Abstract
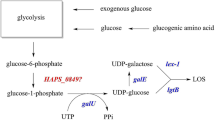
Current clinical treatments for pneumococcal infections have many limitations and are faced with many challenges. New capsular polysaccharide structures must be explored to cope with diseases caused by different serotypes of Streptococcus pneumoniae. UDP-galactose 4-epimerase (GalE) is an essential enzyme involved in polysaccharide synthesis. It is an important virulence factor in many bacterial pathogens. In this study, we found that two genes (galE sp1 and galE sp2 ) are responsible for galactose metabolism in pathogenic S. pneumoniae TIGR4. Both GalESp1 and GalESp2 were shown to catalyze the epimerization of UDP-glucose (UDP-Glc)/UDP-galactose (UDP-Gal), but only GalESp2 was shown to catalyze the epimerization of UDP-N-acetylglucosamine (UDP-GlcNAc)/UDP-N-acetylgalactosamine (UDP-GalNAc). Interestingly, GalESp2 had 3-fold higher epimerase activity toward UDP-Glc/UDP-Gal than GalESp1. The biochemical properties of GalESp2 were studied. GalESp2 was stable over a wide range of temperatures, between 30 and 70°C, at pH 8.0. The K86G substitution caused GalESp2 to lose its epimerase activity toward UDP-Glc and UDP-Gal; however, substitution C300Y in GalESp2 resulted in only decreased activity toward UDP-GlcNAc and UDP-GalNAc. These results indicate that the Lys86 residue plays a critical role in the activity and substrate specificity of GalESp2.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- CPS:
-
capsular polysaccharide
- GalE:
-
UDP-galactose 4-epimerase
- UDP-Gal:
-
UDP-galactose
- UDP-GalNAc:
-
UDP-N-acetylgalactosamine
- UDP-Glc:
-
UDP-glucose
- UDP-GlcNAc:
-
UDP-N-acetylglucosamine
References
Kadioglu, A., Weiser, J. N., Paton, J. C., and Andrew, P. W. (2008) The role of Streptococcus pneumoniae virulence factors in host respiratory colonization and disease, Nat. Rev. Microbiol., 6, 288–301.
Cartwright, K. (2002) Pneumococcal disease in Western Europe: burden of disease, antibiotic resistance and management, Eur. J. Pediatrics, 161, 188–195.
Winkelstein, J. A., Bocchini, J. A., Jr., and Schiffman, G. (1976) The role of the capsular polysaccharide in the activation of the alternative pathway by the pneumococcus, J. Immunol., 116, 367–370.
Austrian, R. (1981) Pneumococcus: the first one hundred years, Rev. Infect. Diseases, 3, 183–189.
Jiang, S. M., Wang, L., and Reeves, P. R. (2001) Molecular characterization of Streptococcus pneumoniae type 4, 6B, 8, and 18C capsular polysaccharide gene clusters, Infect. Immun., 69, 1244–1255.
Trzcinski, K., Thompson, C. M., and Lipsitch, M. (2003) Construction of otherwise isogenic serotype 6B, 7F, 14, and 19F capsular variants of Streptococcus pneumoniae strain TIGR4, Appl. Environ. Microbiol., 69, 7364–7370.
Frey, P. A. (1996) The Leloir pathway: a mechanistic imperative for three enzymes to change the stereochemical configuration of a single carbon in galactose, FASEB J., 10, 461–470.
Holden, H. M., Rayment, I., and Thoden, J. B. (2003) Structure and function of enzymes of the Leloir pathway for galactose metabolism, J. Biol. Chem., 278, 43885–43888.
Beerens, K., Soetaert, W., and Desmet, T. (2013) Characterization and mutational analysis of the UDP-Glc(NAc) 4-epimerase from Marinithermus hydrothermalis, Appl. Microbiol. Biotechnol., 97, 7733–7740.
Bengoechea, J. A., Pinta, E., Salminen, T., Oertelt, C., Holst, O., Radziejewska-Lebrecht, J., Piotrowska-Seget, Z., Venho, R., and Skurnik, M. (2002) Functional characterization of Gne (UDP-N-acetylglucosamine-4-epimerase), Wzz (chain length determinant), and Wzy (O-antigen polymerase) of Yersinia enterocolitica serotype O:8, J. Bacteriol., 184, 4277–4287.
Agarwal, S., Gopal, K., Upadhyaya, T., and Dixit, A. (2007) Biochemical and functional characterization of UDP-galactose 4-epimerase from Aeromonas hydrophila, Biochim. Biophys. Acta, 1774, 828–837.
Thoden, J. B., and Holden, H. M. (1998) Dramatic differences in the binding of UDP-galactose and UDP-glucose to UDP-galactose 4-epimerase from Escherichia coli, Biochemistry, 37, 11469–11477.
