Abstract
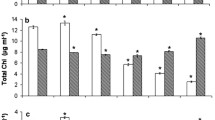
The effect of a typical polyaromatic hydrocarbon, naphthalene (Naph), on photosystem 2 (PS-2) photochemical activity in thylakoid membrane preparations and 20-day-old pea leaves was studied. Samples were incubated in water in the presence of Naph (0.078, 0.21, and 0.78 mM) for 0.5–24 h under white light illumination (15 μmol photons·m−2·s−1). The PS-2 activity was determined by studying fast and delayed chlorophyll (Chl) a fluorescence. Incubation of samples in water solutions at Naph concentrations of 0.21 and 0.78 mM led to a decrease in the maximum PS-2 quantum efficiency (Fv/Fm), noticeable changes in the polyphasic induction kinetics of fluorescence (OJIP), and a decrease in the amplitudes of the fast and slow components of delayed fluorescence of Chl a. The rate of release of electrolytes from leaves that were preliminarily incubated with Naph (0.21 mM) was also increased. Significant decrease in the fluorescence parameters in thylakoid membrane preparations was observed at Naph concentration of 0.03 mM and 12-min exposure of the samples. Chlorophyll (a and b) and carotenoid content (mg per gram wet mass) was insignificantly changed. The quantum yields of electron transfer from QA to QB (φET2o) and also to the PS-1 acceptors (φRE1o) were reduced. These results are explained by the increase in the number of QB-non-reducing centers of PS-2, which increased with increasing Naph concentration and exposure time of leaves in Naph solution. The suppression of PS-2 activity was partly abolished in the presence of the electron donor sodium ascorbate. Based on these results, it is suggested that Naph distorts cell membrane intactness and acts mainly on the PS-2 acceptor and to a lesser degree on the PS-2 donor side.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Abbreviations
- Asc:
-
sodium ascorbate
- Chl:
-
chlorophyll
- DFl:
-
delayed fluorescence
- Fo :
-
minimum fluorescence
- Fm :
-
maximum fluorescence
- Fv :
-
variable fluorescence (Fv = Fo − Fm)
- FFl:
-
fast fluorescence
- Naph:
-
naphthalene
- PA:
-
photosynthetic apparatus
- PAHs:
-
polycyclic aromatic hydrocarbons
- PS-2:
-
photosystem 2
- QA :
-
primary quinone acceptor of PS-2
- QB :
-
secondary quinone acceptor of PS-2
- RC:
-
reaction center
- TM:
-
thylakoid membranes
References
Fang, G.-C., Wu, Y.-S., Chen, J.-C., Chang, C.-N., and Ho., T.-T. (2006) Characteristic of polycyclic aromatic hydrocarbon concentrations and source identification for fine and coarse particulates at Taichung Harbor near Taiwan Strait during 2004–2005, Sci. Total Environ., 366, 729–738.
Simonich, S., and Hites, R. A. (1994) Importance of vegetation in removing polycyclic aromatic hydrocarbons from the atmosphere, Nature, 370, 49–51.
Chirkova, T. V. (2002) Physiological Basis of Plant Resistance [in Russian], St. Petersburg State University, St. Petersburg.
Kummerova, M., Krulova, J., Zezulka, S., and Triska, J. (2006) Evaluation of fluoranthene phytotoxicity in pea plants by Hill reaction and chlorophyll fluorescence, Chemosphere, 65, 489–496.
Kummerova, M., and Vanova, L. (2007) Chlorophyll fluorescence as an indicator of fluoranthene phototoxicity, Plant Soil Environ., 53, 430–436.
Jajoo, A., Rao, M. N., Tomar, R. S., Grieco, M., Tikkanen, M., and Aro, E.-M. (2014) Inhibitory effects of polycyclic aromatic hydrocarbons (PAHs) on photosynthetic performance are not related to their aromaticity, J. Photochem. Photobiol. B, 135, 151–155.
Bigler, W., and Schreiber, U. (1990) Chlorophyll luminescence as an indicator of stress-induced damage to the photosynthetic apparatus, Photosynth. Res., 25, 161–171.
Stirbet, A., and Govindjee (2011) On the relation between the Kautsky effect (chlorophyll a fluorescence induction) and photosystem II: basics and applications of the OJIP fluorescence transient, J. Photochem. Photobiol. B, 104, 36–57.
Kalaji, H. M., Golstev, V., Bosa, K., Allakhverdiev, S. I., Strasser, R. J., and Govindjee (2012) Experimental in vivo measurements of light emission in plants: a perspective dedicated to David Walker, Photosynth. Res., 114, 69–96.
Strasser, R. J., Srivastava, A., and Tsimilli-Michael, M. (2000) in Probing Photosynthesis: Mechanisms, Regulation and Adaptation (Yunus, M., Pathre, U., and Mohanty, P., eds.) Taylor and Francis, London, pp. 445–483.
Strasser, R. J., Tsimilli-Michael, M., and Srivastava, A. (2004) in Chlorophyll a Fluorescence: a Signature of Photosynthesis, Advances in Photosynthesis and Respiration (Papageorgiou, G. C., and Govindjee, eds.) Springer, Dordrecht, pp. 321–362.
