Abstract
Although fish are often exposed to food restrictions in natural or aquafarming conditions, the relationship between altered energy status and the reproduction is not well understood. The aim of the present investigation was to elucidate the effect of chronic food-deprivation on energy status and reproductive axis in male Mozambique tilapia Oreochromis mossambicus. The controls received food ad libitum, whereas the fish in the starvation group were deprived of food for 21 days. The liver showed significantly higher levels of gluconeogenesis and triglycerides, whereas the levels of blood glucose and total protein content in the liver were significantly lower in starved fish compared to controls. Furthermore, the mean numbers of different spermatogenic cells, such as spermatogonia-A, primary spermatocytes, secondary spermatocytes, early spermatids, and late spermatids were significantly lower, but the number of spermatogonia-B did not show a significant difference compared to controls. In starved fish, there was a significant increase in germ cell apoptosis at different stages of development concomitant with faintly immunoreactive androgen receptors in the Sertoli cells of the testis compared with controls. In addition, the percent areas of gonadotropin-releasing hormone—immunoreactive fibres and luteinizing hormone—immunoreactive content—in the proximal pars distalis part of the pituitary gland were significantly lower in starved fish compared with controls. Collectively, these results suggest that decreased energy status negatively affects the gonadotropin-releasing hormone—luteinizing hormone – testicular axis, leading to inhibition of spermatogenesis before entry of germ cells into meiosis through increased apoptosis and decreased expression of androgen receptors in the testis of the Mozambique tilapia.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
Nutrition and food-intake are known to influence health, growth and reproduction in fish (Lall and Tibbetts, 2009). Fish are often subjected to food-deprivation conditions of variable duration during aquacultural practices or in their natural habitat due to food scarcity and/or environmental and seasonal alterations. For example, fish may starve due to handling stress, contamination in the environment and extreme cold as may occur during winter (Pérez-Jiménez et al., 2010; Jobling, 2015). During food-deprivation, fish may employ various behavioural, physiological and structural mechanisms to meet the metabolic needs using their energy reserves (Navarro and Gutiérrez, 1995). Diverse intermediate metabolic pathways are activated when nutrients stored in fish are consumed as energy during scarcity of food. Many researchers have evaluated a variety of blood metabolites, such as glucose, free fatty acids and triglycerides during fasting periods in fish (Costas et al., 2011; Ashouri et al., 2020). In the first stage of starvation, liver glycogen is mobilized to maintain the glucose homeostasis in some fish species such as the white sturgeon Acipenser transmontanus, the red-tailed brycon Brycon cephalus and the gilthead sea bream Sparus aurata (Hung et al., 1997; Figueiredo-Garutti et al., 2002; Metón et al., 2003). Besides, glucose homeostasis is achieved through gluconeogenesis by increasing the plasma levels of glucogenic amino acids (Gillis and Ballantyne, 1996) and triglycerides (Yarmohammadi et al., 2012) in fish.
Nevertheless, the metabolic response to food-deprivation is complex and not the same in different species. For example, mostly protein and lipid are utilized as energy sources, and glucose is conserved during fasting in fishes like the European eel Anguilla anguilla and the plaice Pleuronectes platessa (Dave et al., 1975; Moon and Johnston, 1980), whereas hepatic levels of protein, lipid, and glycogen are significantly decreased during starvation in the golden perch Macquaria ambigua (Collins and Anderson, 1995) and rainbow trout Oncorhynchus mykiss and sturgeon A. naccarii (Furné et al., 2012). However, some fish species such as the Northern pike Esox lucius rely mainly on glycogen storage during starvation (Ince and Thorpe, 1976) or glycogen is utilized only at the initial stage of prolonged starvation as observed in the roach Rutilus rutilus (Méndez and Wieser, 1993). Although significant reductions in plasma glucose and lipid, but not protein, are observed following starvation in the white sturgeon A. transmontanus (Hung et al., 1997), liver triglyceride levels are either unaffected in the Atlantic cod Gadus morhua (Hall et al., 2006) or significantly decreased in the Nile tilapia Oreochromis niloticus (Wang et al., 2019). In contrast, a significant increase in plasma triglyceride level is observed during starvation in the silver catfish Rhamdia quelen (Marqueze et al., 2018).
Unlike mammals, male fish exhibit a cystic type of spermatogenesis, which is controlled by gonadotropin-releasing hormone (GnRH), gonadotropins-follicle stimulating hormone (FSH) and luteinizing hormone (LH), as well as sex-steroids (testosterone/11-ketotestosterone, 11-KT) (Hatef and Unniappan, 2019). Since a cyst consists of cluster of specific germ cells derived from a single spermatogonia, fish serves a good model to study the effect of starvation on the reproductive axis. Effect of food-deprivation on reproduction has been examined in different species of fish. For example, starvation causes a significant reduction in the gonadosomatic index (GSI) in the Atlantic cod G. morhua (Dutil et al., 2003) and the Nile tilapia O. niloticus (Sales et al., 2020), and affects the testicular development and maturation in the iteroparous Arctic charr Salvelinus alpinus (Rice, 1999). In the African catfish Clarias gariepinus, although partial or full feed restriction does not alter the GSI, it affects the testicular activity depending on the phase of the reproductive cycle (Suchiang and Gupta, 2011). Similarly, exposure to starvation for 6 or 12 days does not affect the GSI, but suppresses the germ cell development concomitant with decrease in LH secretion in the Mozambique tilapia O. mossambicus (Pikle et al., 2017). In addition, starvation for 7–28 days lowers plasma concentration of testosterone and 11-KT concurrent with a decrease in the percentage of spermatogonia, but an increase in apoptosis of spermatocytes, spermatids and spermatozoa in the Nile tilapia (Sales et al., 2020).
Furthermore, a few studies have demonstrated the decreased GnRH mRNA/gene expression in fishes such as the winter flounder Pseudopleuronectes americanus (Tuziak and Volkoff, 2013), the Ya-fish Schizothorax prenanti (Wang et al., 2014), and the catfish C. gariepinus (Minari et al., 2022) exposed to starvation ranging between 3 and 14 days. Although these studies have revealed the impact of fasting on the GnRH or LH or testes in different species of fish discretely, detailed studies focusing on the hypothalamic-pituitary-testicular axis in same species are lacking. In particular, the present investigation aims to examine the effect of starvation on hypothalamic-hypophysial secretion and different stages of germ cell development and androgen receptor expression in the testis to get an insight into the possible mechanism by which starvation may affect the germ cell activity in the Mozambique tilapia O. mossambicus. We have also examined the biochemical markers to determine the association between energy status and reproduction in this fish. In this investigation, the Mozambique tilapia is used as an experimental model, as this fish is a commercially important food fish and exhibits cystic spermatogenesis throughout the year (Dadzie, 1969). Since the availability of fertile males is critical in fish farming, it is necessary to understand the impact of chronic starvation on the germ cell developmental stages in this species.
Whereas fishes employ various behavioural, physiological, and structural mechanisms to complete their metabolic needs using their energy reserves (Navarro and Gutiérrez, 1995), chronic and acute starvation may interfere with metabolically dependent reproduction in fish through distinct mechanisms (Hatef and Unniappan, 2019). Therefore, elucidating the impact of starvation along the hypothalamic-hypophysial-testicular axis is helpful in understanding the relationship between feeding status and reproduction in fish.
MATERIALS AND METHODS
Animals
Adult O. mossambicus were collected from ponds in and around Dharwad (75°01′ E, 15°27′ N) and brought to the laboratory. The fish were acclimatized to the laboratory conditions for one month prior to the commencement of experiment. The sexual maturity of males was confirmed by their ability to release milt upon gentle pressing at the ventral surface near the genital opening. The fish weighing 25–35 g were maintained at a stocking density of five fish per aquaria (70 L, size 36 × 12 × 18 inches; length × width × height) under natural conditions (photoperiod 11.45 ± 0.23 h, water temperature 29.86 ± 1.02°C, pH 7.46; dissolved oxygen 7.53 ± 0.25 mg/L). Water was replaced daily to avoid algal growth and accumulation of planktons that might interfere with the nutritional status of the fish in all aquaria during the period of experimentation.
