Abstract
A seabird (Phalacrocorax carbo) colony overwintering for three consecutive years in a pine forest near the Black Sea coast caused severe damage to the vegetation. The impact of excessive nutrients input from this colony increased soil acidity, N, P, Cu and S-content in soil and might therefore affect soil macrofauna. We compared the abundance of main functional trait guilds and the total abundance of soil macrofauna within impact and control pine forests. No significant difference between the sites was noted in taxonomic richness, total macrofauna abundance and abundance of mobile, belowground, phytophagous and predatory invertebrates. However, the abundance of poorly mobile, poorly mobile epibiontic, and these both traits of saprophagous macrofauna guilds was significantly higher in the control sites. We conclude that three-year wintering of a seabird colony in forest can lead to drastic changes in the macrofauna communities structure due to allochthonous input of nutrients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
The transfer of organic matter from aquatic to terrestrial ecosystems (“aquatic subsidy”) is an important or even main source of nutrients and chemical compounds in terrestrial food web [1–3]. Marine-terrestrial resource flows in most cases are due to wave activity and tidal phenomena. However, an aquatic subsidy may be introduced by living organisms (in the form of excrement, seafood residues, feathers and dead individuals) such as seabirds (e.g., cormorants), the main food of which are marine vertebrates and invertebrates [3–5]. The input and accumulation of allochthonous organic matter during abiotic processes is mainly limited to the supralittoral zone. Seabirds can transfer allochthonous resources to remote ecosystems that may not have direct contact with the sea.
Nesting and overwintering sites of bird colonies, in particular of the great cormorant (Phalacrocorax carbo), are usually confined to coastal cliffs, but can occupy forest areas. Populating of a new habitat is accompanied by the input of allochthonous matter into the terrestrial ecosystem, the amount of which depends on the colony size. For example, great cormorant forms colonies of 50–5000 pairs with nesting density up to 2000 nests/ha. The input of guano and organic residues from such a colony into the soil can lead to an increase in the content of nitrogen and phosphorus, as well as to a change in the soil acidity, salinity, and humidity [6, 7]. Changes in soil characteristics are usually affect the vegetation [3, 7]. Very high guano concentrations can degrade vegetation [8, 9], due to the toxicity of excessive ammonium and increased soil acidity [6, 10]. Changes in soil properties and vegetation cover cannot but affect animal communities [11, 12], including soil invertebrates. However, insufficient attention has been paid to this issue. Previous studies are usually limited to areas of permanent concentrations of seabirds and focus on specific taxonomic groups of soil invertebrates (e.g., [13–15]) and parasitic organisms (e.g., [16, 17]). We suggest that populating of a new area by birds should lead to a reduction in the abundance and taxonomic diversity of soil invertebrates, as well as affect the structure of invertebrate communities due to changes in soil properties and plant conditions.
This work aims to assess seabird impact on the structure of soil macrofauna communities in coastal forest ecosystems. Forest ecosystems of Utrish State Nature Reserve separated from the sea by cliff were chosen for the study. A three–year overwintering of the great cormorant (P. carbo) colony in these ecosystems led to the abundant accumulation of excrements and drying out of the trees [18].
MATERIAL AND METHODS
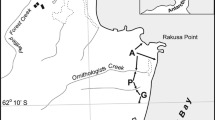
The material was collected in June 2018 at two sites of bird colonies overwintering (hereinafter referred to as impact sites) and at two control sites (44°43ʹ06 N, 37°26ʹ36 E) in Anapa district of Krasnodar krai near Maly Utrish Cape. Nesting of the great cormorant in the winter period in the studied area has been observed since 2015 (according to Nature Reserve monitoring data). The sites were located on a cliff about 13-m high. The vegetation in impact and control sites was represented by xerophytic communities of the Sub–Mediterranean type [19] with prevalence of Pinus brutia var. pityusa and single trees of Juniperus excelsa, Carpinus orientalis, and Quercus pubescens; the shrub layer was represented by Ruscus ponticus; the grass layer was dominated by cereals Hordeum leporinum and Bromus sterilis. The projective grass cover of the impact sites reached 80%, within the control sites, it did not exceed 20%. The litter depth within the impact and control sites was similar (Table 1). However, visually in the impact sites the needle litter prevailed. The soil and plants of the impact sites were covered with bird guano feathers, and food residues (fish, scales, and bones); tree and shrub vegetation was strongly depressed and desiccated (see also [18]).
