Abstract
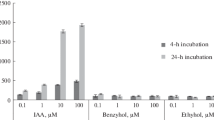
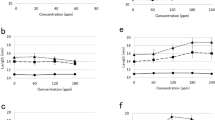
The interaction between auxin and oligosaccharin OS-RG of natural origin (DP ∼10) during the process of adventitious root formation was studied. Oligosaccharin isolated from pea seedlings increased by 20–25% the number of roots induced by IAA on both the segments of buckwheat (Fagopyrum esculentum Moench) hypocotyls and explants produced from the leaves of transgenic (rolB-GUS) tobacco Nicotiana tabacum L., cv Petit Havana. The highest effect was obtained after short-term treatment of explants with oligosaccharin before hormone adding. The optimal time of pretreatment depended on the used model system and was from 1–2 to 5–24 h for buckwheat hypocotyl segments and explants from tobacco leaves, respectively. Treatment with OS-RG after IAA did not affect the number of roots induced by the hormone. By using the explants from the leaves of transgenic tobacco plants harboring the reporter GUS gene under the control of the auxin-inducible promoter of the rolB gene permitted to reveal the explant response to the hormone at the early stages of root formation. The dynamics of GUS activity after IAA addition was characterized by the presence of two peaks. The histological analysis showed that the first peak coincided with the formation of 4–5-layer primordia, whereas the second peak — with the emergence of essentially developed roots. Pretreatment of explants from tobacco leaves with OS-RG activated IAA-induced GUS-activity and accelerated the response, i.e., the shift of the first peak to the beginning of culture without change in the position of the second peak. Thus, obtained data indicate that OS-RG action precedes that of the hormone at early stages of rhizogenesis. Possible mechanisms of interaction between IAA and oligosaccharin in the process of root formation are discussed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Dastidar, M.G., Jouannet, V., and Maizel, A., Root branching: mechanisms, robustness and plasticity, Dev. Biol., 2012, vol. 1, pp. 329–343.
Peret, B., Larrieu, A., and Bennett, M.J., Lateral root emergence: a difficult birth, J. Exp. Bot., 2009, vol. 60, pp. 3637–3643.
Lavenus, J., Goh, T., Roberts, I., Guyomarch, H., Lucas, M., de Smet, I., Fukaki, H., Beeckman, T., Bennett, M., and Laplaze, L., Lateral root development in Arabidopsis: fifty shades of auxin, Trends Plant Sci., 2013, vol. 18, pp. 450–457.
Moriwaki, T., Miyazawa, Y., Kobayashi, A., Uchida, M., Watanabe, C., Fujii, N., and Takahashi, H., Hormonal regulation of lateral root development in Arabidopsis modulated by MIZ1 and requirement of GNOM activity for MIZ1 function, Plant Physiol., 2011, vol. 157, pp. 1209–1220.
Parizot, B., de Rybel, B., and Beecman, T., VisuaLRTC: a new view on lateral root initiation by combining specific transcriptome data sets, Plant Physiol., 2010, vol. 153, pp. 34–40.
Li, S.W. and Xue, L., The interaction between H2O2 and NO, Ca2+, cGMP, and MAPKs during adventitious rooting in mung bean seedlings, In Vitro Cell Dev. Biol. — Plant, 2010, vol. 46, pp. 142–148.
Takahashi, F., Sato-Nara, K., Kobayashi, K., Suzuki, M., and Suzuki, H., Sugar-induced adventitious roots in Arabidopsis seedlings, J. Plant Res., 2003, vol. 116, pp. 83–91.
Albersheim, P., Darvil, A., McNeil, M., Valent, B., Sharp, J., Nothnagel, E., Davis, K., Yamazaki, N., Gollin, D., York, W., Dudman, W., Darvill, J., and Dell, A., Oligosaccharines, naturally occurring carbohydrates with biological regulatory functions, Structure and Function of Plant Genomes, Ciferri, O. and Dure, L., Eds., New York: Plenum Press, 1983, pp. 293–311.
Zabotina, O., Zabotin, A., Biologically active oligosaccharide functions in plant cell: updates and prospects, Oligosaccharides: Sources, Properties and Applications, Gordon, N.S., Ed., New York: Nova Science Publ., Inc., 2010, pp. 1–34.
Tran Than Van K., Toubart, P., and Cousson, A., Manipulation of the morphogenetic pathways of tobacco explants by oligosaccharins, Nature, 1985, vol. 314, pp. 615–617.
Bellincampi, D., Cardarelli, M., Zaghi, D., Serino, G., Salvi, G., Gatz, C., Cervone, F., Altamura, M., Costantino, P., and de Lorenzo, G., Oligogalacturonides prevent rhizogenesis in rolB transformed tobacco explants by inhibiting auxin-induced expression of the rolB gene, Plant Cell, 1996, vol. 8, pp. 477–487.