Fish were divided into two groups with two replicates (5 fish per replicate, 10 fish per group). The control fish were fed (Taiyo pet feed, Chennai, India) ad libitum 3 g per aquaria daily for a period of 21 days, whereas the fish in second group were starved for 21 days. All experimental fish were euthanized following anesthetization with 2-phenoxy ethanol (1 : 1500, Sigma-Aldrich, USA). During autopsy, weights of the body, testis and liver were recorded, and GSI (100 × testis mass/body mass) and hepatosomatic index (HSI) (100 × liver mass/body mass) were calculated.
Biochemical Parameters
Blood samples of each fish were collected in a heparinized syringe and transferred to Eppendorf tubes. The Accu-Chek kit with a minimum detection limit of 10 mg/dL was used for determination of blood glucose level. The liver tissue samples from each fish were thawed and centrifuged for 5 min at 5000 rpm using a refrigerated centrifuge (Eppendorf, Germany) for determination of total protein content by Lowry’s method (Lowry et al., 1951), whereas triglyceride levels were estimated by the enzymatic glycerol phosphate-oxidase-peroxidase-aminoantipyrine (GPO-PAP) method using a triglyceride kit according to the GPO-Trinder method provided by the manufacturer (Erba Diagnostics, Mannhelm, GmbH, Germany). For the estimation of gluconeogenesis levels in the liver, the method described by Hanson (1981) was followed. Briefly, the saline (0.7%) was saturated with O2 : CO2 (95 : 5) at 37°C for 1 h in a CO2 incubator (Eppendorf, Germany). The slices of liver weighing about 60–100 mg from both experimental groups were cut and stored in ice-cold saline solution (0.7%) on a slide previously washed with saline. These slices were transferred to series of conical flasks (10 in each group) consisting of 3.8 mL of gassed saline solution and 0.2 mL of 0.2 M substrate (pyruvate, the precursor of glucose), whereas only 4 mL of gassed saline solution was taken as controls for the precursor. The contents in all conical flasks were gassed briefly in a CO2 incubator, covered with aluminium foil and then incubated at 37°C for 1 h with continuous gentle agitation. The slices were removed, allowed to drain briefly, and dried at 100°C for 15 min in an oven, and then 4 mL of 20% perchloric acid was added in centrifuge tubes. The mixture was stored at 0°C for 10 min and then insoluble proteins were removed by centrifugation. Glucose levels were assayed in 1.0 mL aliquots with two replicates for each supernatant using the glucose oxidase method. The liver glucose levels were estimated prior to the incubation using the glucose oxidase method. After applying Hanson’s method, the initial values of glucose were deducted from the gluconeogenesis assay data. The rate of glucose biosynthesis from the precursor was calculated and represented as µg/µL dry liver/h.
Histology and Quantification of Germ Cells
From each fish, single testis from the pair was fixed in Bouin’s fluid and used for histological studies, whereas the other testis was processed for immunohistochemical studies. Serial sections (5 µm) were cut from paraffin wax-embedded testis tissue and stained with hematoxylin and eosin. The numbers of different kinds of spermatogenic cells were counted as described earlier for this species (Pikle et al., 2017; Konkal and Ganesh 2018; Shinde and Ganesh, 2022). Different germ cell types, namely, spermatogonia-A (SG-A), spermatogonia-B (SG-B), primary spermatocytes, secondary spermatocytes, early spermatids, late spermatids and spermatozoa were identified based on their morphology and size. The number of SG-A was counted from every fourth section, whereas the numbers of SG-B, primary spermatocytes, secondary spermatocytes, early spermatids, and late spermatids were counted in every alternate section. Four cysts were randomly chosen per section and the mean values were calculated from a total of 100 cysts in the testis sections. By measuring each lobule in both the vertical and horizontal planes, the mean diameter of each lobule was calculated. The space between the seminiferous lobules was identified as interstitial space. The mean diameter of interstitial space was calculated by measuring this space in both the vertical and horizontal planes, and then the mean value was calculated. This method was also used to measure the diameters of 100 randomly selected testis lobules from each experimental fish. The photomicrography was done using CellSens image analysis software in DP74 CCD colour camera attached to a BX53 Fluorescent microscope (Olympus, Tokyo, Japan).
Immunohistochemistry and Immunofluorescence Labelling
After anaesthetization, the fish were perfused through the dorsal aorta with 15 ml of chilled phosphate-buffered saline (PBS, pH 7.4) initially and then 15 mL of cold 4% paraformaldehyde. The brains intact with the pituitary glands and single testis were fixed in cold 4% paraformaldehyde for 12 h. After a thorough rinse in PBS, the tissues were transferred to ice-cold 30% sucrose solution. Frozen sections of the brain through the pituitary (14 µm) and the testis (12 µm) were cut in transverse plane using a cryostat (CM1510S, Leica, Germany). For the detection of androgen receptors and apoptosis (caspase-3 active enzyme, a marker for apoptosis), the testis sections were labelled with rabbit anti-androgen receptor antibody (SP107, Sigma-Aldrich, USA; 1 : 3000) and rabbit anti-caspase-3 active antibody (RA15046, Neuromics, USA; 1 : 500) respectively, following streptavidin-biotin-peroxidase method (Konkal and Ganesh, 2020). The caspase-3 labelled sections were counterstained with haematoxylin. For the labelling of LH and GnRH-immunoreactivity in the pituitary gland, the sections were incubated with rabbit anti-human LHβ antisera (1 : 4000; National Hormone and Peptide Program, Harbor-UCLA Medical Centre, CA, USA) and rabbit anti-LHRH antibody (20075; Immunostar, USA; 1 : 500) respectively, following immunofluorescence protocol as described (Vijayalaxmi and Ganesh, 2017).
Control Procedures
The control procedures to affirm the specificity of the antibodies were employed as follows: (i) exclusion of primary antibody and replacement with 1% bovine serum albumin, (ii) omission of secondary antibody. In addition, for GnRH and LH, diluted primary antisera were preabsorbed with GnRH and LH peptides (Sigma-Aldrich, USA), respectively 24 h before incubation. These control procedures confirmed that the immunolabelling was due to the primary antibody employed.
Semiquantitative Analysis of Immunolabelling of Androgen Receptors, Apoptotic Germ Cells, Gonadotropin Releasing Hormone and Luteinizing Hormone
For quantitative analysis of immunohistochemical/immunofluorescence data, ImageJ version 1.46 (NIH, Bethesda, MD, USA) was used following the method described by Bhat and Ganesh (2020). The intensity of androgen receptor immunolabelling was measured in testis sections from each experimental fish (5 exemplars) and provided as arbitrary units (AU; mean ± standard error, SE) in the parenthesis, whereas the numbers of caspase-3 active-labelled apoptotic germ cells in cysts were counted from 100 lobules of each experimental testis. The percent areas of GnRH and LH immunoreactivities (5 each) were evaluated from each experimental pituitary gland.
Statistical Analysis
The data for all parameters were expressed as means ± SE. For all parameters, the normal data distribution and equality of variances were determined. Since these tests were passed, a parametric Student t‑test was conducted using SigmaStat 3.5 software. The significant differences for all statistical comparisons were determined at р < 0.05 level.
RESULTS
Biochemical Parameters
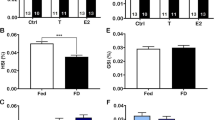
The total protein content of the liver and blood glucose levels were significantly lower (р < 0.02) in fasted fish compared to controls (Figs. 1a, 1b). However, the hepatic levels of triglyceride and gluconeogenesis were significantly increased (р < 0.03) in starved fish compared to those of controls (Figs. 1c, 1d).
Effect of food deprivation in the levels of hepatic levels of protein, μg/mL (a); blood glucose, mg/dL (b); triglyceride, mg/dL (c); and gluconeogenesis, μg/μL (d) in Oreochromis mossambicus. Here and in Figs. 2, 4, 5, 7 and 9: (◼)—controls, (◻)—fasting; values are means ± SE; Student t-test, * significant difference (p < 0.05).
