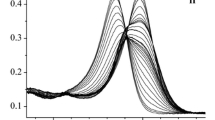
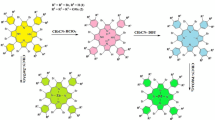
Abstract
Acidity and metal ion coordination are described for three porphyrin derivatives, different in their macrocycle conformation and electronic substitution effects due to bromine substitution in pyrrole rings and trifluorometyl or phenyl groups in meso-positions. Combination of these facts allows modulating both steric and electronic effects on the macrocycle π-conjugated system. The role of electronic substitution effects in the macrocycle deprotonation and metal ion complex formation is found dominating with comparable resonance and inductive contributions, whereas non-planar conformation of reactive species contributes to the reaction rates to a lesser extent. The interaction of two single-electron (a1ueg) and (a2ueg) configurations is studied as a function of non-planar distortions of the molecular structure for the three tetrapyrrole compounds. The additive influence of disturbing factors on the configuration interaction of single-electron (a1ueg) and (a2ueg) configurations in the tetrapyrrole macrocycle is demonstrated.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Berezin, B.D., Coordination Compounds of Porphyrins and Phthalocyanines, New York: Wiley, Toronto, 1981.
Davis, D.J., The Porphyrins, Dolphin, D., Ed., New-York: Academic Press, 1978, vol. 5, p. 127.
Sheidt, W.R. and Lee, Y., Struct. Bonding, 1987, vol. 64, p. 1.
Berova, N., Pescitelli, G., Petrovic, A.G., and Proni, G., Chem. Commun., 2009, p. 5958. doi https://doi.org/10.1039/B909582A
Bottari, G., Trukhina, O., Ince, M., and Torres, T., Coord. Chem. Rev., 2012, vol. 256, p. 2453. doi https://doi.org/10.1016/j.ccr.2012.03.011
Tashiro, K. and Aida, T., Chem. Soc. Rev., 2007, vol. 36, p. 189. doi https://doi.org/10.1039/B614883M
Wienkers, M., Ramos, J., Jemal, H., and Cardenas, C., Org. Lett., 2012, vol. 14, p. 1370. doi https://doi.org/10.1021/ol203249x
Shelton, A.H., Rodger, A., McMillin, D.R., Biochemistry, 2007, vol. 46, p. 9143. doi https://doi.org/10.1021/bi700293g
Allard, M., Dupont, C., Muñoz Robles, V., Douce, N., Lledós, A., Maréchal, J.-D, Urvoas, A., Mahy, J.-P., and Ricoux, R., ChemBioChem, 2012, vol. 13, p. 240. doi https://doi.org/10.1002/cbic.201100659
Venkatramaiah, N., Pereira, C.F., Mendes, R.F., Paz, F.A.A., and Tomé João, P.C., Anal. Chem., 2015, vol. 87, no. 8, p. 4515. doi https://doi.org/10.1021/acs.analchem.5b00772
Romero-Nieto, C., Garcia, R., Herranz, M.A., Ehli, C., Ruppert, M., Hirsch, A., Guldi, D.M., and Martin, N.J., J. Am. Chem. Soc., 2012, vol. 134, p. 9183. doi https://doi.org/10.1021/ja211362z
Yoon, H., Lee, C.H., and Jang, W.D., Chem. Eur. J., 2012, vol. 18, p. 12479. doi https://doi.org/10.1002/chem.201200371
Hermida-Ramon, J.M. and Estevez, C.M., Chem. Eur. J., 2007, vol. 13, p. 4743. doi https://doi.org/10.1002/chem.200601836
Klarner, F.G. and Kahlert, B., Acc. Chem. Res., 2003, vol. 36, p. 919. doi https://doi.org/10.1016/S1874-6004(08)80008-1
Carraro, M., Modugno, G., Fiorani, G., Maccato, C., Sartorel, A., and Bonchi, M., Eur. J. Org. Chem., 2012, p. 281. doi https://doi.org/10.1002/ejoc.201101122
Steinle, E.D., Schaller, U., and Meyerhoff, M.E., Anal. Sci., 1998, vol. 14, p. 79. doi https://doi.org/10.2116/analsci.14.79
Ivanova, Yu.B., Dao, T.N., Kruk, N.N., and Syrbu, S.A., Russ. J. Gen. Chem., 2013, vol. 83, no. 6, p. 1155. doi https://doi.org/10.1134/S107036321306025X
Ivanova, Yu.B., Dao Tkhe Nam, Glazunov, A.V., Semeikin, A.S., and Mamardashvili, N.Zh., Russ. J. Gen. Chem., 2012, vol. 82, no. 7, p. 1272. doi https://doi.org/10.1134/S1070363212070158
Vlascici, D., Cosma, E.F., Pica, E.M., Cosma, V., Bizerea, O., Mihailescu, G., and Olenic, L., Sensors, 2008, vol. 8, p. 4995. doi https://doi.org/10.3390/s8084995
Pukhovskaya, S.G., Fien, C.D., Domanina, E.N., Semeikin, A.S., Nam, D.T., and Ivanova, Yu.B., Russ. J. Phys. Chem. A, 2017, vol. 91, no. 9, p. 1692. doi https://doi.org/10.1134/S0036024417090242
Ivanova, Yu.B., Pukhovskaya, S.G., Semeykin, A.S., and Syrbu, S.A., Russ. J. Gen. Chem., 2013, vol. 83, p. 1406. doi https://doi.org/10.1134/S1070363213070177
Pukhovskaya, S.G., Efimovich, V.A., Semeikin, A.S., and Golubchikov, O.A., Russ. J. Inorg. Chem., 2010, vol. 55, p. 1494. doi https://doi.org/10.1134/S0036023610090275
Golubchikov, O.A., Pukhovskaya, S.G., and Kuvshinova, E.M., Russ. Chem. Rev., 2005, vol. 74, p. 249. doi https://doi.org/10.1070/RC2005v074n03ABEH000925
Andrianov, V.G. and Malkova, O.V., Macroheterocycles, 2009, vol. 2, no. 2, p. 130.