Kollarova, K., Henselova, M., and Liskova, D., Effect of auxins and plant oligosaccharides on root formation and elongation growth of mung bean hypocotyls, Plant Growth Regul., 2006, vol. 46, pp. 1–9.
Zabotina, O.A., Gur’yanov, O.P., Malikov, R.G., Ayupova, D.A., Beldman, G., Voragen, A.D., and Lozovaya, V.V., The isolation and biological activity of oligosaccharins from the pea shoots, Russ. J. Plant Physiol., 1995, vol. 42, pp. 366–371.
Zabotina, O.A., Gur’yanov, O.P., Ayupova, D.A., Lozovaya, V.V., Beldman, G., and Voragen, A.D., Isolation and analysis of soluble oligosaccharides stimulating root formation from pea shoots, Biochemistry (Moscow), 1997, vol. 62, pp. 845–849.
Seifert, G.J. and Blaukopf, C., Irritable walls: the plant extracellular matrix and signaling, Plant Physiol., 2010, vol. 153, pp. 467–478.
Ling, A.P.K., Kok, K.M., Hussein, S., and Ong, S.L., Effects of plant growth regulators on adventitious roots induction from different explants of Orthosiphon stamineus, Am.-Eur. J. Sust. Agric., 2009, vol. 3, pp. 493–501.
Falasca, G. and Altamura, M.M., Histological analysis of adventitious rooting in Arabidopsis thaliana (L.) Heynh seedlings, Plant Biosyst., 2003, vol. 137, pp. 265–274.
Lucas, M., Swarup, R., Paponov, I., Swarup, K., Casimiro, I., Lake, D., Peret, B., Zappala, S., Mairhofer, S., Whitworth, M., Wang, J., Ljung, K., Marchant, A., Sandberg, G., Holdsworth, M., Palme, K., Pridmore, T., Mooney, S., and Bennett, M., SHORT-ROOT regulates primary, lateral, and adventitious root development in Arabidopsis, Plant Physiol., 2011, vol. 155, pp. 384–398.
De Smet, I., White, P., Bengough, A., Dupuy, L., Parizot, B., Casimiro, I., Heidstra, R., Laskowski, M., Lepetit, M., Hochholdinger, F., Draye, X., Zhang, H., Broadley, M., Peret, B., Hammond, J., Fukaki, H., Mooney, S., Lynch, J., Nacry, P., Schurr, U., Laplaze, L., Benfey, P., Beeckman, T., and Bennett, M., Analyzing lateral root development: how to move forward, Plant Cell, 2012, vol. 24, pp. 15–20.
Murashige, T. and Skoog, F., A revised medium for rapid growth and bioassays with tobacco tissue culture, Physiol. Plant., 1962, vol. 15, pp. 473–497.
Pelosi, A., Lee, M., Chandler, S., and Hamill, J., Hormonal control of root primordial differentiation and root formation in cultured explants of Eucalyptus globules seedlings, Aust. J. Plant Physiol., 1995, vol. 22, pp. 409–415.
De Klerk, G.J., Hormone requirements during the successive phases of rooting of Malus microcuttings, Current Issues in Plant Cellular and Molecular Biology, Terzi, M., Cella, R., Falavigna, A., Eds., Dordrecht: Kluwer, 1995, pp. 111–116.
Williams, M.E., Nusbaum, H.C., and Sussex, I.M., Formation of lateral root meristems is a two-stage process, Development, 1995, vol. 121, pp. 3303–3310.
De Klerk, G.J., Keppel, M., Brugge, J., and Meekes, H., Timing of the phases in adventitious root formation in apple microcuttings, J. Exp. Bot., 1995, vol. 46, pp. 965–972.
Altamura, M., The role of hormones on morphogenesis of thin layer explants from normal and transgenic tobacco plants, Physiol. Plant., 1992, vol. 84, pp. 555–560.
Altamura, M., Archilletti, T., Capone, I., and Constantino, P., Histological analysis of the expression of Agrobacterium rhizogenes rolB-GUS gene fusions in transgenic tobacco, New Phytol., 1991, vol. 118, pp. 69–78.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © I.A. Larskaya, T.S. Barisheva, A.I. Zabotin, T.A. Gorshkova, 2015, published in Fiziologiya Rastenii, 2015, Vol. 62, No. 2, pp. 186–194.
Rights and permissions
About this article
Cite this article
Larskaya, I.A., Barisheva, T.S., Zabotin, A.I. et al. Character of oligosaccharin OS-RG participation in the IAA-induced formation of adventitious roots. Russ J Plant Physiol 62, 171–178 (2015). https://doi.org/10.1134/S1021443715020120
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1021443715020120