Gonadosomatic Index, Hepatosomatic Index and Testis Histology
The mean GSI was significantly lower (р < 0.05), but the mean hepatosomatic index (HSI) did not show statistically significant difference (р = 0.302) (Fig. 2). Cross sections of the testis revealed the presence of several lobules in the germinal compartment consisting of germinal cysts with different spermatogenic cell types namely, SG-A, SG-B, primary spermatocytes, secondary spermatocytes, early spermatid, late spermatid and spermatozoa (Fig. 3). The morphometry of different spermatogenic cells for this species has been described previously (Shinde and Ganesh, 2022). The mean numbers of SG-B did not differ significantly (р = 0.114) from that of controls, whereas the mean numbers of SG-A, primary spermatocytes, secondary spermatocytes, early spermatids and late spermatids were significantly lower (р < 0.002) in fasting fish group compared to those of controls (Figs. 3, 4). The majority of seminiferous lobule boundaries were distorted in fasting fish and showed abundant spermatozoa in contrast to their sparse distribution in controls (Figs. 3a, 3d, 3e). The mean lobular diameter was significantly lower (р < 0.001), but the interstitial diameter was significantly higher (р < 0.001) in fasting fish compared to controls (Fig. 5).
Cross sections of the testis Oreochromis mossambicus showing the seminiferous lobules (Lb) and cysts (Cy) containing the Sertoli cells (S), spermatogonia-A (SG-A), spermatogonia-B (SG-B), primary spermatocytes (PSC), secondary spermatocytes (SSC), early spermatids (ES), and late spermatids (LS) in controls (a–c) and starvation group (d, e). Ld, Leydig cells; SZ, spermatozoa; L, lumen; Lb, lobule; DLb, distorted lobule. Coloring, haematoxylin and eosin. Scale, μm: 20 (a, d, e), 10 (b, c).
Effect of starvation on different stages of spermatogenesis in Oreochromis mossambicus. The mean number of spermatogonia-A and spermatogonia-B are not significantly different (a, b); however, the numbers of primary spermatocytes (с), secondary spermatocytes (d), early spermatids (e) and late spermatids (f) in starved fish were significantly reduced compared to the control.
Semiquantitative Analysis of Immunolabelling of Androgen Receptors, Apoptotic Germ Cells, Gonadotropin Releasing Hormone and Luteinizing Hormone
The intensity of immunolabelling of androgen receptors in the Sertoli cells (Fig. 6) was significantly lower (P < 0.001) in starved fish (127.64 ± 2.68 AU) compared to controls (54.86 ± 4.60 AU) (Figs. 6g, 6h). Application of anti-caspase-3 active antibody labelled the apoptotic cells in the testis of both experimental groups (Fig. 6). In general, caspase-3 positive labelling was higher in all types of germ cell cysts in fasting fish than in controls (Fig. 6). The mean numbers of apoptotic germ cells namely, SG-A, SG-B, primary spermatocytes, secondary spermatocytes, early spermatids and late spermatids were significantly higher (р < 0.01) in fasting fish compared to those of controls (Fig. 7). However, the number of apoptotic spermatozoa in starved fish did not differ significantly (р = 0.297) from that of controls (Fig. 7). GnRH-immunoreactive fibres were detected in the nucleus lateralis tuberis part of the hypothalamus, hypothalamo-hypophysial tract and proximal pars distalis (PPD) of the pituitary gland, whereas LH-immunoreactivity was confined to the PPD in both experimental groups (Fig. 8). The percent areas of GnRH-immunoreactive fibres and LH-immunoreactive content in the PPD of the pituitary gland were significantly lower (р < 0.001) in starved fish than in controls (Fig. 9).
Photomicrographs of cross sections of the testis showing caspase-3 active (a–f) and androgen receptor (g, h) immunoreactivities in controls (a–c, g) and fasting fish (d–f, h), respectively. Intense immunolabelling of androgen receptors in the Sertoli cell nuclei (→); (a–f) streptavidin-biotin peroxidase labelled sections counterstained with haematoxylin; (g, h) streptavidin-biotin peroxidase labelled. For designations see Fig. 3. Scale: 20 µm.
The effect of fasting on germ cell apoptosis in Oreochromis mossambicus. The average number of spermatogonia-A (SG-A), spermatogonia-B (SG-B), primary spermatocytes (PSC), secondary spermatocytes (SSC), early spermatids (ES) , late spermatids (LS) and spermatozoa (SZ) in starved fish was significantly reduced compared to the control.
Photomicrographs of cross sections of the brain through the proximal pars distalis (PPD) region of the pituitary gland (P) showing GnRH- and LH-immunoreactivities in Oreochromis mossambicus. In controls, GnRH-immunoreactive fibres (→) are seen the nucleus lateralis tuberis (NLT), hypothalamo-hypophysial-tract (HHT) and PPD (a), whereas LH-immunoreactive content (▶) is observed in the PPD (c). The areas occupied by GnRH-immunoreactive fibres (b) and LH-immunoreactive content (d) are decreased in fasting fish in contrast to their respective controls (a, c). Fluorescein labelled (a, b); Texas red labelled (c, d). Scale: 100 µm.
DISCUSSION
Glucose is an important fuel that must be maintained throughout the food deprivation (Gillis and Ballantyne, 1996). Although glucose levels were not affected following starvation in the rainbow trout, white sturgeon, red-tailed brycon and gilthead sea bream S. aurata (Hung et al., 1997; Figueiredo-Garutti et al., 2002; Metón et al., 2003; Azodi et al., 2014), the majority of studies on different fish species have documented reduction in glucose levels following exposure to starvation of variable duration. For example, significant decrease in blood or plasma glucose levels was observed in the rohu Labeo rohita (7 days), the gibel carp Carassius gibelio (7 days), the European seabass Dicentrarchus labrax (22 days), the Atlantic salmon Salmo salar (14 days), the silver catfish R. quelen (7–21 days), the pacu Piaractus mesopotamicus (30 days), the neotropical catfish Lophiosilurus alexandri (50 days), the sunshine bass Morone chrysops (3 weeks), Nile tilapia O. niloticus (14 or 28 days) and the European eel A. anguilla (42 days) (Caruso et al., 2008; Barcellos et al., 2010; Davis and Gaylord, 2011; Viegas et al., 2013; Waagbø et al., 2017; Li et al., 2018; Favero et al., 2017, 2019; Dar et al., 2019; Silva et al., 2019; Sales et al., 2020; Sakyi et al., 2020). A significant decrease in blood glucose levels observed following fasting for 21 days in the Mozambique tilapia agrees with these studies. Maintenance of glucose levels, although significantly lower during fasting from 7–50 days as observed in the above studies, might be due to glycogenolysis and gluconeogenesis (Dai et al., 2021). Since mobilization of glycogen occurs during the initial phase of fasting (Polakof et al., 2006), the Mozambique tilapia exposed to starvation for 21 days in the present study may depend on gluconeogenesis for the maintenance of glucose homeostasis as shown by a significant increase in the hepatic levels of gluconeogenesis.
Nevertheless, the occurrence of gluconeogenesis through break-down of triglycerides or protein appears to be species-specific. Significant decrease in liver protein content has been documented in different fishes including the rainbow trout (McMillan et al., 1992; Karatas et al., 2021), the Arctic charr Salvelinus alpinus (Cassidy et al., 2016), the silver catfish Bagrus bajad (Marqueze et al., 2018) and the sea bream S. aurata (Power et al., 2000) exposed to starvation period ranging from 1–120 days. In the rainbow trout O. mykiss, although serum triglycerides were significantly lower, serum levels of total proteins were not affected (Karatas et al., 2019). Likewise, in the Mozambique tilapia O. mossambicus starved for 6 or 12 days resulted in significantly lower levels of serum triglycerides and liver protein levels (Vijayalaxmi et al., 2019). In the present study, although a significant decrease in hepatic protein levels in the Mozambique tilapia subjected to long-term starvation (21 days) is consistent with the previous study (Vijayalaxmi et al., 2019) and other teleosts (McMillan et al., 1992; Cassidy et al., 2016; Power et al., 2000; Karatas et al., 2021), hepatic levels of triglycerides on day 21 were significantly higher than that of the Mozambique tilapia subjected to 6 or 12 days of fasting. These results clearly suggest that depletion of hepatic protein occurs consistently from day 6 (Vijayalaxmi et al., 2019) to day 21 (current study) in the Mozambique tilapia, but serum/hepatic triglyceride levels may fluctuate depending on the duration of starvation. In other words, a significant decrease in serum triglyceride levels (Vijayalaxmi et al., 2019) appears to be due to the utilization of triglycerides available during short-term starvation period (6 or 12 days) in the Mozambique tilapia; however, a significant increase in hepatic triglyceride levels on day 21 of fasting may be due to the fact that fat stores are mobilized in response to prolonged starvation, resulting in release of glycerol and free fatty acids. Indeed, a significant increase in the hepatic levels of triglycerides was detected in the Persian sturgeon A. persicus starved for 3 weeks (Yarmohammadi et al., 2012). Similarly, a significant increase in the level of triglycerides was evident in the silver catfish R. quelen (Marqueze et al., 2018) during 21 days starvation. On the contrary, hepatic levels of triglycerides in the Atlantic cod G. morhua starved for one month did not show any significant increase (Hall et al., 2006).
