Abstract
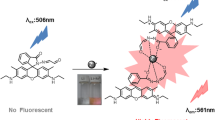
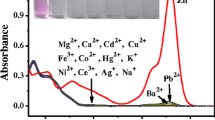
A novel fluorescent chemosensor based on trimesoyl chloride–rhodamine (TR) was successfully synthesized. Rising chromogenic and fluorogenic spectral enhancements could be observed in trimesoyl chloride–rhodamine (TR) probes when Hg2+ and Fe3+ were added, respectively. TR has shown selectivity for Hg2+ and Fe3+ with high sensitivity due to metal ion complexation induced photophysical “turn-on” signaling responses. The detection limit towards Hg2+ was 2.46 × 10−8 M as determined by the 3σ method. At the same time, fluorogenic spectral enhancements were observed in TR, which exhibits a superior sensitive and selective recognition towards Fe3+ with 4.11 × 10−8 M of the detection limit. The test strips were used for colorimetric and simple detection towards Hg2+, which might finally enable the advancement of the Hg2+ sensor in the field of on-site detection.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Notes and references
W. F. Fitzgerald, C. H. Lamborg, and C. R. Hammerschmidt, Marine biogeochemical cycling of mercury, Chem. Rev., 2007, 107, 641–662
T. W. Clarkson, and L. Magos, The toxicology of mercury and its chemical compounds, Crit. Rev. Toxicol., 2006, 36, 609–662
A. Renzoni, F. Zino, and E. Franchi, Mercury levels along the food chain and risk for exposed populations, Environ. Res., 1998, 77, 68–72
V. Dujols, F. Ford, and A. W. Czarnik, A long-wavelength fluorescent chemodosimeter selective for Cu(II) ion in water, J. Am. Chem. Soc., 1997, 119, 7386–7387
K. C. Behera, and B. Bag, Switch in ‘turn-on’ signaling preferences from Fe(III) to Hg(II) as a function of solvent and structural variation in rhodamine-based probes, Dyes Pigm., 2016, 135, 143–153
S. Squadrone, A. Benedetto, P. Brizio, M. Prearo, and M. Abete, Mercury and selenium in European catfish (Silurus glanis) from Northern Italian Rivers: can molar ratio be a predictive factor for mercury toxicity in a top predator, Chemosphere, 2015, 119, 24–30
M. Hong, X. Lu, Y. Chen, and D. Xu, A novel rhodamine-based colorimetric and fluorescent sensor for Hg2+ in water matrix and living cell, Sens. Actuators, B, 2016, 232, 28–36
X. Wu, Q. Niu, T. Li, Y. Cui, and S. Zhang, A highly sensitive, selective and turn-off fluorescent sensor based on phenylamine-oligothiophene derivative for rapid detection of Hg2+, J. Lumin., 2016, 175, 182–186
S. Sharma, N. Kaur, J. Singh, A. Singh, P. Raj, and S. Sankar, Salen decorated nanostructured ZnO chemosensor for the detection of mercuric ions (Hg2+), S, Sens. Actuators, B, 2016, 232, 712–721
J. Wang, and X. Qian, A series of polyamide receptor-based PET fluorescent sensor molecules: positively cooperative Hg2+ ion binding with high sensitivity, Org. Lett., 2006, 17, 3721–3724
L. Prodi, C. Bargossi, M. Montalti, N. Zaccheroni, N. Su, J. S. Bradshaw, R. M. Izatt, and P. B. Savage, An effective fluorescent chemosensor for mercury ions, J. Am. Chem. Soc., 2000, 122, 6769–6770
H. N. Kim, W. X. Ren, J. S. Kim, and J. Yoon, Fluorescent and colorimetric sensors for detection of lead, cadmium, and mercury ions, Chem. Soc. Rev., 2012, 41, 3210–3244
D. J. Shi, J. F. Zeng, J. Ye, M. Ni, Y. M. Chen, and M. Q. Chen, An effective fluorescent chemosensor for mercury ions, J. Chin. Chem. Soc., 2017, 64, 986–992
P. Mahato, S. Saha, E. Suresh, R. D. Liddo, P. P. Parnigotto, M. T. Conconi, M. K. Kesharwani, B. Ganguly, and A. Das, Ratiometric detection of Cr3+ and Hg2+ by a naphthalimide rhodamine based fluorescent probe, Inorg. Chem., 2012, 51, 1769–1777
H. R. Yang, C. M. Han, X. J. Zhu, Y. Liu, K. Y. Zhang, S. J. Liu, Q. Zhao, F. Y. Li, and W. Huang, Upconversion luminescent chemodosimeter based on NIR organic dye for monitoring methylmercury In Vivo, Adv. Funct. Mater., 2016, 26, 1945–1953
K. Kaur, S. Chaudhary, S. Singh, and S. Mehta, Azaindole modified imine moiety as fluorescent probe for highly sensitive detection of Fe3+ ions, Sens. Actuators, B, 2016, 232, 396–401