Within each site five soil samples were randomly collected a corer of 20 cm in diameter, down to the depth of 15 cm. To avoid the edge effect, sampling was carried out in the central part of the site at a 10 × 10 m area. At each site, five samples of leaf litter of dominating tree spices (P. pityusa and Q. pubescens) and cormorant feces were collected, along with five samples of algae Cystoseira barbata (directly from the sea). Then the samples were delivered in isothermal containers at a temperature of 9 ± 1 °C to the laboratory of IEE RAS (Moscow). Animals were extracted for seven days using Tullgren extractors. A solution of alcohol, water, and ethylene glycol at a ratio of 80 : 15 : 5 was used as the fixing liquid. The abundance of soil macrofauna was standardized to m2 (individuals per m2, further ind./m2). All taxa were identified to a family level with the exception of Diptera pupae, Pseudoscorpionida and juvenile individuals of Polyxenida, which were identified to the level of order. In total, 2096 individuals of macrofauna were collected and identified (Table 2). Based on published data [20, 21], animals were allocated to different functional traits [22] according to their feeding preferences (saprophages, predators, and herbivores), predominant vertical distribution (aboveground and belowground) and mobility (relatively mobile and predominantly resident).
Each soil sample was processed in the laboratory to determine following parameters: pH of the soil solution (H2O, ratio 1 : 2.5) and the total content (mg/kg) of Al, P, S, K, Ca, Cr, Mn, Fe, Cu using X-ray fluorescence analyzer S2 PICOFOX, Bruker. The total C and N content in the soil (%) and isotopic the carbon (13C/12C, usually expressed as a standardized ratio δ13C) and nitrogen (15N/14N, usually expressed as δ15N) stable isotope ratios of soil, leaf litter, algae, and P. carbo guano were determined using a Thermo Scientific Delta V Plus continuous-flow mass spectrometer (Thermo Electron, Germany), coupled with a Flash 1112 elemental analyzer (Thermo Electron) at “Instrumental Methods in Ecology” at the Severtsov Institute of Ecology and Evolution, Russian Academy of Sciences.
Soil samples taken within each study site were considered pseudo-replicates and were only used to form a mean value per site. In this regard, the abundance of soil macrofauna is given as the mean between the two sites, for both control (n = 2) and impact (n = 2) treatments ± standard error of the mean (SE). We applied Nested Design ANOVA (sample nested in site) with subsequent control of the false deviations frequency using the Benjamini-Yekutieli algorithm [23] to evaluate differences between control and impact sites in total abundance of soil macrofauna, abundance of different trait combinations, taxonomic diversity and soil parameters. Prior to the analysis data of macrofauna abundance were log-transformed (ln(x+1)).
The dependence of the taxa abundance on soil parameters was evaluated by principal component analysis (PCA) for log-transformed data, n = 20 samples. Soil parameters were chosen as active variables, the abundance of animals as supplementary (passive) variables, and the site type was chosen as a grouping variable. Taxa with the occurrence of at least four per 20 samples were used for analysis.
Statistical data processing was performed using Statistica 13.3 (TIBCO Software, United States).
RESULTS
A total of 40 and 34 taxa were found within control and impact sites respectively (Table 2). However, the taxonomic diversity between sites (13.6 ± 1.6 and 12.5 ± 2.3 taxa per site) did not differ significantly (F = 1.3, p = 0.26).
Total abundance of soil macrofauna did not differ significantly between the control and impact sites (Fig. 1). However, the abundance of aboveground resident groups (e.g., Pseudoscorpiones, Lithobiomorpha, and Juliformia) was significantly lower in impact sites. Among the trophic groups, the abundance of saprotrophic invertebrates was lower in the impact sites compared to the control ones (417.6 ± 252.9 and 826.5 ± 8.8 ind./m2 in the impact and control areas respectively). However, the presence of birds had a statistically significant negative impact only on the abundance of resident and aboveground resident saprophages (primarily Diplopoda and Isopoda) (Fig. 1). The abundance of belowground invertebrates did not differ significantly between the control and impact sites (F = 3.0, p = 0.9). However the abundance of belowground mobile predators (predatory Diptera larvae and Embia (Fig. 1), but statistically insignificant (F = 6.8, p = 0.2).
Soil invertebrates abundance (mean ± SE, n = 2) on control (1) and disturbed (2) sites: Total—total abundance of soil macrofauna, A—aboveground, B—belowground, M—mobile, R—resident, P—predators, S—saprophages; *—statistically significant differences between control and impact sites using analysis of variance.
