Abstract
Preoperative chemoradiotherapy (pCRT) followed by surgery is currently the standard therapy for patients with locally advanced rectal cancer. It is very important to develop biomarkers to prior identify the patients who have a higher likelihood of responding to pCRT. Recently, a series of studies have been conducted to investigate the association of thymidylate synthase (TYMS) polymorphisms with the tumor response to pCRT in rectal cancer, but the results were not consistent and conclusive. In the present study, we performed a systematic literature search for relevant studies up to 30 March 2015 and conducted a meta-analysis to summarize and clarify the association between the TYMS polymorphisms and the tumor response to pCRT in rectal cancer. Finally, 7 studies containing 892 cases for TYMS 2R/3R polymorphism, 7 studies involving 715 cases for TYMS 1494del6 polymorphism and 6 studies containing 616 cases for TYMS 5′ untranslated region (UTR) expression allele polymorphism were analyzed in the meta-analysis. The results suggested that TYMS 2R/3R was associated with the response and the patients with 2R/2R or 2R/3R genotype with rectal cancer might benefit more from pCRT than others. On the contrary, neither 1494del6 nor 5′UTR expression allele polymorphisms was associated with the response to pCRT.
Similar content being viewed by others
Introduction
Colorectal cancer (CRC) is the third most common cancer in the world and the third most common cause of cancer-related death, ∼20% of which is distal to the rectosigmoid junction and is designated as rectal cancer.1 Preoperative 5-fluorouracil (5-FU)-based chemoradiotherapy (pCRT) followed by surgery is currently the standard therapy for patients with locally advanced rectal cancer (clinical TNM (tumor, node, metastasis) stages II–III).2 Other drugs available in colon cancer therapy such as capecitabine, oxaliplatin, irinotecan and cetuximab have also been evaluated in pCRT in rectal cancer.3, 4, 5, 6 Of these drugs, capecitabine is an oral fluoropyrimidine that delivers 5-FU to the tumor and is increasingly used in pCRT in rectal cancer because of its convenience for administration and more favorable toxicity profile than 5-FU.7 Recent clinical trials also suggested that 5-year overall survival is comparable between regiments 5-FU and capecitabine in pCRT setting in rectal cancer.8, 9 After pCRT and surgery treatment, the 5-year rates of local and distant recurrence range from 6 to 9% and from 33 to 36%, revealing an encouraging outcome.10, 11, 12 However, the treatment also exerts some severe side effects such as chemotherapy- and radiotherapy-related toxicity and bowel and sexual dysfunction.13 In addition, some of the patients who received pCRT show poor or no response.14, 15 Hence, it is important to prior identify the patients who have a higher likelihood of responding to pCRT. Several biomarkers have been investigated to see whether they can predict the tumor response to pCRT including genetic polymorphisms in thymidylate synthase (TYMS) gene.
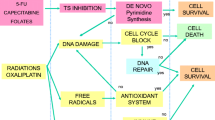
The thymidylate synthase provides thymidylate for DNA synthesis that leads to cell proliferation and is the most important target of 5-FU. When 5-FU is converted into its active metabolite, it forms a stable complex with TYMS and then the activity of the enzyme is inhibited, resulting in cell cycle arrest and cell death.16 There are three main genetic polymorphisms in TYMS that have been described. The first one is located in the 5′-untranslated region (5′UTR) (rs34743033) that consists of a double (2R) or triple (3R) repeat of a 28-base-pair (bp) tandem repeat. The transcription efficiency of TYMS with 2R is lower than that with 3R.17 Another polymorphism is a functional G>C single-nucleotide polymorphism located in the second repeat of the 3R alleles (rs2853542).18 The 3R(G) allele is correlated with greater transcription activity and the transcription efficiency of the 3R(C) alleles is similar to that of the 2R alleles. After combination of the two polymorphisms, the genotypes can be divided into two groups: TYMS high-expression allele (2R/3RG, 3R/3RG and 3RG/3RG) and low-expression allele (2R/2R, 2R/3RC and 3RC/3RC). The third one is a 6 bp insertion at nucleotide 1494 in the 3′UTR that has been reported to be associated with higher intratumoral TYMS expression and may decrease the chemosensitivity to 5-FU.19, 20
Although the association of TYMS polymorphisms with the tumor response to pCRT in rectal cancer has been in a series of studies in recent 10 years, the results were inconsistent and inconclusive. Herein, we performed a systematic review and meta-analysis for the first time to explore the association of three TYMS polymorphisms with the tumor response to pCRT in rectal cancer.
