium-silicate systems is investigated. Solutions of Са(NO3)2 and Na2SiO3 are used for the synthesis; the pH values were varied in the range 7.00–12.00. Freshly precipitated solid phases and products of their annealing at 1000°C were studied by the methods of Fourier IR spectroscopy, X-ray diffraction (XRD), and scanning electron microscopy (SEM).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Meiszterics, A. and Sinko, K., Sol-gel derived calcium silicate ceramics, Colloids Surf., A, 2008, vol. 319, pp. 143–148.
Foley, E.M., Kim, J.J., and Taha, M.M.R., Synthesis and nano-mechanical characterization of calcium–silicate-hydrate (C–S–H) made with 1.5CaO/SiO2 mixture, Cement Concrete Res., 2012, vol. 42, pp. 1225–1232.
Long, L.H., Chen, L.D., and Chang, J., Low temperature fabrication and characterizations of β-CaSiO3 ceramics, Ceram. Int., 2006, vol. 32, pp. 457–460.
Akat’eva, L.V., Development of chemical technological principles of raw material treatment processes for production of calcium silicates and composition materials, Extended Abstract of Doctoral Dissertation, Moscow, 2014.
Mojumdar, S.C. and Raki, L., Preparation, thermal, spectral and microscopic studies of calcium silicate hydrate-poly(acrylic acid) nanocomposite materials, J. Therm. Anal. Calorim., 2006, vol. 85, no. 1, pp. 99–105.
Ni, S., Lin, K., Chang, J., and Chou, L., β-CaSiO3-β-Ca3(PO4)2 composite materials for hard tissue repair: In vitro studies, J. Biomed. Mater. Res., 2008, vol. 85, no. 1, p. 72.
Chen, J.J., Thomas, J.J., Taylor, H.F.W., and Jennings, H.M., Solubility and structure of calcium silicate hydrate, Cement Concrete Res., 2004, vol. 34, p. 1499.
Wu, J., Zhu, Y.J., and Chen, F., Ultrathin calcium silicate hydrate nanosheets with large specific surface areas: synthesis, crystallization, layered self-assembly and applications as excellent adsorbents for drug, protein, and metal ions, Small, 2013, vol. 9, no. 17, pp. 2911–2925.
Chukin, D.G., Khimiya poverkhnosti i stroenie dispersnogo kremnezema (Chemistry of the Surface and Disperse Sicila Structure), Moscow: Paladin, Printa, 2008.
Yu, P., Kirkpatrick, R.J., Poe, B., McMillan, P.F., and Cong, X., Structure of calcium silicate hydrate (C–S–H). Near-, mid-, and far-infrared spectroscopy, J. Am. Ceram. Soc., 1999, vol. 82, no. 3, pp. 742–748.
Yarusova, S.B., Synthesis of calcium silicates in multicomponent systems and their physicochemical properties, Extended Abstract of Cand. Sci. Dissertation, Vladivostok, 2010.
Richardson, I.G., The calcium silicate hydrate, Cement Concrete Res., 2008, vol. 38, pp. 137–158.
Correcher, V., Garcia-Guinea, J., Bustillo, M.A., and Garcia, R., Study of the thermoluminescence emission of a natural α-cristobalite, Radiat. Effects Defects Solids, 2009, vol. 164, no. 1, pp. 59–67.
Finnie, K.S., Thompson, J.G., and Withers, R.L., Phase transitions in cristobalite and related structures studied by variable temperature infer-red emission spectroscopy, J. Phys. Chem. Solids, 1994, vol. 55, no. 1, pp. 23–29.
Luyt, A.S., Dramicanin, M.D., Antic, Z., and Djokovic, V., Morphology, mechanical and thermal properties of composites of polypropylene and nanostructured wollastonite filler, Polym. Test., 2009, vol. 28, pp. 348–356.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.P. Solonenko, A.I. Blesman, D.A. Polonyankin, L.V. Bel’skaya, 2017, published in Fizika i Khimiya Stekla.
Rights and permissions
About this article
Cite this article
Solonenko, A.P., Blesman, A.I., Polonyankin, D.A. et al. Peculiarities of the composition of solid phases formed in aqueous calcium–silicate systems with a medium of variable acidity. Glass Phys Chem 43, 452–458 (2017). https://doi.org/10.1134/S1087659617050182
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1087659617050182