Dao Tkhe Nam, Ivanova, Yu.B., Puhovskaya, S.G., Kruk, M.M., Syrbu, S.A., RSC Adv., 2015, vol. 5, p. 26125. doi https://doi.org/10.1039/c5ra01323b
Nelson, N.Y., Medforth, C.J., Nurco, D.J., Jia, S.-L., Shelnutt, J.A., and Smith, K.M., Chem. Commun., 1999, p. 2071. doi https://doi.org/10.1039/A904532E
Bhyrappa, P. and Krishnan, V., Inorg. Chem., 1991, vol. 30, no. 2, p. 239. doi https://doi.org/10.1021/ic00002a018
Goll, J.G., Moore, K.T., Ghosh, A., Therien, M.J., J. Am. Chem. Soc., 1996, vol. 118, no. 35, p. 8344. doi https://doi.org/10.1021/ja9610904
Kaljurand, A., Kütt, L., Sooväli, T., Rodima, V., Mäemets, I., Leito, I., and Koppel, A., Org. Chem., 2005, vol. 70, no. 3, p. 1019. doi https://doi.org/10.1021/jo048252w
Ivanova, Yu.B., Sheinin, V.B., Mamardashvili, N.Zh., Russ. J. Gen. Chem., 2007, vol. 77, no. 8, p. 1458. doi https://doi.org/10.1134/S107036320708027
Ivanova, Yu.B., Churakhina, Yu.I., and Mamardashvili, N.Zh., Russ. J. Gen. Chem., 2008, vol. 78, no. 4, p. 673. doi https://doi.org/10.1134/S1070363208040269
Ivanova, Yu.B., Chizhova, N.V., and Kruk, N.N., Russ. J. Gen. Chem., 2013, vol. 83, no. 3, p. 558. doi https://doi.org/10.1134/S1070363213030250
D’Souza, F., Zandler, M.E., Tagliatesta, P., Ou, Z., Shao, J., Van Caemelbecke, E., and Kadish, K.M., Inorg. Chem., 1998, vol. 37, no. 18, p. 4567.
Hoffman, P., Labat, G., Robert, A., and Meunier, B., Tetrahedron Lett., 1990, vol. 31, no. 14, p. 1991.
Gouterman, M., The Porphyrins, Dolphin, D., Ed., New York: Academic Press, 1978, vol. 4, p. 1.
Spellane, P.J., Gouterman, M., Antipas, A., Kim, S., and Liu, Y.C., Inorg. Chem., 1980, vol. 19, p. 386–391.
Bernshtein, I.Ya., Spektrofotometricheskii analiz v organicheskoi khimii (Spectrophotometric Analysis in Organic Chemistry), Leningrad: Chemistry, 1986, p. 202.
Berezin, B.D.J., Porphyrins Phthalocyanines, 2003, vol. 7, p. 715.
Pukhovskaya, S.G., Efimovich, V.A., Semeikin, A.S., Kolodina, E.A., and Golubchikov, O.A., Russ. J. Gen. Chem., 2012, vol. 82, no. 3, p. 476. doi https://doi.org/10.1134/S107036321203019X
Pukhovskaya, S.G., Efimovich, V.A., Golubchikov, O.A., Russ. J. Inorg. Chem., 2013, vol. 58, no. 4, p. 406. doi https://doi.org/10.1134/S0036023613040141
Pukhovskaya, S.G., Guseva, L.G., Malkova, O.V., Semeikin, A.S., and Golubchikov, O.A., Russ. J. Gen. Chem., 2003, vol. 73, p. 473.
Berezin, D.B., Ivanova, Yu.B., and Scheinin, V.B., Russ. J. Phys. Chem. A, 2007, vol. 81, no. 12, p. 1986. doi https://doi.org/10.1134/S003602440712014X
Gordon, A.J. and Ford, R.A., The Chemist’s Companion. A Handbook of Practical Data, Techniques and References, New York: Wiley, 1972.
Kruk, M.M. and Braslavsky, S.E., Photobiol. Sci., 2012, vol. 11, p. 972. doi https://doi.org/10.1039/C2PP05368C
Kruk, M.M., Starukhin, A.S., and Wouter, M., Macroheterocycles, 2011, vol. 4, no. 2, p. 69. doi https://doi.org/10.6060/mhc2011.2.01.
Yamauchi, S., Matsukawa, Y., Ohba, Y., and Iwaizumi, M., Inorg. Chem., 1996, vol. 35, p. 2910.
Liulkovich, L.S. and Kruk, M.M., Proceedings of Belarusian State Technological University (BSTU): Physics, Mathematics and Computer Science, 2015, vol. 170, no. 6, p. 63.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Russian Text © The Author(s), 2017, published in Rossiiskii Khimicheskii Zhurnal, 2017, Vol. 61, No. 1, pp. 56–66.
Funding
This work is performed with financial support of the Russian Foundation for Basic Research (grant no. 16-53-00100 Bel-a).
Conflict of Interest
No conflict of interest was declared by the authors.
Rights and permissions
About this article
Cite this article
Pukhovskaya, S.G., Ivanova, Y.B., Semeikin, A.S. et al. Investigation of Acidic and Coordination Properties of Octabromo-Substituted Porphyrins in the System of 1,8-Diazabicyclo[5,4,0]unde-7-ene-Acetonitrile. Russ J Gen Chem 89, 1286–1296 (2019). https://doi.org/10.1134/S1070363219060252
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363219060252