The soils of the impact sites in comparison with the control were characterized by an acid reaction (Table 1, Fig. 2), a high content of nitrogen, phosphorus, sulfur, chromium, and copper. In general, the presence of birds positively correlated with the abundance of a number of invertebrate taxa (Fig. 2), for example, Thripidae, Pyrrhocoridae and predatory Coleoptera (Staphylinidae, Carabidae and Cantharidae). The increase in the abundance of the latter group also correlated with the increase in phosphorus and copper content in the soil. Increasing amounts of nitrogen in the soil were positively correlated with the abundance of larvae of Platypezidae, but negatively affected the abundance of Cicadellidae, adult Polyxenidae, and the larvae of Cecidomyidae. Soil of the control sites characterized by significantly higher values of aluminum and magnesium (Table 1). The abundance of Polyzoniidae and Pseudoscorpionida was positively correlated with its content, while Julida and juvenile Gnaphosidae positively correlated with pH. Litter depth and the content of carbon, iron and calcium in the soil did not differ significantly between sites (Table 1). A close correlation (Fig. 2) was revealed only between the calcium content in soil and the abundance of juvenile Polyxenida.
Ordination diagram showing results of the principal component analysis (PCA) the relationship between soil invertebrates taxa abundance (showed as lines and diamond-shaped symbols) and soil parameters (black lines and dots). 1—Thripidae, 2—Cicadellidae, 3—Pyrrhocoridae, 4—Carabidae, 5—Staphylinidae, 6—Cantharidae, 7—Gnaphosidae, 8—Pseudoscorpionida, 9—Trachelipodidae, 10—Geophilidae, 11—Lithobiidae, 12—juvenile Polyxenida, 13—Polyxenidae, 14—Lophoproctidae, 5—Julidae, 16—Polyzoniidae, 17—Oligotomidae, 18—Cecidomyiidae, 19—Diaspididae, 20—Asilidae, 21—Platypezidae, 22—Stratiomyiidae, 23—Diptera pupae.
According to the isotope analysis, the δ13C and δ15N values of soils in impact sites were significantly higher than in the control in The isotopic signatures of macrophytes (C. barbata) and bird guano were also significantly higher compared to the terrestrial leaf litter (see Table 1).
DISCUSSION
The data only partly confirmed our hypothesis that the excessive input of the marine organic matter into previously unaffected ecosystems would have a negative impact on the total abundance and taxonomic diversity of soil invertebrates. Both indicators did not differ significantly between the impact and control sites. Analysis of published data also showed contradictory information. On the one hand, studies conducted e.g. on the islands of Sweden [10, 15] showed a decrease in the abundance of soil animals in disturbed by seabird bird colonies sites compared to control sites, despite the increase in abundance of some groups (e.g. Astigmata). On the other hand, on islands of the Gulf of California [2, 4, 13, 24], New Zealand and Australia [25, 26] colonized by seabirds, the abundance of phytophages, saprophages, and predatory invertebrates was significantly higher than on islands without bird colonies. However, the mentioned studies concerned a longer period of the seabird colonies presence in comparison with the ones studied by us.
Nevertheless, we have found statistically significant reduction in the abundance of all resident groups, as well as epibiotic saprophages, which inhabit the litter and upper soil layers. The reduction in an abundance of the above groups could be explained as a reaction to the increased content of nitrate (\({\text{NO}}_{3}^{ - }\)) in the litter layer and ground vegetation, the source of which is the guano of cormorants. The increase in soil nitrogen, phosphorus and sulfur content as a result of seabird guano input is consistent with the results of earlier studies [4, 8, 10, 27] and own isotope analysis data. The marine organic matter is characterized by a high content of 13C and 15N [3, 28], due to which the values of δ13C and δ15N of the soil of the impact sites are significantly higher than in the control sites. We also found a significant increase in the amount of chromium and copper in the soil of impact sites, which, according to previous researches [29], may be associated with bioaccumulation of elements in the fish bodies, which are the main object of food for cormorants, as well as in the birds themselves. A significant reduction in the aluminum content in the soils of the impact sites may be caused by a methodological flaw: the interaction of the bird guano with substrates containing many amorphous Fe and Al compounds leads to the formation of the tarankite mineral [30]. These mineral fractions during sample preparation and sifting of the soil were probably excluded from further analysis.
