Abstract
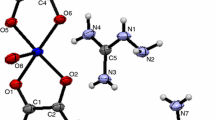
A new uranyl oxalate complex containing aminoguanidinium cation was isolated from an aqueous solution containing uranyl nitrate hexahydrate, oxalic acid dihydrate, and aminoguanidinium bicarbonate in appropriate ratio. The compound was characterized by analytical, spectral (UV-Vis and IR), and thermal (TG-DTA) techniques. The X-ray crystallographic study shows that the complex has a polymeric structure with two types of coordination around the alternative uranyl ions. The oxalate ions exhibit tetradentate bridging and tri-dentate bridging modes. The aminoguanidinium cation is in the outer sphere, acting as charge-neutralizing species. An intense photoluminescence peak is observed at 519 run. The thermal decomposition of the complex yields U3O8. SEM photographs of U3O8 show the presence of irregularly shaped agglomerated particles of sub-micron size.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Katz, J.J., Morss, L.R., and Seaborg, G.T., The Chemistry of the Actinide and Transactinide Elements, Dordrecht: Springer, 2011, vols. 1–6.
Moore, K.T. and Vanderlaan, G., Rev. Mod. Phys., 2009, vol. 81, pp. 235–298.
Ephritikhine, M., Dalton Trans., 2006, pp. 2501–2516.
MacDonald, M.R., Fieser, M.E., Bates, J.E., et al., J. Am. Chem. Soc., 2013, vol. 135, pp. 13310–13313.
Ferri, D., Iuliano, M., Manfredi, C., et al., J. Chem. Soc, Dalton Trans., 2000, pp. 3460–3466.
Poojary, M.D. and Patil, K.C., Proc. Indian Acad. Sci., 1987, vol. 99, pp. 311–315.
Chapelet-Arab, B., Nowogrocki, G., Abraham, F., and Grandjean, S., Radiochim. Acta, 2005, vol. 93, pp. 279–286.
Baeva, E.E., Mikhailov, Yu.N., Gorbunova, Yu.E., et al., Russ. J. Inorg. Chem., 2002, vol. 47, no. 9, pp. 1348–1356.
Subramani, B., Sivasankar, B.N., and Wilfred Sugumar, R., Int. J. Chem., 2014, vol. 3, no. 1, pp. 27–34.
Ragul, R. and Sivasankar, B.N., Sci. Rev. Chem. Commun., 2013, vol. 3, no. 1, pp. 81–93.
Kuppusamy, K., Sivasankar, B.N., and Govindarajan, S., Thermochim. Acta, 1995, vol. 259, pp. 251–262.
Sivasankar, B.N., Sharmila, J.R., and Ragunath, L., Synth. React. Inorg. Met.-Org. Chem., 2004, vol. 34, pp. 1787–1800.
Vikram, L. and Sivasankar, B.N., J. Therm. Anal. Calorim., 2008, vol. 91, pp. 963–970.
Vikram, L. and Sivasankar, B.N., Indian J. Chem., 2007, vol. 46, pp. 568–575.
Erdey, L. and Buzaz, I., Gravimetric Analysis, London: Pergamon, 1965, part II.
Vogels, A.I., A Textbook of Quantitative Inorganic Analysis, London: Longmans, 1962, 3rd ed.
Johnson, C.K., ORTEP ORNL-3794, Tennessee: Oak Ridge National Laboratory, 1976.
Sheldrick, G.M., SHELXL-2014 Programs for Crystal Structure Determination, England: Univ. of Cambridge, 2015.
McGlynn, S.P. and Smith, J.K., J. Mol. Spectrosc, 1961, vol. 6, pp. 164–187.
Saveleva, Z.A., Romanenko, G.V., Sheludyakova, L.A., and Larionov, S.V., Polyhedron, 2000, vol. 17, pp. 1737–1740.
Ugono, O., Rath, N.P., and Beatty, A.M., Cryst. Growth Des., 2009, vol. 9, pp. 4595–4598.
Packiaraj, S., Pushpaveni, A., Govindarajan, S., and Rawson, J.M., CrystEngComm, 2016, vol. 18, pp. 7978–7993.
Klampfer, P., Benkic, P., Ponikvar, M., et al., Monatsh. Chem., 2003, vol. 134, pp. 1–9.
Selvakumar, R., Geib, S.J., Premkumar, T., and Govindarajan, S., Polyhedron, 2015, vol. 87, pp. 321–328.
Selvakumar, R., Geib, S.J., Premkumar, T., and Govindarajan, S., J. Therm. Anal, Calorim., 2016, vol. 124, pp. 375–385.
Bell, J.T. and Biggers, R.E., J. Mol. Spectrosc, 1968, vol. 25, pp. 312–329.
Lauren, A.B. and Christopher, L.C., Cryst. Growth Des., 2006, vol. 6, pp. 2248–2259.
Zhai, X.S., Zhu, W.G., Xu, W., et al., CrystEngComm, 2015, vol. 17, pp. 2376–2388.
Acknowledgments
One of the authors, (T.M. Ahamed Hussain) is grateful to the University Grants Commission, New Delhi for RGNFD fellowship (Ref. no. F/2016-17/RGNFD-2016-17-TAM-2463).
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Ahamed Hussain, T.M., Sivasankar, B.N. Synthesis, Characterization, and X-ray Structure of Tetraaminoguanidinium Diuranyl Tetraoxalate Monohydrate (HAgun)4[(UO2)2(C2O4)4]·H2O. Radiochemistry 61, 434–442 (2019). https://doi.org/10.1134/S1066362219040064
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1066362219040064