Abstract
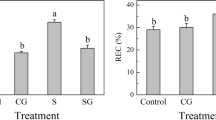
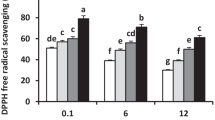
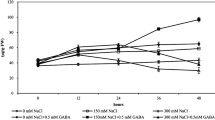
Salinity-alkalinity stress is a pivotal factor influencing plant growth, development, and yield. γ-Aminobutyric acid (GABA) protects plants against a variety of environmental stresses. However, it is remains largely unknown whether exogenous GABA increases the tolerance of Cucumis melon L. seedlings via effects on the chloroplast antioxidant system. In this study, the role of exogenous GABA application on the malondialdehyde content and antioxidant enzyme activities and the ascorbate-glutathione (AsA-GSH) cycle in seedlings of muskmelon was investigated. Plants were treated with foliar spraying of GABA (50 mM) under control or salinity-alkalinity stress conditions. Salinity-alkalinity stress induced cellular membrane damage. Treatment with GABA protected muskmelon seedlings from salinity-alkalinity stress by enhancing antioxidant enzyme activity and reducing malondialdehyde content. These effects of GABA resulted in maintenance of the membrane integrity of the muskmelon seedling. In addition, the status of both GSH and AsA redox played key roles in the regulation of the oxidative stress response in muskmelon seedlings under salinity-alkalinity stress.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- APX:
-
ascorbate peroxidase
- AsA:
-
ascorbate
- DHA:
-
dehydroascorbate
- DHAR:
-
dehydroascorbate reductase
- GABA:
-
γ-aminobutyric acid
- GR:
-
vglutathione reductase
- GSH:
-
glutathione reduced
- GSSG:
-
oxidized glutathione
- MDA:
-
malondialdehyde
- MDHAR:
-
monodehydroascorbate reductase
- O -2 :
-
superoxide anions
- SOD:
-
superoxide dismutase
References
Li, R., Shi, F., and Fukuda, K., Interactive effects of various salt and alkali stresses on growth, organic solutes, and cation accumulation in a halophyte Spartina alterniflora (Poaceae), Environ. Exp. Bot., 2010, vol. 68, pp. 66–74.
Yuan, L.Y., Zhu, S.D., Li, S.H., Shu, S., Sun, J., and Guo, S.R., 24-Epibrassinolide regulates carbohydrate metabolism and increases polyamine content in cucumber exposed to Ca(NO3)2 stress, Acta Physiol. Plant., 2014, vol. 36, no. 11, pp. 2845–2852
Shi, D. and Wang, D., Effects of various salt-alkaline mixed stresses on Aneurolepidium chinense (Trin.) Kitag, Plant Soil, 2005, vol. 271, nos. 1–2, pp. 15–26.
Yang, C.W., Wang, P., Li, C.Y., Shi, D.C., and Wang, D.L., Comparison of effects of salt and alkali stresses on the growth and photosynthesis of wheat, Photosynthetica, 2008, vol. 46, pp. 107–114.
Schieber, M. and Chandel, N.S., ROS function in redox signaling and oxidative stress, Curr. Opin. Cell Biol., 2014, vol. 24, no. 10, pp. 453–462
Reczek, C.R. and Chandel, N.S., ROS-dependent signal transduction, Curr. Opin. Cell Biol., 2015, vol. 33, pp. 8–13.
Bose, J., Rodrigo-Moreno, A., and Shabala, S., ROS homeostasis in halophytes in the context of salinity stress tolerance, J. Exp. Bot., 2014, vol. 65, pp. 1241–1257.
Dietz, K.J., Mittler, R., and Noctor, G., Recent progress in understanding the role of reactive oxygen species in plant cell signaling, Plant Physiol., 2016, vol. 171, pp. 1535–1539.
Gill, S.S. and Tuteja, N., Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants, Plant Physiol. Biochem., 2010, vol. 48, pp. 909–930.
Nahar, K., Hasanuzzaman, M., Alam, M.M., and Fujita, M., Exogenous spermidine alleviates low temperature injury in mung bean (Vigna radiata L.) seedlings by modulating ascorbate-glutathione and glyoxalase pathway, Int. J. Mol. Sci., 2015, vol. 16, pp. 30117–30132.
Meng, J.F., Xu, T.F., Wang, Z.Z., Fang, Y.L., Xi, Z.M., and Zhang, Z.W., The ameliorative effects of exogenous melatonin on grape cuttings under water-deficient stress: antioxidant metabolites, leaf anatomy, and chloroplast morphology, J. Pineal Res., 2014, vol. 57, pp. 200–212.