Although the HSI reflects the energy status of the fish, the effect of starvation on HSI is not consistent among different fish species. The HSI was significantly reduced in food-deprived fishes such as the carp Cyprinus carpio (50 or 90 days), the rainbow trout O. mykiss (10 days), the catfish (54 days), the beluga Huso huso (6 weeks), the grey mullet Mugil cephalus (30 days), and in the Nile tilapia O. niloticus (21 days) (Blasco et al.,1992; Vosylienė et al., 1999; Peterson and Small, 2004; Falahatkar et al., 2012; Pronina and Revakin, 2015; Akbary et al., 2016; Sakyi et al., 2020). However, in the present study, we did not notice any significant difference in the HSI between 21 days starved fish and controls. Similar results were also observed in the Atlantic cod G. morhua exposed to two-month period of starvation (Hall et al., 2006).
The GSI reflects the testicular development, but the effect of starvation on the GSI is not consistent among different teleosts. The mean GSI was significantly lower due to starvation for 84 days in male and female Atlantic cod G. morhua (Dutil et al., 2003) and 28 days of fasting in male Nile tilapia O. niloticus (Sales et al., 2020). A significant decrease in the mean GSI of the Mozambique tilapia following exposure to prolonged starvation (21 days) in the present study agrees well with the previous studies (Dutil et al., 2003; Sales et al., 2020). In contrast, the mean GSI did not show significant difference in the Mozambique tilapia O. mossambicus starved for 6 or 12 days (Pikle et al., 2017; Vijayalaxmi et al., 2019). Additionally, the testis development was not affected in the cichlid Cichlasoma nigrofasciatum subjected to four months starvation (Townshend and Wootton, 1984) or restricted feeding in the matrinxã Brycon amazonicus (Carvalho, 2001). However, the testicular development and maturation was suppressed due to restricted feeding for 2−3 months during spring time in male chinook salmon (Hopkins and Unwin, 1997) or due to starvation for 6 weeks in the iteroparous Arctic charr S. alpinus (Rice, 1999). In the present study, significant decrease in the lobule diameter and number of germ cells associated with a significantly higher interstitial diameter suggests that both spermatogenesis and spermiation was affected due to starvation in the Mozambique tilapia.
We may recall that the SG-A give rise to the SG-B, which divide more rapidly than the former and differentiate into primary spermatocytes (1st meiotic division), secondary spermatocytes (2nd meiotic division), spermatids (differentiation without proliferation), and finally spermatozoa (Schulz et al., 2010). The effect of starvation on different stages of spermatogenesis has been examined in some teleosts, but the progression of spermatogenic germ cell development in response to starvation appears to vary depending on species. In the Nile tilapia subjected to fasting for 21 or 28 days (Sales et al., 2020), a significant decrease in the proportion of SG-A and spermatocytes was observed, but the percentage of SG-B and spermatids was not significantly different. Likewise, the proportion of spermatogonia and spermatocytes was significantly increased, but the percentage of spermatids was not affected in the zebrafish Danio rerio starved for 3 weeks (Fan et al., 2021). Pikle et al., (2017) observed a significant decrease in the mean numbers of spermatogonia, spermatocytes and spermatids in the Mozambique tilapia starved for 6 or 12 days. However, information on the effect of chronic starvation on progression of specific germ cell developmental stages such as SG-A, SG-B, primary spermatocytes, secondary spermatocytes, early spermatids and late spermatids was lacking in the previous study. In this study, exposure of the Mozambique tilapia to 21 days starvation did not affect the mean numbers of SG-B, but caused a significant reduction the mean numbers of SG-A, primary spermatocytes, secondary spermatocytes, early spermatids and late spermatids. The non-significant effect of starvation on SG-B in the Mozambique tilapia or Nile tilapia can be attributed to the rapid division of these germ cells, as reported by Schulz et al. (2010). Similarly, mitotic divisions were unaffected in the Mozambique tilapia exposed to aquacultural stressors (Shinde and Ganesh, 2022). In addition, germ cell loss at an early stage of development due to apoptosis cannot be ruled out. Although germ cell populations can be lost due to apoptosis before differentiating to spermatozoa under normal circumstances in fish (Vilela et al., 2003; Almeida et al., 2008), in the present study, we observed a high incidence of apoptosis in specific stages of germ cell development, as shown by significantly higher numbers of SG-A, SG-B, primary spermatocytes, secondary spermatocytes, early spermatids, and late spermatids positive for caspase-3 active antibody in the spermatogenic cysts in starved fish. However, the number of spermatozoa undergoing apoptosis did not differ significantly between controls and starved fish. Since activated capase-3 is one of among the several apoptotic markers that has been conserved in all vertebrates including fish (Takle and Anderson, 2007), these results suggest that starvation affects the recruitment of germ cells at early stage of spermatogenesis and triggers apoptosis at SG-A stage, leading to significant dcrease in the number of spermatogenic cells at advanced stages in the Mozambique tilapia.
Consistent with the previous study (Pikle et al., 2017), we observed densely packed spermatozoa in the lumens of the lobules in the Mozambique tilapia subjected to 21 days of starvation in the present study. Similar results were also reported by Sasayama and Takahashi (1972) in the goldfish Carassius auratus exposed to starvation for 15–45 days. Pikle et al., (2017) suggested that this strategy may ensure the rapid completion of spermatogenesis under unfavourable conditions like food-deprivation. On the contrary, a significant decrease in the percentage of spermatozoa was observed in the zebrafish fasted for 3 weeks (Fan et al., 2021) and in the Nile tilapia starved for 21 or 28 days (Sales et al., 2020).
Fish reproduction is controlled predominantly by the hypothalamic-pituitary-gonad axis, which includes the release of GnRH from the hypothalamic neurons and the secretion of FSH and LH from the pituitary gonadotropes (Yaron et al., 2003). In addition, spermatogenesis is regulated by several factors, including testicular steroidogenesis, in teleosts (Hatef and Unniappan, 2019; Kitano et al., 2022). The role of gonadotropins in the regulation of testicular activity appears to be complicated due to the complex relationship between FSH and LH signalling pathways, especially in teleosts. While FSH and LH bind to their specific receptors on the Sertoli cells and Leydig cells, respectively in the majority of the teleosts (Sambroni et al., 2007; Rocha et al., 2007; Schulz et al., 2010) in some salmonids (Oba et al., 1999), catfish C. gariepinus (Vischer et al., 2003), and zebrafish D. rerio (So et al., 2005), activation of the FSH receptors by LH has also been reported. However, using FSH receptor knockout male medaka that had normal testes and were fertile, Kitano et al., (2022) suggested that FSH receptors are not absolutely necessary for spermatogenesis. In addition, LH receptors are also expressed in the Sertoli cells of the testes in many teleosts, including zebrafish D. rerio (Miwa et al., 1994; Oba et al., 2001; García-López et al., 2010). Since LH regulates Leydig cell sex-steroid production, which is necessary for spermatogenesis (Schulz et al., 2010), and LH can stimulate FSH receptors (So et al., 2005), it is possible that impairment in LH can disrupt spermatogenesis. In teleosts, the median eminence is absent, and the hypothalamic secretions are directly discharged into the anterior pituitary gland, which contains gonadotropin-secreting cells as well (Fryer and Maler, 1981). The results of our study provide neuroanatomical evidence for the starvation-induced impairment in the release of hypothalamic GnRH into the PPD region of the pituitary gland, where gonadotropin-secreting cells are located, as shown by a significant decrease in the density of GnRH-ir fibres and LH-ir content in the PPD of the pituitary gland in the Mozambique tilapia. In addition, faintly labelled androgen receptors were observed in the Sertoli cells of the starved fish. Among other teleosts, decreased levels of GnRH mRNA/gene expression were observed in the winter flounder, Ya-fish, Nile tilapia, and catfish (Tuziak and Volkoff, 2013; Wang et al., 2014; Park et al., 2016; Minari et al., 2022) that were exposed to starvation for 3–14 days. Likewise, a significant reduction in LH-immunoreactive content was observed in 12‑day-starved Mozambique tilapia (Pikle et al., 2017). In contrast, food restriction did not affect expression of the GtH gene in the goldfish (Sohn et al., 1998) or mRNA expression of GnRH in wild-type male zebrafish D. rerio that were starved for one week (London and Volkoff, 2019). Kawauchi et al., (1989) reported the hyperplausia of LH cells at the time of final maturation in teleosts, and pituitary levels of GnRH were significantly increased in male and female zebrafish starved for 14 days (Marvel et al., 2021). These discrepancies might be due to the duration of starvation or species-specific responses.
