Abstract
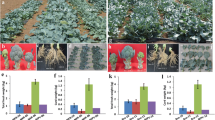
Vernalization plays a key role in the bolting and flowering of Chinese cabbage (Brassica rapa L. ssp. pekinensis). Plants can switch from vegetative to reproductive growth and then bolt and flower under low temperature induction. The economic benefits of Chinese cabbage will decline significantly when the bolting happens before the vegetative body fully grows due to a lack of the edible value. It was found that continuous seedling breeding reduced the heading of Chinese cabbage and led to bolt and flower more easily. In the present study, two inbred lines, termed A161 and A105, were used as experiment materials. These two lines were subjected to vernalization and formed four types: seeds-seedling breeding once, seedling breeding twice, seedling breeding thrice and normal type. Differences in plant phenotype were compared. DNA methylation analysis was performed based on MSAP method. The differential fragments were cloned and analyzed by qPCR. Results showed that plants after seedling breeding thrice had a loosen heading leaves, elongated center axis and were easier to bolt and flower. It is suggested that continuous seedling breeding had a weaker winterness. It was observed that genome methylation level decreased with increasing generation. Four differential genes were identified, short for BraAPC1, BraEMP3, BraUBC26 and BraAL5. Fluorescent qPCR analysis showed that expression of four genes varied at different reproduction modes and different vernalization time. It is indicated that these genes might be involve in the development and regulation of bolting and flowering of plants. Herein, the molecular mechanism that continuous seedling breeding caused weaker winterness was analyzed preliminarily. It plays an important guiding significance for Chinese cabbage breeding.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Lin, S. I., Wang, J. G., Poon, S. Y., et al., Differential regulation of FLOWERING LOCUS C expression by vernalization in cabbage and Arabidopsis, Plant Physiol., 2005, vol. 137, no. 3, pp. 1037–1048.
Greenup, A., Peacock, W. J., Dennis, E. S., and Treyaskis, B., The molecular biology of seasonal floweringresponses in Arabidopsis and the cereals, Ann. Bot., 2009, vol. 103, pp. 1165–1172.
Yuan, Y. X., Wu, J., Sun, R. F., et al., A naturally occurring splicing site mutation in the Brassica rapa FLC1 gene is associated with variation in flowering time, J. Exp. Bot., 2009, vol. 60, no. 4, pp. 1299–1308.
Lou, P., Xie, Q., Xu, X., et al., Genetic architecture of the circadian clock and flowering time in Brassica rapa, Theor. Appl. Genet., 2011, vol. 123, no. 3, pp. 397–409.
Turnbull, C., Long-distance regulation of flowering time, J. Exp. Bot., 2011, vol. 62, no. 13, pp. 4399–4413.
Burn, J. E., Bagnall, D. J., Metzger, J. D., et al., DNA methylation, vernalization, and the initiation of flowering, Proc. Natl. Acad. Sci. U. S. A., 1993, vol. 90, no. 1, pp. 287–291.
Li, M. L., Wang, Q. M., and Zhu, Z. J., Effects of vernalization on DNA methylation, content of GAand protein in Chinese cabbage, Acta Hort. Sin., 2002, vol. 29, no. 4, pp. 353–357.
Finnegan, E. J., Genger, R. K., Kovac, K., et al., DNA methylation and the promotion of flowering by vernalization, Proc. Natl Acad. Sci. U. S. A., 1998, vol. 95, pp. 5824–5829.
Shen, J. Y., Hou, P., Ji, M. J., et al., Progresses in DNA methylation analysis, Chem. Life, 2003, vol. 23, no. 2, pp. 149–151.
Qu, X., Elimination of Potato Virus and Genetic Variation by Low Temperature, Henan Univ. (in Chinese), 2012.
Pan, Y. J., Fu, B. Y., Wang, D., et al., Spatial and temporal profiling of DNA methylation induced by drought stress in rice, Sci. Agric. Sin., 2009, vol. 42, no. 9, pp. 3009–3018.
Fan, J. C., Liu, B., Wang, J. Y., and Sheng, L. X., Effect of napthalene on the DNA methylation patterns and level in rice (Oryza sativa L.), Environ. Sci., 2010, vol. 31, no. 3, pp. 793–800.
Xiong, W. S., Studies on the Relationship of DNA Methylation with Heterosis in Oilseed Rape, Southwest Univ. (in Chinese), 2009.
Meng, H. B., Du, X., Jiang, Y. X., et al., Comparison between tetraploid turnip (Brassica rapa) and its diploid progenitor of DNA methylation under cadmium stress, J. Nucl. Agric. Sci., 2010, vol. 24, no. 6, pp. 1297–1304.
