Abstract
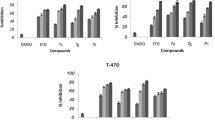
Several derivatives of 6-amino-4-aryl-2-thioxo-1,2,3,4-tetrahydropyrimidine-5-carbonitriles were synthesized via Biginelli type reaction and tested for their anti-proliferative activity on human breast cancer (MCF-7) and human colon carcinoma (HT29) cell lines. Malignant and non-malignant cells were cultivated in RPMI medium and incubated with different concentrations of these pyrimidines. Cell viability was evaluated by MTT assay. Apoptotic cells were determined using DAPI (4'-6-diamidino-2-phenylindole) and propidium iodide staining of DNA fragmentation by flow cytometry (sub-G1 peak). 6-Amino-4-(4-chlorophenyl)-2-thioxo-1,2,3,4-tetrahydropyrimidine-5-carbonitrile and 6-amino-4-[4-dimethylamino)phenyl]-2-thioxo-1,2,3,4-tetrahydropyrimidine-5-carbonitrile decreased the viability of MCF-7 and HT29 cells, in contrast to L929 cells. These compounds induced a sub-G1 peak inflow cytometry histograms of treated cells indicating that apoptosis is involved in their toxicity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
The International Agency for Research on Cancer (IARC) Latest World Cancer Statistics, 2013, December 12. http://www.iarc.fr/en/media-centre/pr/2013/ pdfs/pr223_E.pdf
Goldstein, C.M., Mehren, M.V., Gradel, T., Kilpatrick, D., Vaders, L., Brady, D., and Vogel, L., Breast Cancer Research, Fox Chase Cancer Center Scientific Report, Philadelphia, 2000.
Parkin, D.M., Bray, F., Ferlay, J., and Pisani, P., CA Cancer. J. Clin., 2005, vol. 55, pp. 74–108.
Eidinoff, M.L., Knoll, J.E., Marano, B.J., and Klein, D., Cancer. Res., 1961, vol. 21, pp. 1377–1385.
Callery, P., and Gannett, P., in Cancer and Cancer Therapy, William, D.A. and Lemke, T.L., Eds., Phila delphia, PA, USA: Lippincot Williams and Wilkins, 2002, pp. 934–935.
Donnini, S., Monti, M., Castagnini, C., Solito, R., Botta, M., Schenone, S., Giachetti, A., and Ziche, M., Int. J. Cancer, 2006, vol. 120, pp. 995–1004.
Bazgir, A., Khanaposhtani, M. M., and Soorki, A.A., Bioorg. Med. Chem., 2008, vol. 18, pp. 5800–5803.
Shaban, N.Z., Masoud, M.S., Mawlawi, M.A., Awad, D., and Sadek, O.M., J. Physiol. Biochem., 2012, vol. 68, pp. 475–484.
Sawyer, R.C., Stolfi, R.L., Martin, D.S., and Balis, M.E., Biochem. Pharmacol., 1989, vol. 38, pp. 2305–2311.
Ghoshal, K., and Jacob, S.T., Biochem. Pharmacol., 1997, vol. 53, pp. 1569–1575.
Yang, S., Li, Z., Jin, L., Song, B., Liu, G., Chen, J., Chen, Z., Hu, D., Xue, W., and Xu, R., Bioorg. Med. Chem. Lett., 2007, vol. 17, pp. 2193–2196.
Prachayasittikul, S., Worachartcheewan, A., Nantasenamat, C., Chinworrungsee, M., Sornsongkhram, N., Ruchirawat, S., and Prachayasittikul, V., Eur. J. Med. Chem., 2011, vol. 46, pp. 738–742.
Sharma, S.K., Kumar, P., Narasimhan, B., Ramasamy, K., Mani, V., Mishra, R.K., and Majeed, A.B., Eur. J. Med. Chem., 2012, vol. 48, pp. 16–25.
Taher, A.T. and Helwa, A.A., Chem. Pharm. Bull., 2012, vol. 60, pp. 521–530.
Taher, A.T. and Abou-Seri, S.M., Molecules, 2012, vol. 17, pp. 9868–9886.
Fargualy, A.M., Habib, N.S., Ismail, K.A., Hassan, A.M., and Sarg, M.T., Eur. J. Med. Chem., 2013, vol. 66, pp. 276–295.
Ghodasara, H.B., Trivedi, A.R., Kataria, V.B., Patel, B.G., and Shah, V.H., Med. Chem. Res., 2013, vol. 22, pp. 6121–6128.
