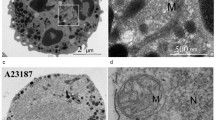
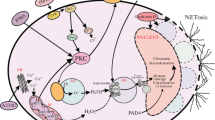
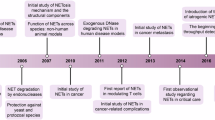
Abstract
Recent insights into the specific type of cell death characteristic of neutrophils, called NETosis, are summarized. NETosis is a process of generation of Neutrophil Extracellular Traps (NETs), whose main components are DNA, granular antimicrobial peptides, and nuclear and cytoplasmic proteins. The structure of NETs determines their bactericidal, fungicidal, antiprotozoal, and antiviral properties. Therefore, NETs production by neutrophils is an essential immune response to infection. In addition to the antimicrobial function, NETosis is involved in many inflammatory and autoimmune disorders and participates in the regulation of noninfectious processes. The molecular mechanisms of NET formation, bactericidal effect, and involvement in some noninfectious, autoimmune, and inflammatory processes are discussed in detail in this review.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- APC:
-
antigen-presenting cell
- CIC:
-
circulating immune complex
- IFN:
-
interferon
- LPS:
-
lipopolysaccharide
- MHC:
-
major histocompatibility complex
- pDC:
-
plasmacytoid dendritic cell
- TNF:
-
tumor necrosis factor
References
Brinkmann, V., Reichard, U., Goosmann, C., Fauler, B., Uhlemann, Y., Weiss, D. S., Weinrauch, Y., and Zychlinsky, A. (2004) Neutrophil extracellular traps kill bacteria, Science, 303, 1532–1535.
Steinberg, B. E., and Grinstein, S. (2007) Unconventional roles of the NADPH oxidase: signaling, ion homeostasis, and cell death, Sci. STKE, 379, pe11.
Yousefi, S., Gold, J. A., Andina, N., Lee, J. J., Kelly, A. M., Kozlowski, E., Schmid, I., Straumann, A., Reichenbach, J., Gleich, G. J., and Simon, H.-U. (2008) Catapult-like release of mitochondrial DNA by eosinophils contributes to antibacterial defense, Nature Med., 14, 949–953.
Von Kockritz-Blickwede, M., Goldmann, O., Thulin, P., Heinemann, K., Norrby-Teglund, A., Rohde, M., and Medina, E. (2008) Phagocytosis-independent antimicrobial activity of mast cells by means of extracellular trap formation, Blood, 111, 3070–3080.
Chuammitri, P., Ostojic, J., Andreasen, C. B., Redmond, S. B., Lamont, S. J., and Palic, D. (2009) Chicken heterophil extracellular traps (HETs): novel defense mechanism of chicken heterophils, Vet. Immunol. Immunopathol., 129, 126–131.
Chow, O. A., von Kockritz-Blickwede, M., Bright, A. T., Hensler, M. E., Zinkernagel, A. S., Cogen, A. L., Gallo, R. L., Monestier, M., Wang, Y., Glass, C. K., and Nizet, V. (2010) Statins enhance formation of phagocyte extracellular traps, Cell Host Microbe, 8, 445–454.
Altincicek, B., Stotzel, S., Wygrecka, M., Preissner, K. T., and Vilcinskas, A. (2008) Host-derived extracellular nucleic acids enhance innate immune responses, induce coagulation, and prolong survival upon infection in insects, J. Immunol., 181, 2705–2712.
Wen, F., White, G. J., VanEtten, H. D., Xiong, Z., and Hawes, M. C. (2009) Extracellular DNA is required for root tip resistance to fungal infection, Plant Physiol., 151, 820–829.
Hawes, M. C., Curlango-Rivera, G., Wen, F., White, G. J., Vanetten, H. D., and Xiong, Z. (2011) Extracellular DNA: the tip of root defenses? Plant Sci., 180, 741–745.
Urban, C. F., Ermert, D., Schmid, M., Abu-Abed, U., Goosmann, C., Nacken, W., Brinkmann, V., Jungblut, P. R., and Zychlinsky, A. (2009) Neutrophil extracellular traps contain calprotectin, a cytosolic protein complex involved in host defense against Candida albicans, PLoS Pathog., 5, e1000639.
Yousefi, S., Mihalache, C., Kozlowski, E., Schmid, I., and Simon, H. U. (2009) Viable neutrophils release mitochondrial DNA to form neutrophil extracellular traps, Cell Death Differ., 16, 1438–1444.
