Abstract
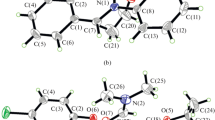
Manganese(III) complexes derived from the bis-Schiff bases N,N′-bis(5-fluorosalicylidene)-1,2-diaminoethane (H2La) and 3,4-bis(2-hydroxybenzylideneamino)pyridine (H2Lb), respectively, have been prepared and characterized by elemental analyses, IR, and single crystal X-ray crystallographic determination (CIF files CCDC nos. 997243 (I), 995896 (II)). The crystal of [MnLa(μ1,3-N3)] n (I) is orthorhombic: space group Pca21, a = 10.723(1), b = 13.430(1), c = 11.112(1) Å, V = 1600.2(2) Å3, Z = 4, R 1 = 0.0264, wR 2 = 0.0649. The crystal of [MnLb(N3)(CH3OH)] (II) is monoclinic: space group C2/c, a = 22.792(1), b = 14.4442(7), c = 12.8637(6) Å, β = 119.262(1)°, V = 3694.5(3) Å3, Z = 8, R 1 = 0.0367, wR 2 = 0.0776. The bis-Schiff base ligands coordinate to the metal atoms through phenolate O and imine N atoms. Each metal atom in the complexes is in octahedral coordination. The effects of the complexes on the antimicrobial activity against Staphylococcus aureus, Escherichia coli, and Candida albicans were studied.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Pradeep, C.P. and Das, S.K., Coord. Chem. Rev., 2013, vol. 257, nos 11–12, p. 1699.
Qian, S.-S., Li, H.-H., Zhu, H., et al., Synth. React. Inorg. Met.-Org. Nano-Met. Chem., 2013, vol. 43, no. 4, p. 412.
Moradi-Shoeili, Z., Amini, Z., Boghaei, D.M., et al., Polyhedron, 2013, vol. 53, p. 76.
Li, J.-N., Synth. React. Inorg. Met.-Org. Nano-Met. Chem., 2013, vol. 43, no. 7, p. 826.
Chityala, V.K., Kumar, K.S., Subhashini, N.J.P., et al., J. Coord. Chem., 2013, vol. 66, no. 2, p. 274.
Mukherjee, T., Pessoa, J.C., Kumar, A., et al., Dalton Trans., 2013, vol. 42, no. 7, p. 2594.
Yuan, C.X., Lu, L.P., Gao, X.L., et al., J. Bio. Inorg. Chem., 2009, vol. 14, no. 6, p. 841.
Sonmez, M., Celebi, M., and Berber, I., Eur. J. Med. Chem., 2010, vol. 45, no. 5, p. 1935.
Xue, L.W., Zhao, G.Q., Han, Y.J., et al., Russ. J. Coord. Chem., 2011, vol. 37, no. 4, p. 262.
Xue, L.W., Han, Y.J., Zhao, G.Q., et al., Russ. J. Coord. Chem., 2012, vol. 38, no. 1, p. 24.
Zhang, M., Xian, D.-M., Li, H.-H., et al., Aust. J. Chem., 2012, vol. 65, no. 4, p. 343.
Bruker, SMART and SAINT, Area Detector Control and Integration Software, Madison (WI, USA): Bruker Analytical X-ray Instruments Inc., 1997.
Sheldrick, G.M., SADABS, Program for Empirical Absorption Correction of Area Detector Data, Goöttingen (Germany): Univ. of Göttingen, 1997.
North, A.C.T., Phillips, D.C., and Mathews, F.S., Acta Crystallogr., Sect. A: Found Crystallogr., 1968, vol. 24, no. 3, p. 351.
Sheldrick, G.M., SHELXL-97, Program for the Refinement of Crystal Structures, Göttingen (Germany): Univ. of Göttingen, 1997.
Lal, R.A., Choudhury, S., Ahmed, A., et al., J. Coord. Chem., 2009, vol. 62, no. 23, p. 3864.
Ray, A., Banerjee, S., Butcher, R.J., et al., Polyhedron, 2008, vol. 27, no. 11, p. 2409.
Mandal, S., Saha, R., Mahanti, B., et al., Inorg. Chim. Acta, 2012, vol. 387, no. 1, p. 387.
Ko, H.H., Lim, J.H., Kim, H.C., et al., Inorg. Chem., 2006, vol. 45, no. 22, p. 8847.
Yuan, M., Zhao, F., Zhang, W., et al., Inorg. Chem., 2007, vol. 46, no. 26, p. 11235.
Habibi, M.H., Askari, E., Amirnasr, M., et al., Spectrochim. Acta, A, 2011, vol. 79, no. 3, p. 666.
Vazquez-Fernandez, M.A., Fernandez-Garcia, M.I., Gonzalez-Noya, A.M., et al., Polyhedron, 2012, vol. 31, no. 1, p. 379.
Zhang, N., Huang, C.-Y., Shi, D.-H., et al., Inorg. Chem. Commun., 2011, vol. 14, no. 10, p. 1636.
Barry, A., In: Antibiotics in Laboratory Medicine, Lorian, V., Ed., Baltimore: Williams and Wilkins, 1991.
Rosu, T., Negoiu, M., Pasculescu, S., et al., Eur. J. Med. Chem., 2010, vol. 45, no. 2, p. 774.
Searl, J.W., Smith, R.C., and Wyard, S., J. Proc. Phys. Soc., 1961, vol. 78, no. 505, p. 1174.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Liu, Q.R., Xue, L.W. & Zhao, G.Q. Manganese(III) complexes derived from bis-Schiff bases: Synthesis, structures, and antimicrobial activity. Russ J Coord Chem 40, 757–763 (2014). https://doi.org/10.1134/S1070328414100066
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328414100066