Bauer, A. J., Rayment, I., Frey, P. A., and Holden, H. M. (1992) The molecular structure of UDP-galactose 4-epimerase from Escherichia coli determined at 2.5 Å resolution, Proteins, 12, 372–381.
Brahma, A., and Bhattacharyya, D. (2004) UDP-galactose 4-epimerase from Kluyveromyces fragilis: existence of subunit independent functional site, FEBS Lett., 577, 27–34.
Alphey, M. S., Burton, A., Urbaniak, M. D., Boons, G. J., Ferguson, M. A., and Hunter, W. N. (2006) Trypanosoma brucei UDP-galactose-4′-epimerase in ternary complex with NAD+ and the substrate analogue UDP-4-deoxy-4-fluoro-alpha-D-galactose, Acta Crystallogr., 62, 829–834.
Shaw, M. P., Bond, C. S., Roper, J. R., Gourley, D. G., Ferguson, M. A., and Hunter, W. N. (2003) High-resolution crystal structure of Trypanosoma brucei UDP-galactose 4′-epimerase: a potential target for structure-based development of novel trypanocides, Mol. Biochem. Parasitol., 126, 173–180.
Daenzer, J. M., Sanders, R. D., Hang, D., and Fridovich-Keil, J. L. (2012) UDP-galactose 4′-epimerase activities toward UDP-Gal and UDP-GalNAc play different roles in the development of Drosophila melanogaster, PLoS Genet., 8, e1002721.
Thoden, J. B., Wohlers, T. M., Fridovich-Keil, J. L., and Holden, H. M. (2001) Human UDP-galactose 4-epimerase. Accommodation of UDP-N-acetylglucosamine within the active site, J. Biol. Chem., 276, 15131–15136.
Thoden, J. B., Wohlers, T. M., Fridovich-Keil, J. L., and Holden, H. M. (2000) Crystallographic evidence for Tyr 157 functioning as the active site base in human UDP-galactose 4-epimerase, Biochemistry, 39, 5691–5701.
Bradford, M. M. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding, Anal. Biochem., 72, 248–254.
Guo, H., Li, L., and Wang, P. G. (2006) Biochemical characterization of UDP-GlcNAc/Glc 4-epimerase from Escherichia coli O86:B7, Biochemistry, 45, 13760–13768.
Kowal, P., and Wang, P. G. (2002) New UDP-GlcNAc C4 epimerase involved in the biosynthesis of 2-acetamino-2-deoxy-L-altruronic acid in the O-antigen repeating units of Plesiomonas shigelloides O17, Biochemistry, 41, 15410–15414.
Ishiyama, N., Creuzenet, C., Lam, J. S., and Berghuis, A. M. (2004) Crystal structure of WbpP, a genuine UDP-N-acetylglucosamine 4-epimerase from Pseudomonas aeruginosa: substrate specificity in UDP-hexose 4-epimerases, J. Biol. Chem., 279, 22635–22642.
Ferrando, M. L., Baarlen, P. V., Orru, G., Piga, R., Bongers, R. S., Wels, M., Greeff, A. D., Smith, H. E., and Wells, J. M. (2014) Carbohydrate availability regulates virulence gene expression in streptococcus suis, PLoS One, 9, e89334.
Chung, S. K., Ryu, S. I., and Lee, S. B. (2012) Characterization of UDP-glucose 4-epimerase from Pyrococcus horikoshii: regeneration of UDP to produce UDP-galactose using two-enzyme system with trehalose, Biores. Technol., 110, 423–429.
Liden, M., Tryggvason, K., and Eriksson, U. (2003) Structure and function of retinol dehydrogenases of the short chain dehydrogenase/reductase family, Mol. Aspects Med., 24, 403–409.
Beerens, K., Soetaert, W., and Desmet, T. (2015) UDP-hexose 4-epimerases: a view on structure, mechanism and substrate specificity, Carbohydr. Res., 414, 8–14.
Thoden, J. B., Henderson, J. M., Fridovich-Keil, J. L., and Holden, H. M. (2002) Structural analysis of the Y299C mutant of Escherichia coli UDP-galactose 4-epimerase. Teaching an old dog new tricks, J. Biol. Chem., 277, 27528–27534.
Dalrymple, S. A., Ko, J., Sheoran, I., Kaminskyj, S. G., and Sanders, D. A. (2013) Elucidation of substrate specificity in Aspergillus nidulans UDP-galactose-4-epimerase, PLoS One, 8, e76803.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Biokhimiya, 2018, Vol. 83, No. 1, pp. 117-126.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Chen, L.L., Han, D.L., Zhai, Y.F. et al. Characterization and mutational analysis of two UDP-galactose 4-epimerases in Streptococcus pneumoniae TIGR4. Biochemistry Moscow 83, 37–44 (2018). https://doi.org/10.1134/S0006297918010054
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297918010054