Maianski, N. A., Geissler, J., Srinivasula, S. M., Alnemri, E. S., Roos, D., and Kuijpers, T. W. (2004) Functional characterization of mitochondria in neutrophils: a role restricted to apoptosis, Cell Death Differ., 11, 143–153.
Pilsczek, F. H., Salina, D., Poon, K. K., Fahey, C., Yipp, B. G., Sibley, C. D., Robbins, S. M., Green, F. H., Surette, M. G., Sugai, M., Bowden, M. G., Hussain, M., Zhang, K., and Kubes, P. J. (2010) A novel mechanism of rapid nuclear neutrophil extracellular trap formation in response to Staphylococcus aureus, J. Immunol., 185, 7413–7425.
Fuchs, T. A., Abed, U., Goosmann, C., Hurwitz, R., Schulze, I., Wahn, V., Weinrauch, Y., Brinkmann, V., and Zychlinsky, A. (2007) Novel cell death program leads to neutrophil extracellular traps, J. Cell Biol., 176, 231–241.
Kessenbrock, K., Krumbholz, M., Schonermarck, U., Back, W., Gross, W. L., Werb, Z., Grone, H. J., Brinkmann, V., and Jenne, D. E. (2009) Netting neutrophils in autoimmune small-vessel vasculitis, Nature Med., 15, 623–625.
Garcia-Romo, G. S., Caielli, S., Vega, B., Connolly, J., Allantaz, F., Xu, Z., Punaro, M., Baisch, J., Guiducci, C., Coffman, R. L., Barrat, F. J., Banchereau, J., and Pascual, V. (2011) Netting neutrophils are major inducers of type I IFN production in pediatric systemic lupus erythematosus, Sci. Transl. Med., 3, 73ra20.
Lande, R., Ganguly, D., Facchinetti, V., Frasca, L., Conrad, C., Gregorio, J., Meller, S., Chamilos, G., Sebasigari, R., Riccieri, V., Bassett, R., Amuro, H., Fukuhara, S., Ito, T., Liu, Y. J., and Gilliet, M. (2011) Neutrophils activate plasmacytoid dendritic cells by releasing self-DNA-peptide complexes in systemic lupus erythematosus, Sci. Transl. Med., 3, 73ra19.
Neeli, I., Dwivedi, N., Khan, S., and Radic, M. (2009) Regulation of extracellular chromatin release from neutrophils, J. Innate Immun., 1, 194–201.
Lim, M. B. H., Kuiper, J. W. P., Katchky, A., Goldberg, H., and Glogauer, M. (2011) Rac2 is required for the formation of neutrophil extracellular traps, J. Leukoc. Biol., 90, 771–776.
Oehmcke, S., Morgelin, M., and Herwald, H. (2009) Activation of the human contact system on neutrophil extracellular traps, J. Innate Immun., 1, 225–230.
Guimaraes-Costa, A. B., Nascimento, M. T., Froment, G. S., Soares, R. P., Morgado, F. N., Conceiçao-Silva, F., and Saraiva, E. M. (2009) Leishmania amazonensis promastigotes induce and are killed by neutrophil extracellular traps, Proc. Natl. Acad. Sci. USA, 106, 6748–6753.
Clark, S. R., Ma, A. C., Tavener, S. A., McDonald, B., Goodarzi, Z., Kelly, M. M., Patel, K. D., Chakrabarti, S., McAvoy, E., Sinclair, G. D., Keys, E. M., Allen-Vercoe, E., Devinney, R., Doig, C. J., Green, F. H., and Kubes, P. (2007) Platelet TLR4 activates neutrophil extracellular traps to ensnare bacteria in septic blood, Nature Med., 13, 463–469.
Neeli, I., Khan, S. N., and Radic, M. (2008) Histone deimination as a response to inflammatory stimuli in neutrophils, J. Immunol., 180, 1895–1902.
Vorobjeva, N. V. (2013) NADPH oxidase of neutrophils and diseases associated with its dysfunction, Immunologiya, 34, 232–238.
Pinegin, B. V., and Mayansky, A. N. (2007) Neutrophils: structure and function, Immunologiya, 6, 374–382.
Bianchi, M., Hakkim, A., Brinkmann, V., Siler, U., Seger, R. A., Zychlinsky, A., and Reichenbach, J. (2009) Restoration of NET formation by gene therapy in CGD controls aspergillosis, Blood, 114, 2619–2622.