Materials and methods
Publication search
A systematic search was performed for published articles on the relationship between TYMS polymorphisms and response to pCRT in rectal cancer. Two Chinese databases (Chinese National Knowledge Infrastructure and Wanfang databases) and three English databases (PubMed, EMBASE and Web of science) were utilized to search the available articles with the last search update on 30 March 2015. The following keywords were used: ‘thymidylate synthase OR TYMS’, ‘Rectal cancer OR rectal carcinoma’, ‘polymorphism OR polymorphisms’, and ‘Chemoradiation OR chemoradiotherapy’. Two independent authors screened and selected the retrieved articles according to the inclusion and exclusion criteria. The review articles and the references of selected articles were also screened to identify additional eligible studies.
Inclusion and exclusion criteria
Inclusion criteria were as follows: (1) evaluating the relationship between TYMS 2R/3R, expression allele and 1494del6 polymorphisms with response to pCRT; (2) the response was evaluated by tumor regression grade; (3) genotype frequency data could be obtained. Exclusion criteria were as follows: (1) the data of tumor regression grade (TRG) were not specific to polymorphism; (2) study with insufficient or duplicate data; and (3) abstracts, letters or review articles.
Data extraction
Data were carefully collected in duplicate by two independent investigators. The following information was extracted: name of first author, year of publication, the clinical characteristics of cases, country of origin, ethnicity, disease stage, chemotherapy drugs, radiation dose, sample source, response evaluation method (TRG 1–2 vs 3–5 or 1 vs 2–5) and genotype frequency of TYMS 2R/3R, 1494del6 and expression allele in different TRG grades, respectively. Inconsistency was solved by discussion.
Quality score assessment
The methodological quality of every eligible article in the present meta-analysis was assessed by two investigators independently through the Newcastle–Ottawa Scale based on three aspects, selection, comparability and exposure, with scores ranging from 0 to 9. The Newcastle–Ottawa Scale score of ⩾7 was considered as high quality.
Statistical analysis
In the study, TRG grades were defined as follows: grade 1: the absence of residual cancer; grade 2: the presence of rare residual cancer cells; grade 3: an increase in the number of residual cancer cells but with fibrosis predominating; grade 4: residual cancer outgrowing fibrosis; and grade 5: the absence of regressive changes.21, 22 Patients were subdivided into responders and nonresponders (TRG 1–2 vs 3–5 or 1 vs 2–5). When the response in one study was evaluated by both TRG 1–2 vs 3–5 and 1 vs 2–5, the TRG 1–2 vs 3–5 data and TRG 1 vs 2–5 data were treated as two studies. The crude odds ratios (ORs) and 95% confidence intervals (95% CIs) were calculated to assess the strength of association between TYMS 2R/3R, expression allele and 1494del6 polymorphisms and response to pCRT in rectal cancer. A P-value of <0.05 was considered as statistically significance. ORs were pooled for allelic comparison, codominant model, dominant model and recessive model, respectively. The statistically significant level was determined by Z-test with P-value <0.05. The heterogeneity was assessed by χ2 based on Q-statistic test with a P-value of <0.1 considered significant. If P>0.1, the pooled OR and 95% CIs were calculated by the fixed effects model (Mantel–Haenszel method), otherwise the random effects model (DerSimonian–Laird method) was used.23 Sensitivity analysis was also conducted to evaluate the effect of each study on the combined ORs by omitting each study in every turn. Besides, subgroup analyses according to evaluation definition or ethnicity were also performed. Potential publication bias was checked by Begg’s funnel plots and Egger’s test.24, 25 Stata 12.0 software (StataCorp, College Station, TX, USA) was used to perform all analyses.