Low abundance of aboveground saprotrophic animals in impact sites can also be caused by a decrease in the quality of incoming litter [31, 32]. The death of trees, primarily the dominants of the tree layer (P. pityusa) led to a sharp, abundant fall of coniferous litter [18], the decomposition of which by soil organisms is much slower than that of the leaf litter [31, 32]. Probably, annual overwintering of the cormorant within the same area is a factor enhancing the negative impact on resident groups of animals due to the reduction of the time interval for the possibility of restoration in the territory [21]. On the contrary, the abundance of all mobile groups did not significantly differ between impact and control sites due to the high activity of movement, including through disturbed sites, which are very local with respect to the area of the entire forest ecosystem [20].
The absence of differences in the abundance of belowground animals between the sites may be due to the process of sedimentation and drying of bird guano mainly on ground vegetation and litter layer. This prevents the further penetration of excess nitrates into the lower soil layers, which is also aided by the arid climate and low precipitation level in the region [19]. The abundance of coniferous litter, high projective cover of the grass layer, and the decrease in evaporation due to an increase in the albedo of areas covered by bird guano, have so far leveled out possible changes in soil moisture, which could be caused by a reduction in shading of the sites via death of the tree layer. However, this needs further verification.
The abundance of phytophages also did not differ significantly between control and impact sites, which is largely caused by the rapid growth of xerophytic cereals in the grass layer due to an increase in the content of phosphorus and nitrogen in the soil. A decrease in the projective cover of crowns [33, 34] as well as with a simultaneous decreased pressure of aboveground predators (e.g., spiders).
CONCLUSIONS
The results obtained generally confirm and supplement the data of previous studies. Overwintering of the great cormorant colonies led to drastical changes in soil and vegetation conditions of the studied forest ecosystems and, as a result, to significant changes in the structure of soil invertebrate communities. Despite the absence of significant differences in the total abundance and taxonomic diversity of soil macrofauna between control and impact sites, the analysis of functional traits allowed us to observe a significant decrease in the abundance of resident and aboveground saprophages. This makes the above approach a promising tool for studying ecosystem disturbances, including forecasting and impact assessment.
It should be noted that our study is preliminary and was conducted for the first time both in forest ecosystems that had not previously been affected by seabirds, and on the Black Sea coast as a whole. Accordingly, the results do not allow us to clearly postulate the patterns of changes in the abundance and structure of invertebrate communities with such ecosystem disturbances. The question raised obviously requires further research, including increasing the sample size, the number of sampling sites and the studied groups of animals (e.g., chortobiontic animals, soil micro- and mesofauna).
REFERENCES
Collier, K.J., Bury, S., and Gibbs, M., A stable isotope study of linkages between stream and terrestrial food webs through spider predation, Freshwater Biol., 2002, vol. 47, pp. 1651–1659. https://doi.org/10.1046/j.1365-2427.2002.00903.x
Polis, G.A., Anderson, W.B., and Holt, R.D., Toward an integration of landscape ecology and food web ecology: The dynamics of spatially subsidized food webs, Annu. Rev. Ecol. Syst., 1997, vol. 28, pp. 289–316. https://doi.org/10.1146/annurev.ecolsys.28.1.289
Anderson, W.B. and Polis, G.A., Marine subsidies of island communities in the Gulf of California: evidence from stable carbon and nitrogen isotopes, Oikos, 1998, vol. 81, pp. 75–80. https://doi.org/10.2307/3546469
Polis, G.A. and Hurd, S.D., Allochthonous input across habitats, subsidized consumers, and apparent trophic cascades: examples from the ocean–land interface, in Food Webs: Integration of Patterns and Dynamics, Polis, G.A. and Winemiller, K.O., Eds., New York: Chapman and Hall, 1996, pp. 275–285.
