Abstract
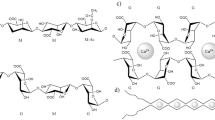
A novel nano flocculant was prepared through the modification of sodium alginate. In the preparation partially oxidised sodium alginate reacted with triethylenetetramine (TETA) and then reacted with carbon disulfide. The structure of the flocculants was confirmed by elemental analysis, Fourier transform infrared and UV spectrophotometry. The flocculant showed nanoparticle sizes with diameters of 300–350 nm in aqueous solution, and became precipitates after flocculation with Pb2+ ions. The flocculant was employed to adsorb Pb2+ in wastewater, and the influence of flocculant concentration, flocculation temperature, pH, and time on the adsorption properties were investigated. The results showed that adsorption capacity of the flocculant for Pb2+ could reach up to 3.43 mmol g–1, and the removal rate for Pb2+ was over 97%. The adsorption processes corresponded to pseudo second order kinetics and Langmuir isotherm model. The adsorption mechanism involved electrostatic attraction, chemical chelation, netting and sweeping actions.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Li, P.Z., Lin, C.Y., Cheng, H.G., Duan, X.L., and Lei, K., Ecotoxicology & Environmental Safety, 2015, vol. 113, pp. 391–399.
Zhang, X.W., Yang, L.S., Li, Y.H., Li, H.R., Wang, W.Y., and Ye, B.X., Environ. Monit. Assess, 2012, vol. 184, pp. 2261–2273.
Fu, F.L. and Wang, Q., J. Environ. Manage, 2011, vol. 92, pp. 407–418.
Zhang, N., Zang, G.L., Shi, C., Yu, H.Q., and Sheng, G.P., J. Hazard. Mater., 2016, vol. 316, pp. 11–18.
Leea, C.S., Robinsonb, J., and Chong, M.F., Process Saf. Environ., 2014, vol. 92, pp. 489–508.
Aljuboori, A.H.R., Idris, A., Hussain H., Al-joubory, R., Uemura, Y., and Ibn Abubakar, B.S.U., J. Environ. Manage, 2015, vol. 150, pp. 466–471.
Yang, Z., Jia, S.Y., Zhang, T.T., et al., Sep. Purif. Technol., 2015, vol. 149, pp. 398–406.
Huang, Y.F., Wua, D.H., Wang, X.D., Huang, W., Lawless, D., and Feng, X.S., Sep. Purif. Technol., 2016, vol. 158, pp. 124–136.
Alizadeh, B., Ghorbani, M., and Salehi, M. A., J. Mol. Liq., 2016, vol. 220, pp. 142–149.
Hana, R.P., Zoua, W.H., Zhang, Z.P., Shi, J., and Yang, J.J., J. Hazard. Mater., 2006, vol. 137, pp. 384–395.
Mostafa, M.S., Bakr, A.A., El Naggar, A.M.A., and Sultan, E.A., J. Colloid Interf. Sci., 2016, vol. 461, pp. 261–272.
Mohammadi, S.Z., Hamidian, H., and Moeinadini, Z., Ind. Eng. Chem., 2014, vol. 20, pp. 4112–4118.
Guo, J.Y., Yu, J., Xin, X., et al., Bioresource Technol., 2015, vol. 177, pp. 393–397.
Pawar, S.N. and Edgar, K.J., Biomaterials, 2012, vol. 33, pp. 3279–3305.
Leea, K.Y., Mooney, D.J., Prog. Polym. Sci., 2012, vol. 37, pp.106–126.
Dekamina, M.G., Peymana, S.Z., Karimia, Z., Javanshira, S., Reza Naimi-Jamala, M., and Barikani, M., Inter. J. Biol. Macromol., 2016, vol. 87, pp. 172–179.
Burckbuchler, V., Kjøniksen, A., Galant, C., Amiel, R.L.C., Knudsen, K.D., and Nyström, B., Biomacromolecules. 2006, vol. 7, pp. 1871–1878.
Augst, A.D. and Kong, H.J., Mooney, D.J., Macromol. Biosci. 2006, vol. 6, pp. 623–633
Wang, F., Lu, X.W., and Li, X.Y., J. Hazard. Mater. 2016, vol. 308, pp.75–83.
Ren, H.X., Gao, Z.M., Wu, D.J., Jiang, J.H., Sun, Y., and Luo, C.W., Carbohyd. Polym. 2016, vol. 137, pp. 402–409.
Lu, T., Xiang, T. and Huang, X.L., et al., Carbohyd. Polym. 2015, vol. 133, pp. 587–595.
Balakrishnan, B., Lesieur, S., Labarreb, D., and Jayakrishnan, A., Carbohyd. Res., 2005, vol. 340, pp. 1425–1429.
Mallampati, R. and Valiyaveettil, S., ACS Appli. Mater. Inter. 2013, vol.5, pp. 4443–4449.
Wang, G., Chang, Q., Han, X.T., and Zhang, M.Y., J. Hazard. Mater. 2013, vols. 248–249, pp. 115–121.
Kano, N. and Kawashima, T., Top. Curr. Chem., 2005, vol. 251, pp. 141–180.
Jiang, Y., Zhang, H.W., Li, H.T., Wu, M.Z., Zhang, S.X., and Wang, J.Y., J. Mol. Struct. 2004, vol. 702, pp. 33–37.
Zeng, H.L., Xiang, X.Y., Tan, M.Z., Zhu, G.C., He, Q., and Ma, J.Y., Asian J. Chem. 2013, vol. 25, pp. 1467–1470.
Matisa, K.A., Zouboulisa, A.I., Lazaridisa, N.K., and Hancock, I.C., Int. J. Miner. Process, 2003, vol. 70, pp. 99–108.
Feng, J., Yang, Z.H., Zeng, G.M., et al., Bioresource Technol., 2013, vol. 148, pp. 414–421.
Dahiya, S., Tripathi, R.M., and Hegde, A.G., J. Hazard. Mater., 2008, vol. 150, pp. 376–386.
Ho, Y.S. and McKay, G., Process Biochem. 1999, vol. 34, pp. 451–465.
Chiou, M.S. and Li, H.Y., Chemosphere, 2003, vol. 50, pp. 1095–1105.
Ngah, W.S.W. and Fatinathan, S., Chem. Eng. J., 2008, vol. 143, pp. 62–72.
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Tian, Z., Zhang, L. & Ni, C. Preparation of modified alginate nanoflocculant and adsorbing properties for Pb2+ in wastewater. Russ J Appl Chem 90, 641–647 (2017). https://doi.org/10.1134/S1070427217040218
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427217040218