Results
Study characteristics
The literature selection process is shown in Figure 1. A total of 86 documents were initially identified. After excluding overlapped records, reviews, meeting abstracts and irrelevant papers, 18 records were further evaluated. Subsequently, seven papers were excluded according to the inclusion and exclusion criteria, of which six papers did not report TRG data and one reported TYMS expression allele-directed therapy in rectal cancer. Finally, 11 eligible articles were included in the meta-analysis.26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36 Quality assessment was performed and all the studies arrived at a high Newcastle–Ottawa Scale NOS score (⩾7, data not show). Of these studies, Hur et al.31 investigated the association in the Asian population and other studies were performed in the Caucasian population. In two papers, the response was evaluated by both TRG 1–2 vs 3–5 and TRG 1 vs 2–5.31, 34 The characteristics of each included study are listed in Table 1. These studies were published from 2006 to 2015. Seven included studies containing 892 cases for TYMS polymorphism 2R/3R,26, 27, 31, 32, 33, 34, 36 seven studies involving 715 cases for 1494del6,28, 29, 30, 33, 34, 35 and six studies containing 616 cases for TYMS 5′UTR expression allele26, 28, 29, 31, 32, 35, 36were finally analyzed in the meta-analysis.
Flowchart of study selection. 2R/3R, double/triple repeat; CNKI, Chinese National Knowledge Infrastructure; TRG, tumor regression grade; TYMS, thymidylate synthase.
Meta-analysis results
The association between TYMS 2R/3R polymorphism and the response to pCRT was first analyzed. Overall, significant association was identified in allele model (2R vs 3R, OR=1.54, 95% CI=1.20–1.98, P=0.001) and the codominant model (2R2R vs 3R3R, OR=3.22, 95% CI=1.03–10.53, P=0.044; 2R3R vs 3R3R, OR=1.98, 95% CI=1.31–3.00, P=0.001; Table 1 and Figure 2). Next, subgroup analysis was conducted according to different evaluation methods. In the group of TRG 1–2 vs 3–5, the association was only found in the allele model (2R vs 3R) and the codominant model (2R3R vs 3R3R; Table 1 and Figure 2). In the group of TRG 1 vs 2–5, the association was identified in the allele model (2R vs 3R), codominant model (2R3R vs 3R3R) and dominant model (2R2R or 2R3R vs 3R3R; Table 1 and Figure 2). Furthermore, the effect of ethnicity was also determined in the two evaluation groups. The results revealed a significant association in Caucasians in the codominant model (2R3R vs 3R3R) in TRG 1–2 vs 3–5 group, and in the codominant model (2R3R vs 3R3R) and the dominant model (2R2R or 2R3R vs 3R3R) in TRG 1 vs 2–5 group (Table 2 and Figure 3). We next investigated the association between TYMS 1494del6 polymorphism and the response to pCRT in rectal cancer. However, no significant association was identified in all genetic models in overall analysis or subgroup analysis according to the evaluation methods (Figure 4 and Table 2). As all the studies were performed in Caucasians, the ethnicity effects could not be examined Finally, the association between TYMS 5′UTR expression allele polymorphism and the response to chemoradiotherapy in rectal cancer was analyzed. No significant association was identified in all genetic models in overall analysis or subgroup analysis (Figure 5 and Table 2).
Forest plot for TYMS 2R/3R polymorphism and the response to pCRT in rectal cancer. Five genetic comparison models were used, 2 vs. 3 (a), 22 vs. 33 (b), 23 vs. 33 (c), 22/23 vs. 33 (d), and 22 vs. 23/33 (e). 2R/3R, double/triple repeat; CI, confidence interval; OR, odds ratio; pCRT, preoperative chemoradiotherapy; TRG, tumor regression grade; TYMS, thymidylate synthase.
Forest plot for TYMS 2R/3R polymorphism and the response to pCRT in rectal cancer subgroup analyzed according to ethnicity. (a) TRG 1-2 was defined as response. (b) TRG 1 was defined as response. 2R/3R, double/triple repeat; CI, confidence interval; OR, odds ratio; pCRT, preoperative chemoradiotherapy; TRG, tumor regression grade; TYMS, thymidylate synthase.