Polis, G.A. and Hurd, S.D., Linking marine and terrestrial food webs: allochthonous input from the ocean supports high secondary productivity on small islands and coastal land communities, Am. Nat., 1996, vol. 147, pp. 396–423. https://doi.org/10.1086/285858
Havik, G., Catenazzi, A., and Holmgren, M., Seabird nutrient subsidies benefit non-nitrogen fixing trees and alter species composition in South American coastal dry forests, PLoS One, 2014, vol. 9, no. 1, e86381. https://doi.org/10.1371/journal.pone.0086381
Kolb, G.S., Jerling, L., Essenberg, C., et al., The impact of nesting cormorants on plant and arthropod diversity, Ecography, 2012, vol. 35, pp. 726–740. https://doi.org/10.1111/j.1600-0587.2011.06808.x
Breuning-Madsen, H., Ehlers, C.B., and Borggaard, O.K., The impact of perennial cormorant colonies on soil phosphorus status, Geoderma, 2008, vol. 148, pp. 51–54. https://doi.org/10.1016/j.geoderma.2008.09.002
McGrath, D.M. and Murphy, S.D., Double-crested cormorant (Phalacrocorax auritus) nesting effects on understory composition and diversity on island ecosystems in Lake Erie, Environ. Manage., 2012, vol. 50, pp. 304–314. https://doi.org/10.1007/s00267-012-9878-6
Kolb, G.S., Palmborg, C., Taylor, A.R., et al., Effects of nesting cormorants (Phalacrocorax carbo) on soil chemistry, microbial communities and soil fauna, Ecosystems, 2015, vol. 18, pp. 643–657. https://doi.org/10.1007/s10021-015-9853-1
Balčiauskas, L., Balčiauskienė, L., and Jasiulionis, M., Mammals under a colony of great cormorants: Population structure and body condition of yellow-necked mice, Turk. J. Zool., 2015, vol. 39, no. 5, pp. 941–948. https://doi.org/10.3906/zoo-1407-27
Balčiauskas, L., Skipitytė, R., Jasiulionis, M., et al., Immediate increase in isotopic enrichment in small mammals following the expansion of a great cormorant colony, Biogeosciences, 2018, vol. 15, pp. 3883–3891. https://doi.org/10.5194/bg-15-3883-2018
Duffy, D., Ants, ticks, and nesting seabirds: Dynamic interactions, in Bird–Parasite Interactions, Loye, J. and Zuk, M., Eds., New York: Oxford Univ. Press, 1991, pp. 242–257.
Zmudczyńska, K., Olejniczak, I., Zwolicki, A., et al., Influence of allochtonous nutrients delivered by colonial seabirds on soil collembolan communities on Spitsbergen, Polar Biol., 2012, vol. 35, pp. 1233–1245. https://doi.org/10.1007/s00300-012-1169-4
Kolb, G.S., Jerling, L., and Hambäck, P.A., The impact of cormorants on plant-arthropod food webs on their nesting islands, Ecosystems, 2010, vol. 13, pp. 353–366. https://doi.org/10.1007/s10021-010-9323-8
Kanarek, G., Sitko, J., Rolbiecki, L., and Rokicki, J., Digenean fauna of the great cormorant Phalacrocorax carbo sinensis (Blumenbach, 1798) in the brackish waters of the Vistula Lagoon and the Gulf of Gdansk (Poland), Wiad Parazytol., 2003, vol. 49, pp. 293–299.
Chaika, K.V., The role of great cormorant (Phalacrocorax carbo L.) in regulation of bioresources of the Curonian Bay ecosystem, Cand. Sci. (Biol.) Dissertation, Kaliningrad, 2017.
Dzhangirov, M.Yu. and Suvorov, A.V., Curresnt state of Pitsunda pine (Pinus pityuosa Steven) phytocenoses in the Utrish Nature Reserve, in Okhrana bioty v gosudarstvennom prirodnom zapovednike “Utrish” (Conservation of the Biota in the Utrish State Nature Reserve), Maikop, 2015, vol. 3, pp. 129–141.
Seregin, A.P. and Suslova, E.G., Vascular plant flora in the environs of Malyi Utrish village, in Landshaftnoe i biologicheskoe raznoobrazie Severo-Zapadnogo Kavkaza (Landscape and Biological Diversity in the Northwestern Caucasus), Moscow: Mosk. Gos. Univ., 2007, pp. 104–174.
Zaitsev, A.S., Gongalsky, K.B., Bengtsson, J., and Persson, T., Connectivity of litter islands remaining after a fire and unburnt forest determines the recovery of soil fauna, Appl. Soil. Ecol., 2014, vol. 83, pp. 101–108. https://doi.org/10.1016/j.apsoil.2014.01.007
Korobushkin, D.I., Gorbunova, A.Yu., Zaitsev, A.S., and Gongalsky, K.B., Trait-specific response of soil macrofauna to forest burning along a macrogeographic gradient, Appl. Soil Ecol., 217, vol. 112, pp. 97–100. https://doi.org/10.1016/j.apsoil.2016.12.004
Wong, M.K.L., Guenard, B., and Lewis, O., Trait-based ecology of terrestrial arthropods, OSF Preprints, 2018. https://doi.org/10.31219/osf.io/bvq2j.