Overall, our results suggest that depletion of energy, as indicated by an increased level of gluconeogenesis due to chronic starvation, can negatively influence the GnRH-LH-testicular axis in the Mozambique tilapia. While this axis is under the control of several factors, the activation of the stress axis during starvation cannot be excluded. For example, exposure of the gilthead sea bream to 14 days of starvation resulted in increased plasma cortisol levels (Polakof et al., 2006), and treatment with cortisol suppressed GnRH mRNA and circulating levels of LH and FSH in cinnamon clownfish Amphiprion melanopus (Choi et al., 2017). Indeed, the Mozambique tilapia also shows a significant cortisol response to aquacultural stressors, resulting in the suppression of the pituitary-testicular axis (Shinde and Ganesh, 2022). In addition, the negative effect of starvation on FSH and the involvement of other factors that can interact with GnRH, such as kisspeptins (Park et al., 2016), neuropeptide Y (Narnaware et al., 2001) and thyroid hormones (Cerdá-Reverter et al., 1996; Pavlov et al., 2020), merit further studies.
CONCLUSIONS
In conclusion, the results of the present study suggest that increased gluconeogenesis and lipid mobilization due to starvation can affect the GnRH-LH-testicular axis as shown by the suppression of release of hypophysiotropic GnRH into the PPD and secretion of LH at the pituitary level, and decreased expression of androgen receptors concomitant with increased rate of germ cells apoptosis prior to meiosis in the Mozambique tilapia. Since the success of fish farming depends on the availability of potentially fertile males, it is suggested that starvation-induced energy crisis must be avoided to ensure the commercial benefits.
REFERENCES
Akbary, P. and Jahanbakhshi, A., Effect of starvation on growth, biohemical, hematological and non-specific immune parameters in two different size groups of grey mullet, Mugil cephalus (Linnaeus, 1758), Acta Ecol. Sin., 2016, vol. 36, no. 3, pp. 205−211. https://doi.org/10.1016/j.chnaes.2016.04.008
Almeida, F.F., Kristoffersen, C., Taranger, G.L., and Schulz, R., Spermatogenesis in Atlantic cod (Gadus morhua): A novel model of cystic germ cell development, Biol. Reprod., 2008, vol. 78, no. 1, pp. 27–34. https://doi.org/10.1095/biolreprod.107.063669
Ashouri, G., Mahboobi-Soofiani, N., Hoseinifar, S.H., et al., Compensatory growth, plasma hormones and metabolites in juvenile Siberian sturgeon (Acipenser baerii, Brandt 1869) subjected to fasting and re-feeding, Aquacult. Nutr., 2020, vol. 26, no. 2, pp. 400−409. https://doi.org/10.1111/anu.13002
Azodi, M., Ebrahimi, E., Motaghi, E., and Morshedi, V., Metabolic responses to short starvation and re-feeding in rainbow trout (Oncorhynchus mykiss), Ichthyol. Res., 2014, vol. 62, no. 2, pp. 177–183. https://doi.org/10.1007/s10228-014-0421-z
Barcellos, L.J.G., Marqueze, A., Trapp, M., et al., The effects of fasting on cortisol, blood glucose and liver and muscle glycogen in adult jundiá Rhamdia quelen, Aquaculture, 2010, vol. 300, nos. 1−4, pp. 231–236. https://doi.org/10.1016/j.aquaculture.2010.01.013
Bhat, S.K. and Ganesh, C.B., Domperidone treatment attenuates stress-induced suppression of reproduction in viviparous mosquitofish Gambusia affinis. J. Fish. Biol., 2020, vol. 96, no. 1, pp. 37–48. https://doi.org/10.1111/jfb.14183
Blasco, J., Fernández, J., and Gutiérrez, J., Fasting and refeeding in carp, Cyprinus carpio L.: The mobilization of reserves and plasma metabolite and hormone variations, J. Comp. Physiol. B., 1992, vol. 162, pp. 539–546. https://doi.org/10.1007/BF00264815
Caruso, G., Denaro, M. G., and Genovese, L., Temporal changes in digestive enzyme activities in the gastrointestinal tract of European eel (Anguilla anguilla) (Linneo 1758) following feeding, Mar. Freshw Behav. Physiol., 2008, vol. 41, no. 4, pp. 215–228. https://doi.org/10.1080/10236240802492931
Carvalho, E.G., Restriҫão alimentar e reproduҫão peixes, Ph. D. Thesis, Jaboticabal: Centro de Aquicultura da UNESP, 2001.