Hakkim, A., Fuchs, T. A., Martinez, N. E., Hess, S., Prinz, H., Zychlinsky, A., and Waldmann, H. (2011) Activation of the Raf-MEK-ERK pathway is required for neutrophil extracellular trap formation, Nature Chem. Biol., 7, 75–77.
Papayannopoulos, V., Metzler, K. D., Hakkim, A., and Zychlinsky, A. (2010) Neutrophil elastase and myeloperoxidase regulate the formation of neutrophil extracellular traps, J. Cell Biol., 191, 677–691.
Metzler, K. D., Fuchs, T. A., Nauseef, W. M., Reumaux, D., Roesler, J., Schulze, I., Wahn, V., Papayannopoulos, V., and Zychlinsky, A. (2011) Myeloperoxidase is required for neutrophil extracellular trap formation: implications for innate immunity, Blood, 117, 953–959.
Palmer, L. J., Cooper, P. R., Ling, M. R., Wright, H. J., Huissoon, A., and Chapple, I. L. C. (2012) Hypochlorous acid regulates neutrophil extracellular trap release in humans, Clin. Exp. Immunol., 167, 261–268.
Wang, Y., Li, M., Stadler, S., Correll, S., Li, P., Wang, D., Hayama, R., Leonelli, L., Han, H., Grigoryev, S. A., Allis, C. D., and Coonrod, S. A. (2009) Histone hypercitrullination mediates chromatin decondensation and neutrophil extracellular trap formation, J. Cell Biol., 184, 205–213.
Li, P., Li, M., Lindberg, M. R., Kennett, M. J., Xiong, N., and Wang, Y. (2010) PAD4 is essential for antibacterial innate immunity mediated by neutrophil extracellular traps, J. Exp. Med., 207, 1853–1862.
Hemmers, S., Teijaro, J. R., Arandjelovic, S., and Mowen, K. A. (2011) PAD4-mediated neutrophil extracellular trap formation is not required for immunity against influenza infection, PLoS ONE, 6, e22043.
Remijsen, Q., Vanden Berghe, T., Wirawan, E., Asselbergh, B., Parthoens, E., De Rycke, R., Noppen, S., Delforge, M., Willems, J., and Vandenabeele, P. (2011) Neutrophil extracellular trap cell death requires both autophagy and superoxide generation, Cell Res., 21, 290–304.
Farley, K., Stolley, J. M., Zhao, P., Cooley, J., and Remold-O’Donnell, E. (2012) A serpin B1 regulatory mechanism is essential for restricting neutrophil extracellular trap generation, J. Immunol., 189, 4574–4581.
Monfregola, J., Johnson, J. L., Meijler, M. M., Napolitano, G., and Catz, S. D. (2012) MUNC13-4 protein regulates the oxidative response and is essential for phagosomal maturation and bacterial killing in neutrophils, J. Biol. Chem., 287, 44603–44618.
Hakkim, A., Furnrohr, B. G., Amann, K., Laube, B., Abed, U. A., Brinkmann, V., Herrmann, M., Voll, R. E., and Zychlinsky, A. (2010) Impairment of neutrophil extra-cellular trap degradation is associated with lupus nephritis, Proc. Natl. Acad. Sci. USA, 107, 9813–9818.
Brinkmann, V., and Zychlinsky, A. (2007) Beneficial suicide: why neutrophils die to make NETs, Nat. Rev. Microbiol., 5, 577–582.
Wartha, F., Beiter, K., Albiger, B., Fernebro, J., Zychlinsky, A., Normark, S., and Henriques-Normark, B. (2007) Capsule and D-alanylated lipoteichoic acids protect Streptococcus pneumoniae against neutrophil extracellular traps, Cell. Microbiol., 9, 1162–1171.
Sumby, P., Barbian, K. D., Gardner, D. J., Whitney, A. R., Welty, D. M., Long, R. D., Bailey, J. R., Parnell, M. J., Hoe, N. P., Adams, G. G., Deleo, F. R., and Musser, J. M. (2005) Extracellular deoxyribonuclease made by group A Streptococcus assists pathogenesis by enhancing evasion of the innate immune response, Proc. Natl. Acad. Sci. USA, 102, 1679–1684.
