Abstract
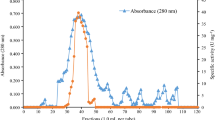
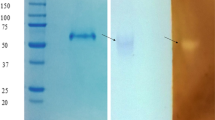
The extracellular amylolytic activity elaborated by the nematophagous fungus Arthrobotrys conoides was found to resolve into 2 amylolytic peaks when fractionated on Sephadex G-100 column. Around 80% of the eluted glucoamylase activity was attributed to peak I (GA A). GA A being cationic in nature was purified about 70-fold with 57% yield by negative chromatography on DEAE Sephadex at pH 7.0. The enzyme was stable over a broad pH range of 4.8–9.0. KM for the linear polysaccharide amylose was 0.34 mg/mL. Enzyme showed high affinity for the branched polysaccharides as the KM values for amylopectin, glycogen and starch were 0.056, 0.062 and 0.065 mg/mL, respectively. The enzyme clearly demonstrated raw starch digestibility. Probable involvement of Trp and His residues in enzyme catalysis was elucidated using group-specific reagents.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Pathak, E., El-Borai, F.E., Campos-Herrera, R., Johnson, E.G., Stuart, R.J., Graham. J.H., et al., Fungal Biol., 2012, vol. 116, no. 5, pp. 563–573.
Gomes, A.P.S., Araújo, J.V., and Ribeiro, R.C.F., Braz. J. Med. Biol. Res., 1999, vol. 32, no. 1, pp. 79–83.
Hanying, Y., Wei, X., and Xingxi, G., Afr. J. Biotechnol., 2012, vol. 11, no. 14, pp. 3370–3378.
Zouhar, M., Douda, O., Nováková, J., Doudová, E. Mazáková, J., Wenzlová, J., et al., Helminthologia, 2013, vol. 50, no. 2, pp. 127–131.
Pandit, R., Kunjadia, P., Mukhopadhyaya, P., and Kunjadia, A., J. Agricult. Technol., 2014, vol. 10, no. 3, pp. 559–570.
Futatsugi, M., Ogawa, T., and Fukuda, H., J. Ferment. Bioeng., 1993, vol. 76, no. 6, pp. 521–525.
Tanuja, S., Srinivas, N.D., Rao, R., and Gowthaman, M.K., Process Biochem., 1997, vol. 32, no. 8, pp. 635–641.
Ellaiah, P., Adinarayana, K., Bhavani, Y., Padmaja, P., and Srinivasulu B., Process Biochem., 2002, vol. 38, no. 4, pp. 615–620.
Kumar, S. and Satyanarayana, T., Biotechnol. Progress, 2003, vol. 19, no. 3, pp. 936–944.
Miller, G.L., Anal Chem., 1959, vol. 31, no. 3, pp. 426–428.
Dahlquist, A., Anal Biochem., 1964, vol. 7, pp. 18–25.
Jaffar, M.B., Bharata Ratnam, P., Norouzian, D., Irani, S.D., and Shetty, P., Indian J. Exp. Biol., 1993, vol. 31, no. 1, pp. 87–89.
Yu., R. and Hang, Y.D., Food Chem., 1991, vol. 40, pp. 301–308.
Cereia, M, Terenzi H.F., Jorge, J.A., Greene, L.J., Rosa, J.C., and Polizeli, M.L.T.M., J. Basic Microbiol., 2000, vol. 40, no. 2, pp. 83–92.
Tachibana, S. and Yasuda, M., Biosci. Biotechnol. Biochem., 2007, vol. 71, no. 10, pp. 2573–2576.
Ferreira-Nozawa, M.S., Rezende, J.L., Guimarães, L.H.S., Terenzi, H.F., Jorge, J.A., and Polizeli, M.L.T.M., Braz. J. Microbiol., 2008, vol. 39, pp. 344–352.
Koç, O. and Metin, K., Afr. J. Biotechnol., 2010, vol. 9, no. 23, pp. 3414–3424.
Bagheri, A., Khodarahmi, R., and Mostafaie, A., Food Chem., 2014, vol. 161, pp. 270–278.
Patel, M.N. and Wright, D.J., Parasitology, 1997, vol. 114, pp. 591–596.
Plant Nematology, Perry, R.N. and Moens, M., Eds., 2nd ed., Wallingford: Oxfordshire, UK, 2013.
Meagher, M.M., Nikholov, Z.L., and Reilly, P.J., Biotechnol. Bioeng., 1989, vol. 34, pp. 681–688.
Sauer, J., Sigurskjold, B.W., Christensen, U., Frandsen, T.P., Mirgorodskaya, E., Harrison, M., et al., Biochim. Biophys. Acta, 2000, vol. 1543, pp. 275–293.
Spande, T. F., Green, N. M., and Witkop, B., Biochemistry, 1966, vol. 5, no. 6, pp. 1926–1933.
Takahashi, K., J. Biochem. (Tokyo), 1970, vol. 68, no. 5, pp. 659–664.
Siksnys, V. and Pleckaityte, M., Biochim. Biophys. Acta, 1992, vol. 1160, no. 2, pp. 199–205.
Fu, H.W., Moomaw, J.F., Moomaw, C. R., and Casey, P.J., J. Biol. Chem., 1996, vol. 271, no. 45, pp. 28541–28548.
Yu, Y., Li, R., Xu, C., Ruan, K., Shen, Y., and Govindjee, Physiol. Plantarum, 2001, vol. 111, no. 1, pp. 108–115.
Miles, E.W., Methods Enzymol., 1977, vol. 47, pp. 431–442.
Colanduoni, J. and Villafrancal, J.J., J. Biol. Chem., 1985, vol. 280, no. 28, pp. 15042–15050.
Bhagwat, A.S. and Ramakrishna, J., Biochim. Biophys. Acta–Enzymology, 1981, vol. 662, no. 2, pp. 181–189.
Shenoy, B.C., Katwa, L.C., Appu Rao, A.G., and Rao, M.R., J. Biosci., 1985, vol. 7, no. 3/4, pp. 399–419.
Kiernan, J.A., Histological and Histochemical Methods. Theory and Practice, 2nd ed., Oxford (England): Pergamon Press, 1990.
Wong, S.S. and Wong, L.J., Enzyme Microb. Technol., 1992, vol. 14, no. 11, pp. 866–874.
Bisaria, V.S. and Mishra S., Crit. Rev. Biotechnol., 1989, vol. 9, no. 2, pp. 61–103.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Shetty, P. Glucoamylase from the Predacious Fungus Arthrobotrys conoides: a Cationic Enzyme with High Debranching Activity and Raw Starch Digestibility. Appl Biochem Microbiol 52, 176–182 (2016). https://doi.org/10.1134/S0003683816020150
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0003683816020150