Abstract
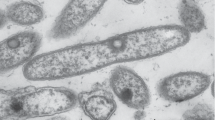
A new facultative methylotroph, strain Side1T, was isolated from the phyllosphere of Bougainvillea sp. L. The isolate is represented by rod-shaped, aerobic gram-negative asporogenous bacteria which divide by binary fission. Methanol and mono- and trimethylamine were utilized, as well as a limited spectrum of polycarbon substrates, while methane and dichloromethane were not used. Growth occurred at pH 6.0–9.0 with the optimum at pH 7.0 within the temperature range from 20 to 40°C (optimum at 28–30°C) and 0–2.5% NaCl in the medium. The predominant fatty acids were cis-11-octadecenoic (C18:1ω7c), 11-methyl-octadecenoic (C18:ω7c11Me), and stearic (C18:0) acids. Phosphatidylethanolamine, phosphatidylcholine, phosphatidylglycerol, and diphosphatidylglycerol were the dominant phospholipids. Q10 was the dominant ubiquinone. The isolate oxidized methanol and methylamine by the appropriate dehydrogenases. The isocitrate lyase-negative variant of the serine pathway was used. Ammonium assimilation involved glutamate dehydrogenase and the glutamate cycle (glutamate synthase and glutamine synthetase). The strain synthesized indole and siderophores; it solubilized insoluble phosphates. The DNA G+C content (T m) was 65.4 mol %. While the nucleotide sequence of the 16S rRNA gene of strain Side1 exhibited high similarity to those of Methylopila species (M. musalis MUSAT and M. capsulata IM1T), DNA-DNA homology with these cultures was 32–37%. The results obtained supported classification of strain Side1T as a new species Methylopila turkiensis sp. nov. (VKM B-2748T= DSM 27566T).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Fedorov, D.N., Doronina, N.V., and Trotsenko, Yu.A., Phytosymbiosis of aerobic methylobacteria: new facts and views, Microbiology (Moscow), 2011, vol. 80, pp. 443–454.
Kolb, S., Aerobic methanol-oxidizing bacteria in soil, FEMS Microbiol. Lett., 2009, vol. 300, pp. 1–10.
Doronina, N.V., Trotsenko, Yu.A., Krausova, V.I., Boulygina, E.S., and Tourova, T.P., Methylopila capsulata gen. nov., sp. nov., a novel non-pigmented aerobic facultatively methylotrophic bacterium, Int. J. Syst. Bacteriol., 1998, vol. 48, pp. 1313–1321.
Doronina, N.V., Trotsenko, Yu.A., Tourova, T.P., Kuznetsov, B.B., and Leisinger, T., Methylopila helvetica sp. nov. and Methylobacterium dichloromethanicum sp. nov.-novel aerobic facultatively methylotrophic bacteria utilizing dichloromethane, Syst. Appl. Microbiol., 2000, vol. 23, pp. 210–218.
Li, L., Zheng, J.-W., Hang, B.-J., Doronina, N.V., Trotsenko, Yu.A., He, J., and Li, S.-P., Methylopila jiangsuensis sp. nov., an aerobic, facultatively methylotrophic bacterium, Int. J. Syst. Evol. Microbiol., 2011, vol. 61, pp. 1561–1566.
Doronina, N.V., Kaparullina, E.N., Bykova, T.V., and Trotsenko, Yu.A., Methylopila musalis sp. nov., a new aerobic facultatively methylotrophic bacterium isolated from banana fruit, Int. J. Syst. Evol. Microbiol., 2013, vol. 63, pp. 1847–1852.
Poroshina, M.N., Doronina, N.V., Kaparullina, E.N., Kovalevskaya, N.P., and Trotsenko, Yu.A., Halophilic and halotolerant aerobic methylobacteria from the technogenic Solikamsk biotopes, Microbiology (Moscow), 2013, vol. 82, pp. 490–498.
Doronina, N.V., Braus-Stromeyer, S.A., Leisinger, T., and Trotsenko, Y.A., Isolation and characterization of a new facultatively methylotrophic bacterium: description of Methylorhabdus multivorans, gen. nov., sp. nov., Syst. Appl. Microbiol., 1995, vol. 18, pp. 92–98.
Gordon, S.A. and Weber, R.P., Colorimetric estimation of indoleacetic acid, Plant Physiol., 1951, vol. 26, pp. 192–195.