Buchanan, J. T., Simpson, A. J., Aziz, R. K., Liu, G. Y., Kristian, S. A., Kotb, M., Feramisco, J., and Nizet, V. (2006) DNase expression allows the pathogen group A Streptococcus to escape killing in neutrophil extracellular traps, Curr. Biol., 16, 396–400.
Berends, E. T. M., Horswill, A. R., Haste, N. M., Monestier, M., Nizet, V., and von Kockritz-Blickwede, M. (2010) Nuclease expression by Staphylococcus aureus facilitates escape from neutrophil extracellular traps, J. Innate Immun., 2, 576–586.
Beiter, K., Wartha, F., Albiger, B., Normark, S., Zychlinsky, A., and Henriques-Normark, B. (2006) An endonuclease allows Streptococcus pneumoniae to escape from neutrophil extracellular traps, Curr. Biol., 16, 401–407.
Weinrauch, Y., Drujan, D., Shapiro, S. D., Weiss, J., and Zychlinsky, A. (2002) Neutrophil elastase targets virulence factors of enterobacteria, Nature, 417, 91–94.
Averhoff, P., Kolbe, M., Zychlinsky, A., and Weinrauch, Y. (2008) Single residue determines the specificity of neutrophil elastase for Shigella virulence factors, J. Mol. Biol., 377, 1053–1066.
Parker, H., Albrett, A. M., Kettle, A. J., and Winterbourn, C. C. (2012) Myeloperoxidase associated with neutrophil extracellular traps is active and mediates bacterial killing in the presence of hydrogen peroxide, J. Leukoc. Biol., 91, 369–376.
Bianchi, M., Niemiec, M. J., Siler, U., Urban, C. F., and Reichenbach, J. (2011) Restoration of anti-Aspergillus defense by neutrophil extracellular traps in human chronic granulomatous disease after gene therapy is calprotectin dependent, J. Allergy Clin. Immunol., 127, 1243–1252.
Saitoh, T., Komano, J., Saitoh, Y., Misawa, T., Takahama, M., Kozaki, T., Uehata, T., Iwasaki, H., Omori, H., Yamaoka, S., Yamamoto, N., and Akira, S. (2012) Neutrophil extracellular traps mediate a host defense response to human immunodeficiency virus-1, Cell Host Microbe, 12, 109–116.
Fadeel, B. (2009) Babies born without safety NET, Blood, 113, 6270–6271.
Yost, C. C., Cody, M. J., Harris, E. S., Thornton, N. L., McInturff, A. M., Martinez, M. L., Chandler, N. B., Rodesch, C. K., Albertine, K. H., Petti, C. A., Weyrich, A. S., and Zimmerman, G. A. (2009) Impaired neutrophil extracellular trap (NET) formation: a novel innate immune deficiency of human neonates, Blood, 113, 6419–6427.
Yost, C. C., and Zimmerman, G. A. (2009) Response: gestational age as a factor in neutrophil extracellular trap formation, Blood, 114, 4911–4912.
Yoo, D. G., Winn, M., Pang, L., Moskowitz, S. M., Malech, H. L., Leto, T. L., and Rada, B. (2014) Release of cystic fibrosis airway inflammatory markers from Pseudomonas aeruginosa-stimulated human neutrophils involves NADPH oxidase-dependent extracellular DNA trap formation, J. Immunol., 192, 4728–4738.
Marcos, V., Zhou, Z., Yildirim, A. O., Bohla, A., Hector, A., Vitkov, L., Wiedenbauer, E. M., Krautgartner, W. D., Stoiber, W., Belohradsky, B. H., Rieber, N., Kormann, M., Koller, B., Roscher, A., Roos, D., Griese, M., Eickelberg, O., Doring, G., Mall, M. A., and Hartl, D. (2010) CXCR2 mediates NADPH oxidase-independent neutrophil extra-cellular trap formation in cystic fibrosis airway inflammation, Nat. Med., 16, 1018–1023.
Papayannopoulos, V., Staab, D., and Zychlinsky, A. (2011) Neutrophil elastase enhances sputum solubilization in cystic fibrosis patients receiving DNase therapy, PLoS One, 6, e28526.
Fuchs, T. A., Brill, A., Duerschmied, D., Schatzberg, D., Monestier, M., Myers, D. D., Jr., Wrobleski, S. K., Wakefield, T. W., Hartwig, J. H., and Wagner, D. D. (2010) Extracellular DNA traps promote thrombosis, Proc. Natl. Acad. Sci. USA, 107, 15880–15885.