Y. Wang, M. Yang, M. Zheng, X. Zhao, Y. Xie, and J. Jin, 2-Pyridylthiazole derivative as ICT-based ratiometric fluorescent sensor for Fe(III), Tetrahedron Lett., 2016, 57, 2399–2402
J. F. Liu, and Y. Qian, A novel naphthalimide-rhodamine dye: Intramolecular fluorescence resonance energy transfer and ratiometric chemodosimeter for Hg2+ and Fe3+, Dyes Pigm., 2017, 136, 782–790
M. Y. Zhang, C. C. Shen, T. Jia, J. W. Qiu, H. Zhu, and Y. Gao, One-step synthesis of rhodamine-based Fe3+ fluorescent probes via mannich reaction and its application in living cell imaging, Spectrochim. Acta, Part A, 2020, 231, 118105–118112
E. L. Mackenzie, K. Iwasaki, and Y. Tsuji, Intracellular iron transport and storage: from molecular mechanisms to health implications, Antioxid. Redox Signal., 2008, 10, 997–1030
X. Cao, F. Zhang, Y. Bai, X. Ding, and W. Sun, A highly selective “Turn-on” fluorescent probe for detection of Fe3+ in cells, J. Fluoresc., 2019, 29, 425–434
Z. Yang, M. She, B. Yin, J. Cuo, Y. Zhang, and W. Sun, Three rhodamine-based “Off-On” chemosensors with high selectivity and sensitivity for Fe3+ imaging in living cells, J. Org. Chem., 2012, 77, 1143–1147
Y. Liu, C. X. Zhao, X. Y. Zhao, H. L. Liu, Y. B. Wang, Y. G. Du, and D. B. Wei, A selective N, N-dithenoyl-rhodamine based fluorescent probe for Fe3+ detection in aqueous and living cells, J. Environ. Sci., 2020, 90, 180–188
H. Wang, B. B. Chen, and S. Q. Zhu, Chip-based magnetic solid-phase microextraction online coupled with micro HPLC-ICPMS for the determination of mercury species in cells, Anal. Chem., 2016, 88, 796–802
G. Reddi, and C. Rao, Analytical techniques for the determination of precious metals in geological and related materials, Analyst, 1999, 124, 1531–1540
N. Amiri, M. K. Rofouei, and J. B. Ghasemi, Multivariate optimization, preconcentration and determination of mercury ions with (1-(p-acetyl phenyl)-3-(o-methylbenzoate)) triazene in aqueous samples using ICP-AES, Anal. Methods, 2016, 8, 1111–1119
S. Caroli, G. Forte, A. Iamiceli, and B. Galoppi, Determination of essential and potentially toxic trace elements in honey by inductively coupled plasma-based techniques, Talanta, 1999, 50, 327–336
W. S. Zhong, T. Ren, and L. J. Zhao, Determination of Pb (Lead), Cd (Cadmium), Cr (Chromium), Cu (Copper), and Ni (Nickel) in Chinese tea with high-resolution continuum source graphite furnace atomic absorption spectrometry, Food Drug Anal., 2016, 24, 46–55
N. Promphet, P. Rattanarat, R. Rangkupan, O. Chailapakul, and N. Rodthongkum, An electrochemical sensor based on graphene/polyaniline/polystyrene nanoporous fibers modified electrode for simultaneous determination of lead and cadmium, Sens. Actuators, B, 2015, 207, 526–534
B. Kaur, N. Kaur, and S. Kumar, Colorimetric metal ion sensors-a comprehensive review of the years 2011-2016, Coord. Chem. Rev., 2018, 358, 13–69
J. Li, D. Yim, W. D. Jang, and J. Yoon, Recent progress in the design and applications of fluorescence probes containing crown ethers, Chem. Soc. Rev., 2017, 46, 2437–2458
J. Qin, J. Yan, B. Wang, and Z. Yang, Rhodamine-enaphthalene conjugate as a novel ratiometric fluorescent probe for recognition of Al3+, Tetrahedron Lett., 2016, 57, 1935–1939
F. Hu, B. Zheng, D. Wang, M. Liu, J. Du, and D. Xiao, A novel dual-switch fluorescent probe for Cr(III) ion based on PET-FRET processes, Analyst, 2014, 139, 3607–3613
M. Annadhasan, and N. Rajendiran, Highly selective and sensitive colorimetric detection of Hg(II) ions using green synthesized silver nanoparticles, RSC Adv., 2015, 5, 94513–94518
M. Li, H. Zhou, L. Shi, D. Li, and Y. Long, Ion-selective goldethiol film on integrated screen printed electrodes for analysis of Cu(II) ions, Analyst, 2014, 139, 643–648
L. Chang, Q. Gao, S. Liu, D. Luo, B. Han, K. Xia, and C. Zhou, A single polymer chemosensor for differential determination of Hg2+ and Cu2+ in pure aqueous media without mutual interference, Mater. Today Commun., 2019, 19, 148–156
L. Bai, F. Tao, L. Li, A. Deng, C. Yan, G. Li, and L. Wang, A simple turn-on fluorescent chemosensor based on Schiff base-terminated water-soluble polymer for selective detection of Al3+ in 100% aqueous solution, Spectrochim. Acta, Part A, 2019, 214, 436–444
