Abstract
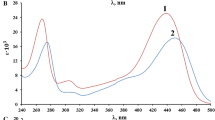
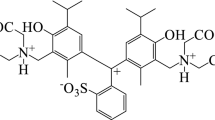
The brilliant blue FCF acid–base properties in aqueous solutions have been studied and its ionization constants have been defined by tristimulus colorimetry and spectrophotometry methods. The scheme of the acid–base dye equilibrium has been proposed and a diagram of the distribution of its ionic-molecular forms has been built. It has been established that the dominant form of the dye was the electroneutral form, which molar absorptivity (ε625 = 0.97 × 105) increases with the increase of the dielectric permittivity of the solvent. It has been shown that the replacement of polar solvents by less polar ones is causing a bathochromic shift of the maximum absorption band of the dye, the value of which is correlated with the value of the Hansen parameter. Tautomerization constants have been defined in a number of solvents and associated with the value of the Dimroth-Reichardt parameter.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
B. M. Ceyhan, F. Gultekin, D. K. Doguc, and E. Kulac, Food Chem. Toxicol. 56, 145 (2013).
K. Venkataraman, The Analytical Chemistry of Synthetic Dyes (Wiley, Chichester, 1977).
K. Yamjala, M. Nainar, and N. Ramisetti, Food Chem. 192, 813 (2016).
G. A. Krestov, V. A. Romanov, and A. G. Zakharov, Izv. Vyssh. Uchebn. Zaved., Khim. Khim. Tekhnol. 25 (7), 854 (1982).
V. A. Shormanov, A. A. Pyatachkov, and G. A. Krestov, Zh. Fiz. Khim. 56, 727 (1982).
D. T. Manallack, Perspect. Med. Chem. 1, 25 (2007).
A. N. Chebotarev, D. V. Snigur, E. V. Bevzyuk, and I. S. Efimova, Metody Ob’ekty Khim. Anal. 9 (1), 4 (2014).
V. M. Ivanov, O. V. Monogarova, and K. V. Oskolok, J. Anal. Chem. 70, 1165 (2015).
V. M. Ivanov and O. V. Kuznetsova, Russ. Chem. Rev. 70, 357 (2001).
A. N. Chebotarev and D. V. Snigur, J. Anal. Chem. 70, 55 (2015).
A. N. Chebotarev and D. V. Snigur, Russ. J. Gen. Chem. 86, 815 (2016).
O. M. Chebotarev, K. V. Bevzyuk, and D. V. Snigur, Ukr. Khim. Zh. 80 (6), 9 (2014).
C. Reichardt, Solvents and Solvent Effects in Organic Chemistry (Wiley-VCH, Weinheim, 2003).
Yu. L. Kaminskii, I. Ya. Bershtein, and O. F. Ginzburg, Dokl. Akad. Nauk 145, 330 (1962).
O. R. Sovin and I. O. Patsai, Metody Ob’ekty Khim. Anal. 7 (2), 74 (2012).
C. M. Hansen, Solubility Parameters: A User’s Handbook (CRC, Taylor Francis Group, Boca Raton, FL, 2007), p. 521.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.N. Chebotarev, K.V. Bevziuk, D.V. Snigur, Ya.R. Bazel, 2017, published in Zhurnal Fizicheskoi Khimii, 2017, Vol. 91, No. 10, pp. 1694–1699.
The article was translated by the authors.
Rights and permissions
About this article
Cite this article
Chebotarev, A.N., Bevziuk, K.V., Snigur, D.V. et al. The brilliant blue FCF ion-molecular forms in solutions according to the spectrophotometry data. Russ. J. Phys. Chem. 91, 1907–1912 (2017). https://doi.org/10.1134/S0036024417100089
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024417100089