Geddings, J. E., and Mackman, N. (2014) New players in hemostasis and thrombosis, Thromb. Haemost., 111, 570–574.
Farquharson, D., Butcher, J. P., and Culshaw, S. (2012) Periodontitis, porphyromonas, and the pathogenesis of rheumatoid arthritis, Mucosal Immunol., 5, 112–120.
Vitkov, L., Klappacher, M., Hannig, M., and Krautgartner, W. D. (2009) Extracellular neutrophil traps in periodontitis, J. Periodontal Res., 44, 664–672.
Yarilin, A. A. (2010) Immunology [in Russian], GEOTAR-Media, Moscow.
Lande, R., Gregorio, J., Facchinetti, V., Chatterjee, B., Wang, Y. H., Homey, B., Cao, W., Wang, Y. H., Su, B., Nestle, F. O., Zal, T., Mellman, I., Schroder, J. M., Liu, Y. J., and Gilliet, M. (2007) Plasmacytoid dendritic cells sense self-DNA coupled with antimicrobial peptide, Nature, 449, 564–569.
Ganguly, D., Chamilos, G., Lande, R., Gregorio, J., Meller, S., Facchinetti, V., Homey, B., Barrat, F. J., Zal, T., and Gilliet, M. (2009) Self-RNA-antimicrobial peptide complexes activate human dendritic cells through TLR7 and TLR8, J. Exp. Med., 206, 1983–1994.
Chamilos, G., Gregorio, J., Meller, S., Lande, R., Kontoyiannis, D. P., Modlin, R. L., and Gilliet, M. (2012) Cytosolic sensing of extracellular self-DNA transported into monocytes by the antimicrobial peptide LL37, Blood, 120, 3699–3707.
Singh, D., Qi, R., Jordan, J. L., San Mateo, L., and Kao, C. C. (2013) The human antimicrobial peptide LL-37, but not the mouse ortholog, mCRAMP, can stimulate signaling by poly(I:C) through a FPRL1-dependent pathway, J. Biol. Chem., 288, 8258–8268.
Lai, Y., Adhikarakunnathu, S., Bhardwaj, K., Ranjith-Kumar, C. T., Wen, Y., Jordan, J. L., Wu, L. H., Dragnea, B., San Mateo, L., and Kao, C. C. (2011) LL37 and cationic peptides enhance TLR3 signaling by viral double-stranded RNAs, PLoS One, 6, e26632.
Chuang, C. M., Monie, A., Wu, A., Mao, C. P., and Hung, C. F. (2009) Treatment with LL-37 peptide enhances anti-tumor effects induced by CpG oligodeoxynucleotides against ovarian cancer, Hum. Gene Ther., 20, 303–313.
Kahlenberg, J. M., and Kaplan, M. J. (2013) Little peptide, big effects: the role of LL-37 in inflammation and autoimmune disease, J. Immunol., 191, 4895–4901.
Kaplan, M. J. (2011) Neutrophils in the pathogenesis and manifestations of SLE, Nature Rev. Rheumatol., 27, 691–699.
Denny, M. F., Yalavarthi, S., Zhao, W., Thacker, S. G., Anderson, M., Sandy, A. R., McCune, W. J., and Kaplan, M. J. (2010) A distinct subset of proinflammatory neutrophils isolated from patients with systemic lupus erythematosus induces vascular damage and synthesizes type I IFNs, J. Immunol., 184, 3284–3297.
Budikhina, A. S., and Pinegin, B. V. (2008) Defensins — multifunctional human cationic peptides, Immunopatol. Allergol. Infektol., 2, 31–40.
Leffler, J., Martin, M., Gullstrand, B., Tyden, H., Lood, C., Truedsson, L., Bengtsson, A. A., and Blom, A. M. (2012) Neutrophil extracellular traps that are not degraded in systemic lupus erythematosus activate complement exacerbating the disease, J. Immunol., 188, 3522–3531.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © N. V. Vorobjeva, B. V. Pinegin, 2014, published in Biokhimiya, 2014, Vol. 79, No. 12, pp. 1580–1591.
Rights and permissions
About this article
Cite this article
Vorobjeva, N.V., Pinegin, B.V. Neutrophil Extracellular Traps: Mechanisms of formation and role in health and disease. Biochemistry Moscow 79, 1286–1296 (2014). https://doi.org/10.1134/S0006297914120025
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297914120025