Prachayasittikul, S., Sornsongkhram, N., Pingarew, R., Techatanachai, S., Ruchirawat, S., and Prachayasittikul, V., Eur. J. Sci. Res., 2009, vol. 36, pp. 236–245.
Tozkoparan, B., Yarim, M., Saraç, S., Ertan, M., Kelicen, P., Altinok, G., and Demirdamar, R., Arch. der Pharmazie, 2000, vol. 333, pp. 415–420.
Bakavoli, M., Rahimizadeh, M., Shiri, A., Akbarzadeh, M., Mousavi, S. H., Atapour-Mashhad, H., and Tayarani-Najaran, Z., J. Chem. Res., 2010, pp. 403–406.
Bakavoli, M., Rahimizadeh, M., Shiri, A., Akbarzadeh, M., Mousavi, S. H., Tayarani-Najaran, Z., Atapour-Mashhad, H., and Nikpour, M., J. Het. Chem., 2011, vol. 48, pp. 183–187.
Pisani, P., Parkin, D.M., and Ferlay, J., Int. J. Cancer, 1993, vol. 55, pp. 891–903.
Fogh, J., Fogh, J.M., and Orfeo, T., J. Natl. Cancer. Inst., 1977, vol. 59, pp. 221–226.
Navio, J.L.G., Lorente, A., and Soto, J.L., Heterocycles, 1982, vol. 19, pp. 305–311.
Fadda, A.A., El-Latif, E.A., Bondock, S., and Samir, A., Synth. Commun., 2008, vol. 38, pp. 4352–4368.
Jung, M.H., Kim, H., Choi, W.K., El-Gamal, M.I., Park, J.H., Yoo, K.H., Sim, T.B., Lee, S.H., Baek, D., Hah, J.M., Cho, J.H., and Oh, C.H., Bioorg. Med. Chem. Lett., 2009, vol. 19, pp. 6538–6543.
Zhao, L., Chen, Z., Wang, J., Yang, L., Zhao, Q., Wang, J., Qi, Q., Mu, R., You, Q.D., and Guo, QL., Cancer. Chemother. Pharmacol., 2009, vol. 10, pp. 1053–1052.
Yu, B., Shi, X.J., Zheng, Y.F., Fang, Y., Zhang, E., Yu, D.Q., and Liu, H.M., Eur. J. Med. Chem., 2013, vol. 69, pp. 323–330.
Kemnitzer, W., Sirisoma, N., May, C., Tseng, B., Drewe, J., and Cai, S.X., Bioorg. Med. Chem. Lett., 2009, vol. 1, pp. 3536–3540.
Atapour-Mashhad, H., Tayarani-Najaran, Z., Davoodnia, A., Moloudi, R., and Mousavi, S. H., Drug. Chem. Toxicol., 2011, vol. 34, pp. 271–276.
Mousavi, S.H., Tayarani-Najaran, Z., and Hersey, P., Iran. J. Basic. Med. Sci., 2008, vol. 11, pp. 121–142.
Brohem, C.A., Sawada, T.C., Massaro, R.R., Almeida, R.L., Rivelli, D.P., Ropke, C.D., da Silva, V.V., de Lima, T.M., Curi, R., Barros, S.B., and Maria-Engler, S.S., Toxicol. In Vitro, 2009, vol. 23, pp. 111–119.
Mosmann, T., J. Immunol. Meth., 1983, vol. 65, pp. 55–63.
Mousavi, S. H., Tavakkol-Afshari, J., Brook, A., and Jafari-Anarkooli, I., Food. Chem. Toxicol., 2009, vol. 47, pp. 1909–1913.
Mousavi, S.H., Atapour-Mashhad, H., Bakavoli, M., Shiri, A., Akbarzadeh, M., and Tayarani-Najaran, Z., Russ. J. Bioorg. Chem., 2015, vol. 41, pp. 201–208.
Mousavi, S.H., Tavakkol-Afshari, J., Brook, A., and Jafari-Anarkooli, I., Food. Chem. Toxicol., 2009, vol. 47, pp. 855–859.
Tayarani-Najaran, Z., Mousavi, S.H., Asili, J., and Emami, S.A., Food. Chem. Toxicol., 2010, vol. 48, pp. 599–604.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Atapour-Mashhad, H., Soukhtanloo, M., Massoudi, A. et al. Synthesis and evaluation of cytotoxicity of 6-amino-4-aryl-2-thioxo-1,2,3,4-tetrahydropyrimidine-5-carbonitriles. Russ J Bioorg Chem 42, 316–322 (2016). https://doi.org/10.1134/S1068162016020047
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162016020047