Cassidy, A.A., Saulnier, R.J., and Lamarre, S.G., Adjustments of protein metabolism in fasting Arctic charr, Salvelinus alpines, PLos One, 2016, vol. 11, no. 4, Article e0153364. https://doi.org/10.1371/journal.pone.0153364
Cerdá-Reverter, J.M., Zanuy, S., Carrillo, M., and Kah, O., Development of enzyme immunoassays for 3,5,3'-triiodo-L-thyronine and L-thyroxine: Time-course studies on the effect of food deprivation on plasma thyroid hormones in two marine teleosts, sea bass (Dicentrarchus labrax L.) and sea bream (Sparus aurata L.), Gen. Comp. Endocrinol., 1996, vol. 103, no. 3, pp. 290–300. https://doi.org/10.1006/gcen.1996.0124
Choi, Y. J., Habibi, H. R., Kil, G. S., et al., Effect of cortisol on gonadotropin inhibitory hormone (GnIH) in the cinnamon clownfish, Amphiprion melanopus, Biochem. Biophys. Res. Commun., 2017, vol. 485, no. 2, pp. 342–348. https://doi.org/10.1016/j.bbrc.2017.02.078
Collins, A.L. and Anderson, T.A., The regulation of endogenous energy stores during starvation and refeeding in the somatic tissues of the golden perch, J. Fish Biol., 1995, vol. 47, no. 6, pp. 1004–1015. https://doi.org/https://doi.org/10.1111/j.1095-8649.1995.tb06024.x
Costas, B., Aragão, C., Ruiz-Jarabo, I., et al., Feed deprivation in Senegalese sole (Solea senegalensis Kaup, 1858) juveniles: Effects on blood plasma metabolites and free amino acid levels, Fish Physiol. Biochem., 2011, vol. 37, no. 3, pp. 495–504. https://doi.org/10.1007/s10695-010-9451-2
Dadzie, S., Spermatogenesis and the stages of maturation in the male cichlid fish Tilapia mossambica, J. Zool., 1969, vol. 159, no. 3, pp. 399–403. https://doi.org/10.1111/j.1469-7998.1969.tb08454.x
Dai, Y.F., Shen, Y.B., Wang, S.T., et al., RNA-Seq Transcriptome analysis of the liver and brain of the black carp (Mylopharyngodon piceus) during fasting, J. Mar. Biotechnol., 2021, vol. 23, no. 3, pp. 389–401. https://doi.org/10.1007/s10126-021-10032-9
Dar, S.A., Srivastava, P.P., Varghese, T., et al., Temporal changes in superoxide dismutase, catalase, and heat shock protein 70 gene expression, cortisol and antioxidant enzymes activity of Labeo rohita fingerlings subjected to starvation and refeeding, Gene, 2019, vol. 692, pp. 94–101. https://doi.org/10.1016/j.gene.2018.12.058
Dave, G., Johansson-Sjöbeck, M.L., Larsson, A., et al., Metabolic and hematological effects of starvation in the European eel, Anguilla anguilla L. I. Carbohydrate, lipid, protein and inorganic ion metabolism, Comp. Biochem. Physiol. A. Comp. Physiol., 1975, vol. 52, no. 3, pp. 423–430. https://doi.org/10.1016/s0300-9629(75)80059-4
Davis, K.B. and Gaylord, T.G., Effect of fasting on body composition and responses to stress in sunshine bass, Comp. Biochem. Physiol. A. Mol. Int. Physiol., 2011, vol. 158, no. 1, pp. 30–36. https://doi.org/10.1016/j.cbpa.2010.08.019
Dutil, J., Winter and spring changes in condition factor and energy reserves of wild cod compared with changes observed during food-deprivation in the laboratory, ICES J. Mar. Sci., 2003, vol. 60, no. 4, pp. 780–786. https://doi.org/10.1016/S1054-3139(03)00024-9
Falahatkar, B., The metabolic effects of feeding and fasting in beluga Huso huso, Mar. Environ. Res., 2012, vol. 82, pp. 69–75. https://doi.org/10.1016/j.marenvres.2012.09.003
Fan, X., Cui, L., Hou, T., et al., Stress responses of testicular development, inflammatory and apoptotic activities in male zebrafish (Danio rerio) under starvation, Dev. Comp. Immunol., 2021, vol. 114, Article 103833. https://doi.org/10.1016/j.dci.2020.103833
Favero, G.C., Gimbo, R.Y., Franco Montoya, L.N., et al., Fasting and refeeding lead to more efficient growth in lean pacu (Piaractus mesopotamicus), Aquacult. Res., 2017, vol. 49, no. 1, pp. 359–366. https://doi.org/ doi:10.1111/are.13466
Favero, G.C., Boaventura, T.P., Ferreira, A.L., et al., Fasting / re-feeding and water temperature promote the mobilization of body reserves in juvenile freshwater carnivorous catfish Lophiosilurus alexandri, Aquaculture, 2019, vol. 511, Article 734223. https://doi.org/10.1016/j.aquaculture.2019.734223
Figueiredo-Garutti, M.L., Navarro, I., Capilla, E., et al., Metabolic changes in Brycon cephalus (Teleostei, Characidae) during post-feeding and fasting, Comp. Biochem. Physiol. Part A. Mol. Integr. Physiol., 2002, vol. 13, no. 2, pp. 467–476. https://doi.org/10.1016/s1095-6433(02)00094-6
Fryer, J.N. and Maler, L., Hypophysiotropic neurons in the goldfish hypothalamus demonstrated by retrograde transport of horseradish peroxidase, Cell Tiss. Res., 1981, vol. 218, pp. 93–102. https://doi.org/10.1007/BF00210094
Furné, M., Morales, A.E., Trenzado, C.E., et al., The metabolic effects of prolonged starvation and refeeding in sturgeon and rainbow trout, J. Comp. Physiol. B., 2012, vol. 182, pp. 63–76. https://doi.org/10.1007/s00360-011-0596-9
García-López, A., de Jonge, H., Nóbrega, R.H., et al., Studies in zebrafish reveal unusual cellular expression patterns of gonadotropin receptor messenger ribonucleic acids in the testis and unexpected functional differentiation of the gonadotropins, Endocrinology, 2010, vol. 151, no. 5, pp. 2349–2360. https://doi.org/10.1210/en.2009-1227
Gillis, T.E. and Ballantyne, J. S., The effects of starvation on plasma free amino acid and glucose concentrations in lake sturgeon, J. Fish Biol., 1996, vol. 49, no. 6, pp. 1306–1316. https://doi.org/10.1111/j.1095-8649.1996.tb01797.x
Hall, J. R., Short, C. E., and Driedzic, W. R., Sequence of Atlantic cod (Gadus morhua) GLUT4, GLUT2 and GPDH: Developmental stage expression, tissue expression and relationship to starvation-induced changes in blood glucose, J. Exp. Biol., 2006, vol. 209, no. 22, pp. 4490–4502. https://doi.org/10.1242/jeb.02532
Hanson, R., Glycolysis and gluconeogenesis, Biochem. Educat., 1981, vol. 9, no. 3, pp. 89–91. https://doi.org/10.1016/0307-4412(81)90209-0
Hatef, A. and Unniappan, S., Metabolic hormones and the regulation of spermatogenesis in fishes, Theriogenology, 2019, vol. 134, pp. 121–128. https://doi.org/10.1016/j.theriogenology.2019.05.021
Hopkins, C. L. and Unwin, M. J., The effect of restricted springtime feeding on growth and maturation of freshwater-reared chinook salmon, Oncorhynchus tshawytscha (Walbaum), Aquacult. Res., 1997. vol. 28, no. 7, pp. 545–549. https://doi.org/10.1046/j.1365-2109.1997.00893.x
Hung, S.S., Liu, W., Li, H., et al., Effect of starvation on some morphological and biochemical parameters in white sturgeon, Acipenser transmontanus, Aquaculture, 1997, vol. 151, pp. 357−363. https://doi.org/10.1016/S0044-8486(96)01506-2
Ince, B.W. and Thorpe, A., The effects of starvation and force-feeding on the metabolism of the Northern pike, Esox lucius L., J. Fish Biol., 1976, vol. 8, no. 1, pp.79−88. https://doi.org/10.1111/j.1095-8649.1976.tb03909.x
Jobling, M., Fish nutrition research: past, present and future, Aquacult. Int., 2015, vol. 24, pp. 767–786. https://doi.org/10.1007/s10499-014-9875-2
Karatas, T., Effect of fasting and re-feeding on growth performance, Biochemical parameters of brown trout (Salmo trutta fario) and rainbow trout (Oncorhynchus mykiss), J. Anim. Plant Sci., 2019, vol. 29, no. 3, pp. 667−673.
Karatas, T., Onalan, S., and Yildirim, S., Effects of prolonged fasting on levels of metabolites, oxidative stress, immune-related gene expression, histopathology, and DNA damage in the liver and muscle tissues of rainbow trout (Oncorhynchus mykiss), Fish Physiol. Biochem., 2021, vol. 47, no. 4, pp. 1119–1132. https://doi.org/10.1007/s10695-021-00949-2
Kawauchi, H., Suzuki, K., Itoh, H., et al., The duality of teleost gonadotropins, Ibid., 1989, vol. 7, nos. 1−6, pp. 29–38. https://doi.org/10.1007/BF00004687
Kitano, T., Takenaka, T., Takagi, H., et al., Roles of gonadotropin receptors in sexual development of medaka, Cells, 2022, vol. 11, no. 3, Article 387. https://doi.org/10.3390/cells11030387
Konkal, P. and Ganesh, C. B., Exposure to low or high light intensity affects pituitary-testicular activity in the fish Oreochromis mossambicus, Aquaculture, 2018, vol. 497, pp. 109–116.https://doi.org/10.1016/j.aquaculture.2018.07.043
Konkal, P. and Ganesh, C.B., Continuous exposure to light suppresses the testicular activity in Mozambique tilapia Oreochromis mossambicus (Cichlidae), J. Ichthyol., 2020, vol. 60, pp. 660–667. https://doi.org/10.1134/S0032945220040074
Lall, S. P. and Tibbetts, S. M., Nutrition, feeding, and behavior of fish, Vet. Clin. North. Am. Exot. Anim. Pract., 2009, vol. 12, no. 2, pp. 361–xi. https://doi.org/10.1016/j.cvex.2009.01.005
Li, H., Xu, W., Jin, J., et al., Effects of starvation on glucose and lipid metabolism in gibel carp (Carassius auratus gibelio var. CAS III), Aquaculture, 2018, vol. 496, pp. 166–175. https://doi.org/10.1016/j.aquaculture.2018.07.015
London, S. and Volkoff, H., Effects of fasting on the central expression of appetite-regulating and reproductive hormones in wild-type and Casper zebrafish (Danio rerio), Gen. Comp. Endocrinol., 2019, vol. 282, Article 113207. https://doi.org/10.1016/j.ygcen.2019.06.011
Lowry, O.H., Rosebrough, N.J., Farr, A L., and Randall, R.J., Protein measurement with the Folin phenol reagent, J. Biol. Chem., 1951, vol. 193, no. 1, pp. 265–275.