Benjamini, Y. and Yekutieli, D., The control of the false discovery rate in multiple testing under dependency, Ann. Stat., 2001, vol. 29, pp. 1165–1188. https://doi.org/10.1214/aos/1013699998
Sanchez Piñero, F. and Polis, G.A., Bottom-up dynamics of allochthonous input: direct and indirect effects of seabirds on islands, Ecology, 2000, vol. 81, pp. 3117–3132. https://doi.org/10.1890/0012-9658(2000)081[3117:budoai]2.0.co;2
Daugherty, C.H., Towns, D.R., Atkinson, I.A.E., and Gibbs, G.W., The significance of the biological resources of New Zealand islands for ecological restoration, in Ecological Restoration of the New Zealand Islands, Towns, D.R. and Daugherty, C.H., Eds., IAE Atchinson, 1990, pp. 9–21.
Towns, D.R., Wardle, D.A., Mulder, C.P., et al., Predation of seabirds by invasive rats: Multiple indirect consequences for invertebrate communities, Oikos, 2009, vol. 118, pp. 420–430. https://doi.org/10.1111/j.1600-0706.2008.17186.x
Birkhofer, K., Schöning, I., Alt, F., et al., General relationships between abiotic soil properties and soil biota across spatial scales and different land-use types, PLoS One, 2012, vol. 7, no. 8, p. e43292. https://doi.org/10.1371/journal.pone.0043292
Korobushkin, D.I., Role of allochthonous carbon in the energy of terrestrial invertebrate communities at different distances from the Black Sea and a freshwater lake (isotopic evidence), Russ. J. Ecol., 2014, vol. 45, no. 3, pp. 223–230. https://doi.org/10.1134/S1067413614030060
Rial, D., Santos-Echeandia, J., Alvarez-Salgado, X.A., et al., Toxicity of seabird guano to sea urchin embryos and interaction with Cu and Pb, Chemosphere, 2016, vol. 145, pp. 384–393. https://doi.org/10.1016/j.chemosphere.2015.11.064
Lindsay, W.L., Chemical Equilibria in Soils, New York: Wiley, 1979.
Striganova, B.R., Pitanie pochvennykh saprofagov (Nutrition of Soil Macrophages), Moscow: Nauka, 1980.
Berg, B., Berg, M.P., Bottner, P., et al., Litter mass loss rates in pine forests of Europe and eastern United States: some relationship with climate and litter quality, Biogeochemistry, 1993, vol. 20, pp. 127–159. https://doi.org/10.1007/BF00000785
Wainright, S.C., Haney, J.C., Kerr, C., et al., Utilization of nitrogen derived from seabird guano by terrestrial and marine plants at St. Paul, Pribilof Islands, Bering Sea, Alaska, Mar. Biol., 1998, vol. 131, pp. 63–71. https://doi.org/10.1007/s002270050297
Ellis, J.C., Marine birds on land: A review of plant biomass, species richness, and community composition in seabird colonies, Plant Ecol., 2005, vol. 181, pp. 227–241. https://doi.org/10.1007/s11258-005-7147-y
ACKNOWLEDGMENTS
We are grateful to Deputy Director of the Utrish State Nature Reserve O.N. Bykhalova for assisting in the organization of field work, as wel as to Yu.M. Lebedev for his help in sampling and extraction of animals, K.B. Gongalsky for identification of woodlice, coleopterans, and dipteran larvae, and R.R. Seifulina for the identification of spiders.
Funding
Statistical data processing and identification of taxa were carried out using software and equipment funded by Russian Foundation of Basic Research, project no. 16-14-00096. The study was supported by Russian Foundation for Basic Research, project 18-34-00573 mol_a.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest. The authors declare that they have no conflict of interest.
Statement of the welfare of animals. This article does not contain any studies involving animals or human participants performed by any of the authors.
Additional information
Translated by S. Kuzmin
Rights and permissions
About this article
Cite this article
Korobushkin, D.I., Saifutdinov, R.A. Influence of Seabird Colonies on Soil Macrofauna Communities at the Black Sea Coast Forests. Russ J Ecol 50, 567–573 (2019). https://doi.org/10.1134/S1067413619060080
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1067413619060080