L. Bai, G. Li, L. Li, M. Gao, H. Li, F. Tao, A. Deng, S. Wang, and L. Wang, Schiff base functionalized PEG as a high efficient fluorescent chemosensor for Al3+ detection in 100% aqueous solution, React. Funct. Polym., 2019, 139, 1–8
T. Geng, C. Guo, Y. Dong, M. Chen, and Y. Wang, Turn-on fluorogenic and chromogenic detection of cations in complete water media with poly (N-vinyl pyrrolidone) bearing rhodamine B derivatives as polymeric chemosensor, Polym. Adv. Technol., 2016, 27, 90–97
G. Li, L. Bai, F. Tao, A. Deng, and L. Wang, A dual chemosensor for Cu2+ and Hg2+based on a rhodamine-terminated water-soluble polymer in 100% aqueous solution, Analyst, 2018, 143, 5395–5403
Q. M. Wang, W. Gao, X. H. Tang, Y. Liu, and Y. Li, A fluorescence turn-on sensor for aluminum ion by a naphthaldehyde derivative, J. Mol. Struct., 2016, 1109, 127–130
W. Y. He, R. Q. Liu, Y. H. Liao, G. H. Ding, J. L. Li, W. Liu, L. Y. Wu, H. J. Feng, Z. F. Shi, and M. X. He, A new 1,2,3-triazole and its rhodamine B derivatives as a fluorescence probe for mercury ions, Anal. Biochem., 2020, 598, 113690–113702
Z. L. Yang, S. Chen, Y. X. Zhao, P. T. Zhou, and Z. H. Cheng, Hg2+ chromogenic and fluorescence indicators based on rhodamine derivatives bearing thiophene group, Sens. Actuators, B, 2018, 266, 422–428
O. Mecit, A fast-response, highly selective, chromogenic and fluorescent chemosensor for the detection of Hg2+ ions, Sens. Actuators, B, 2017, 249, 217–228
S. Saha, P. Mahato, U. Reddy G, E. Suresh, A. Chakrabarty, M. Baidya, S. K. Ghosh, and A. Das, Recognition of Hg2+ and Cr3+ in Physiological Conditions by a Rhodamine Derivative and Its Application as a Reagent for Cell Imaging Studies, Inorg. Chem., 2012, 51, 336–345
F. Abebe, T. Sutton, P. Perkins, and R. Shaw, Two colorimetric fluorescent turn–on chemosensors for detection of Al3+and N3−: Synthesis, photophysical and computational studies, Luminescence, 2018, 33, 1194–1201
W. Y. Lin, L. Yuan, X. W. Cao, W. Tan, and Y. M. Feng, A Coumarin-Based Chromogenic Sensor for Transition-Metal Ions Showing Ion-Dependent Bathochromic Shift, Eur. J. Org. Chem., 2008 4981–4987
C. Chen, R. Wang, L. Guo, N. Fu, H. Dong, and Y. Yuan, A squaraine-based colorimetric and “Turn on” fluorescent sensor for selective detection of Hg2+ in an Aqueous Medium, Org. Lett., 2011, 13, 1162–1165
Z. Guo, S. Park, J. Yoon, and I. Shin, Recent progress in the development of near-infrared fluorescent probes for bioimaging applications, Chem. Soc. Rev., 2014, 43, 16–29
K. Li, Y. Xiang, X. Y. Wang, J. Li, R. R. Hu, A. J. Tong, and B. Z. Tang, Reversible photochromic system based on rhodamine B salicylaldehyde hydrazone metal complex, J. Am. Chem. Soc., 2014, 136, 1643–1649
K. H. Chu, Y. Zhou, Y. Fang, L. H. Wang, J. Y. Li, and C. Yao, Rhodamine-pyrene conjugated chemosensors for ratiometric detection of Hg2+ ions: different sensing behavior between a spirolactone and a spirothiolactone, Dyes Pigm., 2013, 98, 339–346
D. Q. Sun, T. Lu, F. P. Xiao, X. Y. Zhu, and G. Q. Sun, Formulation and aging resistance of modified bio-asphalt containing high percentage of waste cooking oil residues, J. Cleaner Prod., 2017, 161, 1203–1214
Z. L. Yang, S. Chen, Y. X. Zhao, P. T. Zhou, and Z. H. Cheng, Hg2+ chromogenic and fluorescence indicators based on rhodamine derivatives bearing thiophene group, Sens. Actuators, B, 2018, 266, 422–428
Analytical Methods Committee, Recommendations for the Definition, Estimation and Use of the Detection Limit, Analyst, 1987, 112, 199–204
Author information
Authors and Affiliations
Additional information
† Electronic supplementary information (ESI) available. See DOI: 10.1039/d0pp00302f
Rights and permissions
About this article
Cite this article
Hu, JP., Yang, HH., Lin, Q. et al. A rhodamine-based dual chemosensor for the naked-eye detection of Hg2+ and enhancement of the fluorescence emission for Fe3+. Photochem Photobiol Sci 19, 1690–1696 (2020). https://doi.org/10.1039/d0pp00302f
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/d0pp00302f