Marqueze, A., Garbino, C.F., Trapp, M., et al., Protein and lipid metabolism adjustments in silver catfish (Rhamdia quelen) during different periods of fasting and refeeding, Braz. J. Biol., 2018, vol. 78, no. 3, pp. 464–471. https://doi.org/10.1590/1519-6984.169333
Marvel, M., Levavi-Sivan, B., Wong, T.T., et al., Gnrh2 maintains reproduction in fasting zebrafish through dynamic neuronal projection changes and regulation of gonadotropin synthesis, oogenesis, and reproductive behaviours, Sci. Rep., 2021, vol. 11, no. 1, Article 6657. https://doi.org/10.1038/s41598-021-86018-3
McMillan, D.N. and Houlihan, D.F., Protein synthesis in trout liver is stimulated by both feeding and fasting, Fish Physiol. Biochem., 1992, vol. 10, no. 1, pp. 23–34. https://doi.org/10.1007/BF00004651
Méndez, G. and Wieser, W., Metabolic responses to food deprivation and refeeding in juveniles of Rutilus rutilus (Teleostei: Cyprinidae), Environ. Biol. Fishes., 1993, vol. 36, no. 1, pp. 73–81https://doi.org/10.1007/BF00005981
Metón, I., Fernández, F., and Baanante, I.V., Short- and long-term effects of refeeding on key enzyme activities in glycolysis-gluconeogenesis in the liver of gilthead seabream (Sparus aurata), Aquaculture, 2003, vol. 225, nos. 1−4, pp. 99–107.https://doi.org/10.1016/S0044-8486(03)00281-3
Minari, J., Josephine, O.A., and Balikis, O. S., Status of expression of GnRH gene in fed, starved, and refed Clarias gariepinus (African catfish), Adv. Technol., 2022, vol. 2, no. 4, pp. 405–417. https://doi.org/10.31357/ait.v2i4.6032
Miwa, S., Yan, L., and Swanson, P., Localization of two gonadotropin receptors in the salmon gonad by in vitro ligand autoradiography, Biol. Reprod., 1994, vol. 50, no. 3, pp. 629–642. https://doi.org/10.1095/biolreprod50.3.629
Moon, T.W. and Johnston, I.A., Starvation and the activities of glycolytic and gluconeogenic enzymes in skeletal muscles and liver of the plaice, Pleuronectes platessa, J. Comp. Physiol. B., 1980, vol. 136, no. 1, pp. 31–38. https://doi.org/10.1007/BF00688619
Narnaware, Y.K. and Peter, R.E., Effects of food deprivation and refeeding on neuropeptide Y (NPY) mRNA levels in goldfish, Comp. Biochem. Physiol. B. Biochem. Mol. Biol., 2001, vol. 129, nos. 2−3, pp. 633–637. https://doi.org/10.1016/s1096-4959(01)00359-1
Navarro, I. and Gutiérrez, J., Fasting and starvation, Biochem. Mol. Biol. Fish., 1995, vol. 4, pp. 393–434. https://doi.org/10.1016/S1873-0140(06)80020-2
Oba, Y., Hirai, T., Yoshiura, Y., et al., The duality of fish gonadotropin receptors: Cloning and functional characterization of a second gonadotropin receptor cDNA expressed in the ovary and testis of amago salmon (Oncorhynchus rhodurus), Biochem. Biophys. Res. Commun., 1999, vol. 265, no. 2, pp. 366–371. https://doi.org/10.1006/bbrc.1999.1700
Oba, Y., Hirai, T., Yoshiura, Y., et al., Fish gonadotropin and thyrotropin receptors: The evolution of glycoprotein hormone receptors in vertebrates, Comp. Biochem. Physiol. Part B Biochem. Mol. Biol., 2001, vol. 129, nos. 2−3, pp. 441–448. https://doi.org/10.1016/s1096-4959(01)00374-8
Park, J.W., Kwon, Y.J., Jin, H.Y., and Oh, S., Effects of fasting on brain of kiss2 and GnRH I and plasma levels of sex steroid hormones, in Nile tilapia Oreochromis niloticus, Ocean Polar Res., 2016, vol. 38, no. 1, pp. 81−88. https://doi.org/10.4217/OPR.2016.38.1.081
Pavlov, D.S., Pavlov, E.D., and Ganzha, E.V., Change of rheoreaction and concentration of thyroid hormones in blood of juvenile rainbow trout Oncorhynchus mykiss during starvation, J. Ichthyol., 2020, vol. 60, pp. 325–330. https://doi.org/10.1134/S0032945220020149
Pérez-Jiménez, J., Neveu, V., and Vos, F., Identification of the 100 richest dietary sources of polyphenols: an application of the Phenol-Explorer database, Eur. J. Clin. Nutr. 2010, vol. 64, no. 3, pp. S112–S120. https://doi.org/10.1038/ejcn.2010.221
Peterson, B.C. and Small, B.C., Effects of fasting on circulating IGF-binding proteins, glucose, and cortisol in channel catfish (Ictalurus punctatus), Domest. Anim. Endocrinol., 2004, vol. 26, no. 3, pp. 231–240. https://doi.org/10.1016/j.domaniend.2003.10.005
Pikle, R.P., Jatiger, R.M., and Ganesh, C.B., Food-deprivation-induced suppression of pituitary-testicular-axis in the tilapia Oreochromis mossambicus, Int. Aquat. Res., 2017, vol. 9, no. 3, pp. 203–213. https://doi.org/10.1007/s40071-017-0169-y
Pronina, G.I. and Revyakin, A.O., Changes of the morphophysiological parameters of carp Cyprinus carpio at food limitation in aquaculture conditions, J. Ichthyol., 2015, vol. 55, pp. 297–301. https://doi.org/10.1134/S0032945215020162
Polakof, S., Arjona, F.J., Sangiao-Alvarellos, S., et al., Food deprivation alters osmoregulatory and metabolic responses to salinity acclimation in gilthead sea bream Sparus auratus, J. Comp. Physiol. B. Biochem. Syst. Environ. Physiol., 2006, vol. 176, no. 5, pp. 441–452. https://doi.org/10.1007/s00360-006-0065-z
Power, D.M., Melo, J., and Santos, C.R.A, The effect of food deprivation and refeeding on the liver, thyroid hormones and transthyretin in sea bream, J. Fish Biol., 2000, vol. 56, no. 2, pp. 374–387.https://doi.org/10.1111/j.1095-8649.2000.tb02112.x
Rice, C.D., Restricted feeding, spermatogenesis and growth in Arctic Charr (Salvelinus alpinus). The identification of two possible gametogenic control points, MS Thesis, St. John’s: Memorial University of Newfoundland, 1999. http://research.library.mun.ca/id/eprint/997
Rocha, A., Gómez, A., Zanuy, S., et al., Molecular characterization of two sea bass gonadotropin receptors: cDNA cloning, expression analysis, and functional activity, Mol. Cell Endocrinol., 2007, vol. 272, nos. 1−2, pp. 63–76. https://doi.org/10.1016/j.mce.2007.04.007
Sakyi, M.E., Cai, J., Tang, J., et al., Short term starvation and re-feeding in Nile tilapia (Oreochromis niloticus, Linnaeus 1758): Growth measurements, and immune responses, Aquacult. Rep., 2020, vol. 16, Article 100261.https://doi.org/10.1016/j.aqrep.2019.100261
Sales, C.F., Barbosa Pinheiro, A.P., Ribeiro, Y.M., et al., Effects of starvation and refeeding cycles on spermatogenesis and sex steroids in the Nile tilapia Oreochromis niloticus, Mol. Cell. Endocrinol., 2020, vol. 500, Article 110643. https://doi.org/10.1016/j.mce.2019.110643
Sambroni, E., Le Gac, F., Breton, B., and Lareyre, J.J., Functional specificity of the rainbow trout (Oncorhynchus mykiss) gonadotropin receptors as assayed in a mammalian cell line, J. Endocrinol., 2007, vol. 195, no. 2, pp. 213–228. https://doi.org/10.1677/JOE-06-0122