Agafonova, N.V., Kaparullina, E.N., Doronina, N.V., and Trotsenko, Yu.A., Phosphate-solubilizing activity of aerobic methylobacteria, Microbiology (Moscow), 2013, vol. 82, pp. 864–867.
Schwyn, B. and Neilands, J.B., Universal chemical assay for the detection and determination of siderophores, Anal. Biochem., 1987, vol. 160, pp. 47–56.
Arnow, L.E., Colorimetric determination of the components of 3,4-dihydroxyphenylalanine-tyrosine mixtures, J. Biol. Chem., 1937, vol. 118, pp. 531–537.
Doronina, N.V., Trotsenko, Y.A., and Tourova, T.P., Methylarcula marina gen. nov., sp. nov. and Methylarcula terricola sp. nov.: novel aerobic, moderately halophilic, facultatively methylotrophic bacteria from coastal saline environments, Int. J. Syst. Evol. Microbiol., 2000, vol. 50, pp. 1849–1859.
Collins, M.D., Analysis of isoprenoid quinones, in Methods in Microbiology, Gottschalk, G., Ed., New York: Academic, 1985, vol. 18, pp. 329–366.
Sasser, M., Identification of bacteria by gas chromatography of fatty acids, MIDI Technical Note 101, Newark, DE: MIDI, Inc., 1990.
Doronina, N.V., Gogleva, A.A., and Trotsenko, Y.A., Methylophilus glucosoxydans sp. nov., a restricted facultative methylotroph from rice rhizosphere, Int. J. Syst. Evol. Microbiol., 2012, vol. 62, pp. 196–201.
Trotsenko, Y.A., Doronina, N.V., and Govorukhina, N.I., Metabolism of non-motile obligately methylotrophic bacteria, FEMS Microbiol. Lett., 1986, vol. 3, pp. 293–297.
Lowry, O.H., Rosebrough, N.J., Farr, A.L., and Randall, R.J., Protein measurement with the Folin phenol reagent, J. Biol. Chem., 1951, vol. 193, pp. 265–275.
Owen, R.J. and Lapage, S.P., The thermal denaturation of partly purified bacterial deoxyribonucleic acid and its taxonomic applications, J. Appl. Bacteriol., 1976, vol. 41, pp. 335–340.
Doronina, N.V., Govorukhina, N.I., Lysenko, A.M., and Trotsenko, Y.A., Analysis of DNA-DNA homology in obligately methylotrophic bacteria, Microbiology (Moscow), 1988, vol. 57, pp. 629–633.
Lane, D.J., 16S/23S rRNA sequencing, in Nucleic Acid Techniques in Bacterial Systematics, Stackebrandt, E. and Goodfellow, M., Eds., Chichester: Wiley, 1991, pp. 115–175.
McDonald, I.R. and Murrell, J.C., The methanol dehydrogenase structural gene mxaF and its use as a functional gene probe for methanotrophs and methylotrophs, Appl. Environ. Microbiol., 1997, vol. 63, pp. 3218–3224.
Neufeld, J.D., Schafer, H., Cox, M.J., Boden, R., McDonald, I.R., and Murrell, J.C., Stable-isotope probing implicates Methylophaga spp. and novel Gammaproteobacteria in marine methanol and methylamine metabolism, Int. Soc. Microb. Ecol. J., 2007, vol. 1, pp. 480–491.
Van de Peer, Y. and De Wachter, R., TREECON for Windows: a software package for the construction and drawing of evolutionary trees for the Microsoft Windows environment, Comput. Appl. Biosci., 1994, vol. 10, pp. 569–570.
Horneffer, V., Haverkamp, J., Janssen, H.G., Steeg, P.F., and Notz, R., MALDI-TOF-MS analysis of bacterial spores: wet heat-treatment as a new releasing technique for biomarkers and the influence of different experimental parameters and microbiological handling, J. Am. Soc. Mass. Spectrom., 2004, vol. 15, pp. 1444–1454.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © N.V. Agafonova, E.N. Kaparullina, N.V. Doronina, Yu.A. Trotsenko, 2015, published in Mikrobiologiya, 2015, Vol. 84, No. 4, pp. 456–465.
Rights and permissions
About this article
Cite this article
Agafonova, N.V., Kaparullina, E.N., Doronina, N.V. et al. Methylopila turkiensis sp. nov., a new aerobic facultatively methylotrophic phytosymbiont. Microbiology 84, 544–552 (2015). https://doi.org/10.1134/S0026261715040025
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026261715040025