Abstract
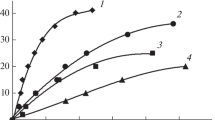
The use of boric acid (H3BO3) and boron oxide (B2O3) for the synthesis of cobalt-based catalysts by the co-precipitation technique was investigated and catalytic activities in hydrogen generation were evaluated. Different cobalt salts [cobalt (II) chloride (CoCl2 · 6H2O), cobalt sulfate (CoSO4 · 5H2O) and cobalt(II) nitrate (Co(NO3)2 · 7H2O)] were used with H3BO3 and B2O3 to prepare Co based catalysts. Crystalline, surface and chemical characteristics were clarified using X-ray diffraction (XRD); low temperature adsorption of nitrogen (BET), scanning electron microscopy (SEM), and inductively coupled plasma optical emission spectroscopy (ICP-OES). Three types of powder samples were obtained according to the different boron sources and cobalt salts, and it was found that an efficient Co based catalyst was obtained by co-precipitation of B2O3 and CoCl2 · 6H2O salt. Additionally, the effect of temperature, stabilizer ratio and NaBH4/catalyst ratio on parameters, characterizing the reaction of hydrogen generation was investigated. The zero order, first order and Langmuir-Hinshelwood kinetic models were used to identify the effect of Co based catalysts on the behavior of the catalytic system in hydrogen generation. Kinetic parameters of hydrogen generation for zero-order kinetic model were calculated for the following conditions: the rate of hydrogen generation is 0.93 L H2 g−1 catalyst min−1, the activation energy is 43.55 kJ mol−1 and the constant of Arrhenius equation is 11 min−1.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Yang, C.C., Chen, M.S., and Chen, Y.W., Int. J. Hydrogen Energy, 2011, vol. 36, no. 2, p. 1418.
Santos, D.M.F. and Sequeira, C.A.C., Renewable Sustainable Energy Rev., 2011, vol. 15, p. 3980.
Umegaki, T., Yan, J.M., Zhang, X.B., Shioyama, H., Kuriyama, N., and Xu, Q., Int. J. Hydrogen Energy, 2009, vol. 34, p. 2303.
Liu, C.H., Wua, Y.C., Chou, C.C., Chen, B.H., Hsueh, C.L., Ku, J.R., and Tsau, F., Int. J. Hydrogen Energy, 2012, vol. 37, p. 2950.
Jeong, S.U., Kim, R.K., Cho, E.A., Kim, H.J., Nam, S.W., Oh, I.H., Hong, S.A., and Kim, S.H., J. Power Sources, 2005, vol. 144, p. 129.
Amendola, S.C., Sharp-Goldman, S.L., Janjua, M.S., Spencer, N.C., Kelly, M.T., Petillo, P.J., and Binder, M., Int. J. Hydrogen Energy, 2000, vol. 25, p. 969.
Retnamma, R., Novais, A.Q., and Rangel, C.M., Int. J. Hydrogen Energy, 2011, vol. 36, p. 9772.
Akdim, O., Demirci, U.B., Muller, D., and Miele, P., Int. J. Hydrogen Energy, 2009, vol. 34, p. 2631.
Khan, R., Kim, S.W., Kim, T.J., and Nam, C.M., Mater. Chem. Phys., 2008, vol. 112, p. 167.
Shen, X., Dai, M., Gao, M., Zhao, B., and Ding, W., Chin. J. Catal., 2013, vol. 34, p. 979.
Hung, A.J., Tsai, S.F., Hsu, Y.Y., Ku, J.R., Chen, Y.H., and Yu, C.C., Int. J. Hydrogen Energy, 2008, vol. 33, p. 6205.
Fogler, S., Elements of Chem. Reaction Engineering, New Jersey: Prentice-Hall, 1999, 3rd ed.
Levenspiel, O., Chem. Reaction Engineering, New York: Wiley, 1999, 3rd ed.
Ozerova, A.M., Simagina, V.I., Komova, O.V., Netskina, O.V., Odegova, G.V., Bulavchenko, O.A., and Rudina, N.A., J. Alloys Compd., 2012, vol. 513, p. 266.
Kantürk Figen, A. and Coşkuner, B., Int. J. Hydrogen Energy, 2013, vol. 38, no. 6, p. 2824.
Kantürk Figen, A., Coşkuner, B., Pişkin, M.B., and Özdemir Dere, Ö., J. Int. Sci. Publ.: Mater., Met., 2013, vol. 7, no. 1, p. 43.
Coşkuner, B., Kantürk Figen, A., and Pişkin, S., Reac. Kinet. Mech. Catal., 2013, vol. 109, no. 2, p. 375.
Fernandes, R., Patel, N., Miotello, A., and Filippi, M., J. Mol. Catal. A: Chem., 2009, vol. 298, p. 1.
Cavaliere, S., Hannauer, J., Demirci, U.B., Akdim, O., and Miele, P., Catal. Today, 2011, vol. 170, p. 3.
Garron, A., Świerczyński, D., Bennici, S., and Auroux, A., Int. J. Hydrogen Energy, 2009, vol. 34, p. 1185.
Andrieux, J., Demirci, U.B., and Miele, P., Catal. Today, 2011, vol. 170, p. 13.
Kaufman, C.M. and Sen, B., J. Chem. Soc., Dalton Trans., 1985, p. 307.
Chamoun, R., Demirci, U.B., Zaatar, Y., Khoury, A., and Miele, P., Int. J. Hydrogen Energy, 2010, vol. 35, p. 6583.
Zhao, J., Ma, H., and Chen, J., Int. J. Hydrogen Energy, 2007, vol. 32, p. 4711.
Krishnan, P., Advani, S.G., and Prasad, A.K., Int. J. Hydrogen Energy, 2008, vol. 33, p. 7095.
Ding, X.L., Yuan, X., Jia, C., and Ma, Z.F., Int. J. Hydrogen Energy, 2010, vol. 35, p. 1107.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Coşkuner, B., Kantürk Figen, A. & Pişkin, M.B. The use of boric acid (H3BO3) and boron oxide (B2O3) for co-precipitation synthesis of cobalt-boron catalysts: Catalytic activity in hydrogen generation. Kinet Catal 55, 809–823 (2014). https://doi.org/10.1134/S002315841466001X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S002315841466001X