Sasayama, Y. and Takahashi, H., Effect of starvation and unilateral castration in male goldfish (Carassius auratus), and a design of bioassay for fish gonadotropin using starved goldfish, Bull. Fac. Fish. Hokkaido. Univ., 1972, vol. 22, pp. 267–279
Schulz, R.W., de França, L.R., Lareyre, J.J., et al., Spermatogenesis in fish, Gen. Comp. Endocrinol., 2010, vol. 165, no. 3, pp. 390–411. https://doi.org/10.1016/j.ygcen.2009.02.013
Shinde, D. and Ganesh, C.B., Chronic exposure to aquacultural stressors affects pituitary-testis axis in the Mozambique tilapia Oreochromis mossambicus, Fish Physiol. Biochem., 2022, vol. 48, no. 2, pp. 437– 448. https://doi.org/10.1007/s10695-022-01061-9
Silva, W., de Souza, E., Hisano, H., and Mattioli, C.C., Effects of cyclical short-term fasting and refeeding on juvenile Lophiosilurus alexandri, a carnivorous Neotropical catfish, Aquaculture, 2019, vol. 505, pp. 12−17. https://doi.org/10.1016/j.aquaculture.2019.02
So, W.K., Kwok, H.F., and Ge, W., Zebrafish gonadotropins and their receptors: II. Cloning and characterization of zebrafish follicle-stimulating hormone and luteinizing hormone subunits: their spatial-temporal expression patterns and receptor specificity, Biol. Reprod., 2005, vol. 72, no. 6, pp. 1382–1396. https://doi.org/10.1095/biolreprod.104.038216
Sohn, Y.C., Suetake, H., Yoshiura, Y., et al., Structural and expression analyses of gonadotropin-I beta subunit genes in goldfish (Carassius auratus), Gene, 1998, vol. 222, no. 2, pp. 257–267. https://doi.org/10.1016/s0378-1119(98)00505-8
Suchiang, P. and Gupta, B. B. P., Effects of partial and full feed restriction on the plasma levels of thyroid hormones and testicular activity in the male air-breathing catfish, Clarias gariepinus during different phases of the breeding cycle, Int. J. Biol., 2011, vol. 3, no. 2, pp. 32−42.https://doi.org/10.5539/ijb.v3n2p32
Takle, H. and Andersen, Ø., Caspases and apoptosis in fish, J. Fish Biol., 2007, vol. 71, pp. 326–349. https://doi.org/10.1111/j.1095-8649.2007.01665.x
Townshend, T.J. and Wootton, R.J., Effects of food supply on the reproduction of the convict cichlid, Cichlasoma nigrofasciatum, Ibid., 1984, vol. 24, no. 1, pp. 91–104.
Tuziak, S.M. and Volkoff, H., Gonadotrophin-releasing hormone in winter flounder (Pseudopleuronectes americanus): Molecular characterization, distribution and effects of fasting, Gen. Comp. Endocrinol., 2013, vol. 184, pp. 9–21. https://doi.org/10.1016/j.ygcen.2012.12.010
Viegas, I., Rito, J., González, J.D., et al., Effects of food-deprivation and refeeding on the regulation and sources of blood glucose appearance in European seabass (Dicentrarchus labrax L.)., Comp. Biochem. Physiol. Part A. Mol. Integr. Physiol., 2013, vol. 166, no. 3, pp. 399–405.https://doi.org/10.1016/j.cbpa.2013.07.013
Vijayalaxmi and Ganesh, C.B., Distribution of endomorphin-like-immunoreactive neurones in the brain of the cichlid fish Oreochromis mossambicus, J. Neuroendocrinol., 2017, vol. 29, no. 3. https://doi.org/10.1111/jne.12460
Vijayalaxmi, Shyamannavar, B., Patil, et al., Effect of food deprivation on neuropeptide Y immunoreactivity and concurrent biochemical and gonadal responses in tilapia Oreochromis mossambicus (Peters, 1852), Indian J. Fish., 2019, vol. 66, no. 3, pp. 95−101. https://doi.org/10.21077/ijf.2019.66.3.81168-12
Vilela, D., Silva, S., Peixoto, M., et al., Spermatogenesis in teleost: Insights from the Nile tilapia (Oreochromis niloticus) model, Fish Physiol. Biochem., 2003, vol. 28, pp. 187–190. https://doi.org/10.1023/B:FISH.0000030523.16010.62
Vischer, H.F., Granneman, J.C.M., Linskens, M.H.K., et al., Both recombinant African catfish LH and FSH are able to activate the African catfish FSH receptor, J. Mol. Endocrinol., 2003, vol. 31, no. 1, pp. 133–140. https://doi.org/10.1677/jme.0.031013
Vosylienė, M.Z. and Kazlauskienė, N., Alterations in fish health state parameters after exposure to different stressors, Acta Zool. Lituanica, 1999, vol. 9, no. 2, pp. 83–94. https://doi.org/10.1080/13921657.1999.10512291
Waagbø, R., Jørgensen, SM., Timmerhaus, G., et al., Short-term starvation at low temperature prior to harvest does not impact the health and acute stress response of adult Atlantic salmon, PeerJ., 2017, vol. 5. pp. 1−22. https://doi.org/10.7717/peerj.3273
Wang, T., Zhou, C., Yuan, D., et al., Schizothorax prenanti corticotropin-releasing hormone (CRH): Molecular cloning, tissue expression, and the function of feeding regulation, Fish Physiol. Biochem., 2014, vol. 40, no. 5, pp. 1407–1415. https://doi.org/10.1007/s10695-014-9935-6
Wang, J., Du, J.J., Jiang, B., et al., Effects of short-term fasting on the resistance of Nile tilapia (Oreochromis niloticus) to Streptococcus agalactiae infection, Fish Shellfish Immunol., 2019, vol. 94, pp. 889−895. https://doi.org/10.1016/j.fsi.2019.09.055
Yarmohammadi, M., Shabani, A., Pourkazemi, M., et al., Effect of starvation and re-feeding on growth performance and content of plasma lipids, glucose and insulin in cultured juvenile Persian sturgeon (Acipenser persicus Borodin, 1897), J. Appl. Ichthyol., 2012, vol. 28, no. 5, pp. 692–696. https://doi.org/10.1111/j.1439-0426.2012.01969.x
Yaron, Z., Gur, G., Melamed, P., et al., Regulation of fish gonadotropins, Int. Rev. Cytol., 2003, vol. 129, pp. 131–185. https://doi.org/10.1016/s0074-7696(05)25004-0
Funding
The first author is grateful to Karnatak University, Dharwad for providing University Research Studentship (URS/2021/626). The infrastructural facility received from Science and Engineering Research Board–Department of Science and Technology (SERB-DST), New Delhi, Government of India is thankfully acknowledged.
Author information
Authors and Affiliations
Contributions
Methodology, formal analysis and investigation, original draft preparation (M. Gouda); Conceptualization, supervision, review and editing (C.B. Ganesh).
Corresponding author
Ethics declarations
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The experimental procedures were carried out according to Institutional Animal Ethics Committee (IAEC) guidelines (No. 639/GO/Re/S/02/CPCSEA).
CONFLICT OF INTEREST
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gouda, M., Ganesh, C.B. Chronic Starvation-Induced Alterations in Biochemical Parameters and Hypothalamic-Pituitary-Testicular Axis in the Mozambique Tilapia Oreochromis mossambicus (Cichlidae). J. Ichthyol. 63, 1172–1187 (2023). https://doi.org/10.1134/S003294522306005X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S003294522306005X