Abstract
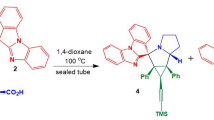
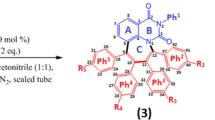
The NMR spectra of several heterocyclic compounds synthesized at Omsk universities revealed manifestation of a diasteomerism at a large (up to fifteen bonds) distance between the stereogenic centers. This phenomenon was explained by a regular helical secondary structure of the tether between the centers.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Talsi, V.P. and Evdokimov, S.N., Russ. J. Org. Chem., 2017, vol.53, p.1630. doi https://doi.org/10.1134/S1070428017110021
Barysheva, N.A., Ikrina, M.A., Simonov, V.D., and Talsi, V.P., Zh. Org. Khim., 1985, vol.21, p.515.
Ovchinnikova, I.G., Candidate Sci. (Chem.) Dissertation, Yekaterinburg, 2007.
Gein, V.L., Zamaraeva, T.M., Dmitriev, M.V., Ovchinnikova, I.G., and Fedorova, O.V., Russ. J. Org. Chem., 2017, vol.53, p.1090. doi https://doi.org/10.1134/S1070428017070223
Ovchinnikova, I.G., Fedorova, O.V., Stepukhin, P.A., Litvinov, I.A., and Rusinov, G.L., Crystall. Rep., 2009, vol.54, p.31. doi https://doi.org/10.1134/S1063774509010064
Ohrui, H. and Matsui, M., Proc. Jpn. Acad. Ser. B, Phys. Biol. Sci., 2007, vol.83, p.127. doi https://doi.org/10.2183/pjab.83.127
Lehn, J.M., Nitrogen Inversion: Experimental & Theory, Berlin: Springer-Verlag, 1970.
Clayden, J., Lemiegre, L., Morris, G.A., Pickworth, M., Snape, T.J, and Jones, L.H., J. Am. Chem. Soc., 2008, vol.130, p.15193. doi https://doi.org/10.1021/ja805758v
Cram, D.J. and Knox, G.R., J. Am. Chem. Soc., 1961, vol.83, p.2204.
Jenneskens, L.W., Klamer, J.C., de Boer, H.J.R., de Wolf, W.H., Bichlhaupt, F., and Stam, C.H., Angew. Chem., 1984, vol.96, p.236. doi https://doi.org/10.1002/anie.198402381
Author information
Authors and Affiliations
Corresponding author
Additional information
Russian Text © V.P. Talsi, S.N. Evdokimov, A.L. Shatsauskas, 2019, published in Zhurnal Organicheskoi Khimii, 2019, Vol. 55, No. 2, pp. 208–214.
Rights and permissions
About this article
Cite this article
Talsi, V.P., Evdokimov, S.N. & Shatsauskas, A.L. Investigation of the Diastomerism of Compounds with a Long-Chain Tether between the Stereogenic Centers. Russ J Org Chem 55, 161–167 (2019). https://doi.org/10.1134/S1070428019020064
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428019020064