Perlman, R. S., Yalkowsky, S. H., and Banerjee, S. (1984) Water solubilities of polynuclear aromatic and heteroaromatic compounds, J. Phys. Chem. Ref. Data, 13, 555–562.
Kreslavski, V. D., Lankin, A. V., Vasilyeva, G. K., Lyubimov, V. Yu., Semenova, G. N., Schmitt, F.-J., Friedrich, T., and Allakhverdiev, S. I. (2014) Effects of polyaromatic hydrocarbons on photosystem II activity in pea leaves, Plant Physiol. Biochem., 81, 135–142.
Mehta, P., Kreslavsky, V., Bharti, S., Allakhverdiev, S. I., and Jajoo, A. (2011) Analysis of salt stress induced changes in photosystem II heterogeneity by prompt fluorescence and delayed fluorescence in wheat (Triticum aestivum) leaves, J. Photochem. Photobiol. B, 104, 308–313.
Lichtenthaler, H. K., and Wellburn, A. R. (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes, Methods Enzymol., 148, 350–382.
Genty, B., Briantais, J-M., and Baker, N. R. (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence, Biochim. Biophys. Acta, 990, 87–92.
Liu, H., Weisman, D., Ye, Y. B., Cui, B., Huang, Y. H., Colon-Carmona, A., and Wang, Z. H. (2009) An oxidative stress response to polycyclic aromatic hydrocarbon exposure is rapid and complex in Arabidopsis thaliana, Plant Sci., 176, 357–382.
Bukhov, N. G., and Carpentier, R. (2000) Heterogeneity of photosystem II reaction centers as influenced by heat treatment of barley leaves, Physiol. Plant., 110, 279–285.
Singh-Tomar, R., and Jajoo, A. (2013) Alteration in PS II heterogeneity under the influence of polycyclic aromatic hydrocarbon (fluoranthene) in wheat leaves (Triticum aestivum), Plant Sci., 209, 58–63.
Jajoo, A., Mekala, N. R., Tomar, R. S., Grieco, M., Tikkanen, M., and Aro, E.-M. (2014) Inhibitory effects of polycyclic aromatic hydrocarbons (PAHs) on photosynthetic performance are not related to their aromaticity, J. Photochem. Photobiol. B, 137, 151–155.
Velthuys, B. R. (1981) Electron-dependent competition between plastoquinone and inhibitors for binding to photosystem II, FEBS Lett., 126, 277–281.
Smirnoff, N., and Wheeler, G. L. (2000) Ascorbic acid in plants: biosynthesis and function, Crit. Rev. Plant Sci., 19, 267–290.
Aksmann, A., and Tukaj, Z. (2008) Intact anthracene inhibits photosynthesis in algal cells: a fluorescence induction study on Chlamydomonas reinhardtii cw92 strain, Chemosphere, 74, 26–32.
Kummerova, M., Vanova, L., Krulova, J., and Zezulka, S. (2008) The use of physiological characteristics for comparison of organic compounds phytotoxicity, Chemosphere, 71, 2050–2059.
Van Gorkom, H. J. (1986) in Light Emission by Plants and Bacteria (Govindjee, Amesz, J., and Fork, D. C., eds.) Academic, Orlando, pp. 267–289.
Rathod, D. P., Brestic, M., and Shao, H. B. (2011) Chlorophyll a fluorescence determines the drought resistance capabilities in two varieties of mycorrhized and nonmycorrhized Glycine max Linn, Afr. J. Microbiol. Res., 5, 4197–4206.
Goltsev, V. N., Kuzmanova, M. A., Kaladzhi, H. M., and Allahverdiev, S. I. (2014) Variable and Delayed Fluorescence of Chlorophyll a Theoretical Basis and Practical Application in the Study of Plants [in Russian], Institute of Computer Science, Izhevsk-Moscow.
Goltsev, V., Zaharieva, I., Chernev, P., and Strasser, R. J. (2009) Delayed fluorescence in photosynthesis, Photosynth. Res., 101, 217–232.
Zhang, L., and Xing, D. (2008) Rapid determination of the damage to photosynthesis caused by salt and osmotic stresses using delayed fluorescence of chloroplasts, Photochem. Photobiol. Sci., 7, 352–360.
Chirkova, T. V. (1997) Cell membranes and plant resistance to stresses, Soros Obrazovat. Zh., 9, 12–17.
Duxbury, C. L., Dixon, D. G., and Greenberg, B. M. (1997) Effects of simulated solar radiation on the bioaccumulation of polycyclic aromatic hydrocarbons by the duckweed Lemna gibba, Environ. Toxicol. Chem., 16, 1739–1748.
Murata, N., Takahashi, S., Nishiyama, Y., and Allakhverdiev, S. I. (2007) Photoinhibition of photosystem II under environmental stress, Biochim. Biophys. Acta, 1767, 414–421.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Russian in Biokhimiya, 2014, Vol. 79, No. 11, pp. 1493–1504.
Originally published in Biochemistry (Moscow) On-Line Papers in Press, as Manuscript BM14-177, September 7, 2014.
Rights and permissions
About this article
Cite this article
Lankin, A.V., Kreslavski, V.D., Khudyakova, A.Y. et al. Effect of naphthalene on photosystem 2 photochemical activity of pea plants. Biochemistry Moscow 79, 1216–1225 (2014). https://doi.org/10.1134/S0006297914110091
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297914110091