Shelp, B.J., Bown, A.W., and McLean, M.D., Metabolism and functions of gamma-aminobutyric acid, Trends Plant Sci., 1999, vol. 11, pp. 446–452.
Bouchereau, A., Aziz, A., Larher, F., and Martin-Tanguy, J., Polyamines and environmental challenges: recent development, Plant Sci., 1999, vol. 140, pp. 103–125.
Hu, X.H., Xu, Z.R., Xu, W.N., Li, J.M., Zhao, N., and Zhou, Y., Application of gamma-aminobutyric acid demonstrates a protective role of polyamine and GABA metabolism in muskmelon seedlings under Ca(NO3)2 stress, Plant Physiol. Biochem., 2015, vol. 92, pp. 1–10.
Malekzadeh, P., Khara, J., and Heydari, R., Alleviating effects of exogenous gamma-aminobutyric acid on tomato seedling under chilling stress, Physiol. Mol. Biol. Plants, 2014, vol. 20, pp. 133–137.
Wang, C.Y., Fan, Q.L., Gao, H.B., Wu, X.L., Li, J.R., Lv, G.Y., and Gong, B.B., Polyamine biosynthesis and degradation are modulated by exogenous gamma-aminobutyric acid in root-zone hypoxia-stressed melon roots, Plant Physiol. Biochem., 2014, vol. 82, pp. 17–26.
Xiang, L.X., Hu, L.P., Xu, W.N., Zhen, A., Zhang, L., and Hu, X.H., Exogenous gamma-aminobutyric acid improves the structure and function of photosystem IIin muskmelon seedlings exposed to salinity-alkalinity stress, PloS One, 2016, vol. 11: e0164847.
Shu, S., Yuan, L.Y., Guo, S.R., Sun, J., and Yuan, Y.H., Effects of exogenous spermine on chlorophyll fluorescence, antioxidant system and ultrastructure of chloroplasts in Cucumis sativus L. under salt stress, Plant Physiol. Biochem., 2013, vol. 63, pp. 209–216.
Li, J.M., Hu, L.P., Zhang, L., Pan, X.B., and Hu, X.H., Exogenous spermidine is enhancing tomato tolerance to salinity–alkalinity stress by regulating chloroplast antioxidant system and chlorophyll metabolism, BMC Plant Biol., 2015, vol. 15: 303.
Xu, P.L., Guo, Y.K., Bai, J.G., Shang, L., and Wang, X.J., Effects of long-term chilling on ultrastructure and antioxidant activity in leaves of two cucumber cultivars under low light, Physiol. Plant., 2008, vol. 132, pp. 467–478.
Giannopolitis, C.N. and Ries, S.K., Superoxide dismutases. I. Occurrence in higher plants, Plant Physiol., 1977, vol. 59, pp. 309–314.
Nakano, Y. and Asada, K., Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts, Plant Cell Physiol., 1981, vol. 22, pp. 867–880.
Gupta, A.S., Webb, R.P., Holaday, A.S., and Allen, R.D., Overexpression of superoxide dismutase protects plants from oxidative stress, Plant Physiol., 1993, vol. 103, pp. 1067–1073.
Zhang, J., Niu, J.P., Duan, Y., Zhang, M.X., Liu, J.Y., Li, P.M., and Ma, F.W., Photoprotection mechanism in the 'Fuji' apple peel at different levels of photooxidative sunburn, Physiol. Plant., 2015, vol. 154, pp. 54–65.
Bradford, M.M., A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding, Anal. Biochem., 1976, vol. 72, pp. 248–254.
Li, P.M. and Cheng, L.L., The shaded side of apple fruit becomes more sensitive to photoinhibition with fruit development, Physiol. Plant., 2008, vol. 134, pp. 282–292.
Stepien, P. and Klobus, G., Antioxidant defense in the leaves of C3 and C4 plants under salinity stress, Physiol. Plant., 2005, vol. 125, pp. 31–40.
Mittler, R., Oxidative stress, antioxidants and stress tolerance, Trends Plant Sci., 2002, vol. 7, pp. 405–410.
Hasanuzzaman, M., Hossain, M.A., and Fujita, M., Exogenous selenium pretreatment protects rapeseed seedlings from cadmium-induced oxidative stress by up-regulating antioxidant defense and methylglyoxal detoxification systems, Biol. Trace Elem. Res., 2012, vol. 149, pp. 248–261.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Chen, H., Liu, T., Xiang, L. et al. GABA Enhances Muskmelon Chloroplast Antioxidants to Defense Salinity-Alkalinity Stress. Russ J Plant Physiol 65, 674–679 (2018). https://doi.org/10.1134/S1021443718050047
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1021443718050047