Abstract
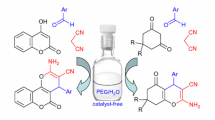
A novel one-pot approach has been developed for a high-yield synthesis of 4-phenyl-4H-pyran derivatives by the three-component condensation of dimethyl 3-oxopentanedioate or 3-oxo-3-arylpropanoic acid esters, aldehydes, with malononitrile in the presence of a DIPEA catalyst in ethanol at room temperature. The significant features of the present strategy include efficiency, high yield, inexpensive catalyst, mild reaction conditions, and simple purification procedure.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Domling, A. and Ugi, I., Angew Chem Int Ed Engl., 2000, vol. 39, p. 3168. https://doi.org/10.1002/1521-3773(20000915) 39:183168::aid-anie31683.0.co;2-u
Ugi, I., Pure. Appl. Chem., 2001, vol. 73, p. 187. https://doi.org/10.1351/pac200173010187
Sunderhaus, J. and Martin, S., Chem. Eur. J., 2009, vol. 15, p. 1300. https://doi.org/10.1002/chem.200802140
Toure, B.B. and Hall, D.G., Chem. Rev., 2009, vol. 41, p. 4439. https://doi.org/10.1021/cr800296p
Ghosh, A.K. and Sarkar, A., Tetrahedron Lett., 2017, vol. 58, p. 3230. https://doi.org/10.1016/j.tetlet.2017.07.010
Babu, N.S., Pasha, N., Rao, K.T.V, Prasad, P.S.S., and Lingaiah, N., Tetrahedron Lett., 2008, vol. 49, p. 2730. https://doi.org/10.1016/j.tetlet.2008.02.154
Kemnitzer, W., Drewe, J., Jiang, S., Zhang, H., Crogan-Grundy, C., Labreque, D., Bubenick, M., Attardo, G., Denis, R., Lamothe, S., Gourdeau, H., Tseng, B., Kasibhatla, S., and Cai, S., J. Med. Chem., 2008, vol. 51, p. 417. https://doi.org/10.1021/jm7010657
Kemnitzer, W., Drewe, J., Jiang, S., Zhang, H., Zhao, J., Crogan-Grundy, C., Xu, L., Lamothe, S., Gourdeau, H., Denis, R., Tseng, B., Kasibhatla, S., and Cai, S., J. Med. Chem., 2007, vol. 50, p. 2858. https://doi.org/10.1021/jm070216c
Li, M.M., Duan, C.S., Yu, Y.Q., and Xu, D.Z., Dyes Pigments, 2018, vol. 150, p. 202. https://doi.org/10.1016/j.dyepig.2017.12.007
Schmitt, F., Kasparkova, J., Brabec, V., Begemann, G., Schobert, R., and Biersack, B., J. Lnorg. Biochem., 2018, vol. 184, p. 69. https://doi.org/10.1016/jjinorgbio.2018.03.013
Sabitha, G., Bhikshapathi, M., Nayak, S., Srinivas, R., and Yadav, J.S., J. Heterocycl. Chem., 2011, vol. 48, p. 267. https://doi.org/10.1002/jhet.544
Babu, N.S., Pasha, N., Rao, K.T.V., Prasad, P.S.S., and Lingaiah, N., Tetrahedron Lett., 2008, vol. 49, p. 2730. https://doi.org/10.1016/j.tetlet.2008.02.154
Keshavarz, M., Iravani, N., Azghandi, M.H.A., and Nazari, S., Res. Chem. Intermed., 2016, vol. 42, p. 4591. https://doi.org/10.1007/slll64-015-2302-0
Kalita, S.J., Saikia, N., Deka, D.C., and Mecadon, H., Res. Chem. Intermed., 2016, vol. 42, p. 6863. https://doi.org/10.1007/slll64-016-2500-4
Gutierrez, L.F., Nope, E., Rojas, H.A., Cubillos, J.A., and Martinez, J.J., Res. Chem. Intermed., 2018, vol. 44, p. 1. https://doi.org/10.1007/slll64-018-3440-y
Dekamin, M.G., Eslami, M., and Maleki, A., Tetrahedron, 2013, vol. 69, p. 1074. https://doi.org/10.1002/chin.201322145
Safaei-Ghomi, J., Teymuri, R., Shahbazi-Alavi, H., and Ziarati, A., Chin. Chem. Lett., 2013, vol. 24, p. 921. https://doi.org/10.1002/chin.201406166
Banerjee, S., Horn, A., Khatri, H., and Sereda, G., Tetrahedron Lett., 2011, vol. 52, p. 1878. https://doi.org/10.1016/j.tetlet.2011.02.031
Niknam, K., Borazjani, N., Rashidian, R., and Jamali, A., Chin. J. Catal, 2013, vol. 34, p. 2245. https://doi.org/10.1016/S1872-2067(12)60693-7
Kidwai, M., Saxena, S., Khan, M.K.R., and Thukral, S.S., Bioorg. Med. Chem. Lett., 2005, vol. 15, p. 4295.
Naimi-Jamal, M.R., Mashkouri, S., and Sharifi, A., Mol. Divers, 2010, vol. 14, p. 473. https://doi.org/10.1007/sll030-010-9246-5
Erichsen, M.N., Huynh, T.H.V., Abrahamsen, B., Bastlund, J.F., Bundgaard, C., and Monrad, O., J. Med. Chem., 2016, vol. vol. 53, p. 7180. https://doi.org/10.1021/jml009154
Gong, K., Wang, H.L., Fang, D., and Liu, Z.L., Catal. Commun., 2008, vol. 9, p. 650. https://doi.org/10.1016/j.catcom.2007.07.010
Waghmare, A.S., Pandit, S.S., and Suryawanshi, D.M., Comb. Chem. High, Throughput Screen, 2018, vol. 21, p. 254. https://doi.org/10.21741386207321666180315095422
Gutierrez, L.F., Nope, E., Rojas, H.A., Cubillos, J.A., and Martinez, J.J., Res. Chem. Intermed., 2018, vol. 44, p. 1. https://doi.org/10.1007/slll64-018-3440-y
Heber, D. and Stoyanov, E.V., ChemInform, 2003, vol. 34, p. 227. https://doi.org/10.1002/chin.200320151
Funding
This study was financially supported by the Shenyang Science & Technology project (project no. 18-013-0-03) and Natural Science Funds for Young Scholar of Liaoning University (project no. LDQN201436).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Li, J., Li, J., Hu, Sy. et al. Efficient Synthesis of 4-Phenyl-4H-pyran Derivatives via a DIPEA-catalyzed One-Pot Three-Component Reaction. Russ J Org Chem 55, 1791–1799 (2019). https://doi.org/10.1134/S107042801911023X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S107042801911023X