Bai, X. L., Genomic DNA Methylation in Maize Association with Heterosis, Shaanxi Normal Univ. (in Chinese), 2008.
Xiong, L. Z., Xu, C. G., Saghai, M. A., and Zhang, Q., Patterns of cytosine methylation in an elite rice hybrid and its parental lines, detected by a methylation-sensitive amplification polymorphism technique, Mol. Gen. Genet., 1999, vol. 261, pp. 439–446.
Ezio, P., Alberto, A., Cinzia, C., and Sergio, L., Analysis of DNA methylation during germination of pepper (Capsicum annuum L.) seeds using methylation-sensitive amplification polymorphism (MSAP), Plant Sci., 2004, vol. 166, no. 1, pp. 169–178.
Sibum, S. and Richard, M. A., Vernalization, epigenetics, how plants remember winter, Curr. Opin. Plant Biol., 2004, no. 7, pp. 4–10.
He, Y. X. and Wang, Z. C., Genetic variation of DNA methylation in Arabidopsis seedlings after cryopreservation, Chin. Bull. Bot., 2009, vol. 44, no. 3, pp. 317–322 (in Chinese).
Dong, F. C. and Song, C. P., Ubiquitin and its physiological function in plant cells, Plant Physiol. Commun., 1999, vol. 35, no. 1, pp. 54–59 (in Chinese).
Xu, L., Ménard, R., Berr, A., et al., E2 ubiquitin-conjugating enzymes, AtUBC1 and AtUBC2, play redundant roles and are involved in activation of FLC expression and repression of flowering in Arabidopsis thaliana, Plant J., 2009, vol. 57, no. 2, pp. 279–288.
Lau, O. S. and Deng, X. W., Effect of Arabidopsis COP10 ubiquitin E2 enhancement activity across E2 families and functional conservation among its canonical homologues, Biochem. J., 2009, no. 418, pp. 683–690.
Yang, D. L., Yao, J., Mei, C. S., et al., Plant hormone jasmonate prioritizes defense over growth by interfering with gibberellin signaling cascade, Proc. Natl Acad. Sci. U. S. A., 2012, vol. 109, no. 19, pp. E1192–E1200.
Jiang, Z. W., Liu, X. G., and Zhou, Z. J., The regulation of histone modification, Prog. Biochem. Biophys., 2009, vol. 36, no. 1, pp. 1252–1259 (in Chinese).
Wang, W., Meng, Z. Q., and Shi, F. X., Modification and biological role of histone, Hereditas (Beijing), 2012, vol. 34, no. 7, pp. 810–818.
Yun, M., Wu, J., Jerry, L. W., and Li, B., Readers of histone modifications, Cell Res., 2011, vol. 21, no. 4, pp. 564–578.
Lee, W. Y., Lee, D., Chung, W. I., and Kwon, C. S., Arabidopsis ING and Alfin1-like protein families localize to the nucleus and bind to H3K4me3/2 via plant homeodomain fingers, Plant J., 2009, vol. 58, no. 3, pp. 511–524.
Song, J. H. and Zhang, L. X., Progress on the transmembrane protein in plants, J. Biol., 2009, vol. 26, no. 6, pp. 62–64 (in Chinese).
Inan, G., Zhang, Q., Li, P. H., et al., A halophyte and cryophyte Arabidopsis relative model system and its applicability to molecular genetic analyses of growth and development of extremophiles, Plant Physiol., 2004, no. 135, pp. 1718–1737.
He, Y., Doyle, M. R., and Amasino, R. M., PAF1-complex-mediated histone methylation of FLOWERING LOCUS C chromatin is required for the vernalizationresponsive, winter-annual habit in Arabidopsis, Genes Dev., 2004, no. 18, pp. 2774–2784.
Song, Y., Gao, J., Yang, F., et al., Molecular evolutionary analysis of the Alfin-like protein family in Arabidopsis lyrata, Arabidopsis thaliana, and Thellungiella halophila, PLoS One, 2013, vol. 8, no. 7. e66838
Gao, C. L., Yu, C. K., Qu, S., et al., The Golgi-localized Arabidopsis endomembrane protein12 contains both endoplasmic reticulum export and Golgi retention signals at its C terminus, Plant Cell, 2012, vol. 24, no. 5, pp. 2086–2104.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Tao, L., Wang, X.L., Guo, M.H. et al. Analysis of genomic DNA methylation and gene expression in Chinese cabbage (Brassica rapa L. ssp. pekinensis) after continuous seedling breeding. Russ J Genet 51, 774–782 (2015). https://doi.org/10.1134/S1022795415080116
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1022795415080116