Forest plot for TYMS 1494del6 polymorphism and the response to pCRT in rectal cancer. Five genetic comparison models were used, 0 vs. 6 (a), 00 vs. 66 (b), 06 vs. 66 (c), 00/06 vs. 66 (d), and 00 vs. 06/66 (e). CI, confidence interval; OR, odds ratio; pCRT, preoperative chemoradiotherapy; TRG, tumor regression grade; TYMS, thymidylate synthase.
Forest plot for TYMS expression allele polymorphism and the response to pCRT in rectal cancer. CI, confidence interval; OR, odds ratio; pCRT, preoperative chemoradiotherapy; TRG, tumor regression grade; TYMS, thymidylate synthase.
Sensitivity analysis and publication bias
Sensitivity analysis was performed to examine the influence set by the individual study on the pooled ORs by deleting each study once in every genetic model. For all three TYMS polymorphisms, we arrived at almost the same results (data not shown). Begg’s funnel plot and Egger’s test were carried out to assess the publication bias among the selected studies. Symmetrical funnel plots were obtained in all the genetic models (Figure 6, and data not shown). Egger’s test was performed to provide the statistical evidence of publication bias and the results did not show any publication bias for all three TYMS polymorphisms (Table 2).
Begg’s funnel plot for publication bias analysis for for TYMS polymorphisms 2R/3R (a), 1494del6 (b), and expression allele (c). 2R/3R, double/triple repeat; TYMS, thymidylate synthase.
Discussion
In the present study, we performed a meta-analysis to explore the association between TYMS polymorphisms and the response to pCRT in rectal cancer and found that TYMS 2R/3R might correlate with the response, whereas 1494del6 and 5′UTR expression allele polymorphisms exhibited no significant association.
pCRT followed by surgery is currently the standard therapy for locally advanced rectal cancer.2 However, the therapy strategy also brings severe side effects and some of the patients undergoing pCRT show poor or no response.14, 15 Thus, it is important to develop some biomarkers to screen and identify the patients who may benefit from pCRT before treatment. Of the biomarkers, TYMS gene is a widely explored one. It is critical for cell proliferation and the most important target of 5-FU.16 In recent 10 years, a series of studies have been conducted to investigate the association of TYMS polymorphisms with the tumor response to pCRT in rectal cancer, but the results were not consistent and inconclusive. Here, we pooled all related studies and identified a significant association between TYMS 2R/3R polymorphism and the response to pCRT in overall and subgroup analysis. The patients carrying 2R/2R or 2R/3R genotypes might benefit more from pCRT therapy. On the contrary, no significant association was found between TYMS expression allele and 1494del6 polymorphisms and response to pCRT in rectal cancer. However, the conclusions should be treated carefully. First, the responses in some studies were grouped both by TRG 1–2 vs 3–5 and TRG 1 vs 2–5 and treated as two approximate studies, and thus was not strict. Second, the included study number and sample size were limited. Third, almost all the original studies were performed in the Caucasian population except one that was carried out in the Asian population. Other important shortages were the differences among the analyzed studies such as the diversity of drug dose and kind, radiation dose, sample source and interval time between pCRT and surgery. In detail, for the dose and kind of drugs, 550 (studies n=7) and 126 (n=4) patients received 5-FU or capecitabine alone, respectively. In addition 186 (n=4) and 160 (n=2) patients received 5-FU or capecitabine combined with other drugs, respectively. Oxaliplatin, cetuximab and leucovorin were mostly used in the combined therapy. Only one study did not use 5-FU or capecitabine for the patients. The dose of capecitabine was almost 825 mg m−2 per 2 days, whereas 5-FU varied from 200 to 400 mg m−2 per day. Unfortunately, the data obtained from the analyzed studies were not sufficient to compare the predictive value of TYMS polymorphisms to the response to pCRT among different drug administration strategies. As for the dose of radiation, it ranged from 41 to 60 Gy (45.0 Gy, studies n=4; 50.4/50.5 Gy, n=3; 45–50.4 Gy, n=2; 41–60 Gy, n=1; and 60 Gy, n=1) respectively. Higher radiation dose might result in a higher rate of tumor regression. After stratification by radiation dose, TYMS polymorphisms 2R/3R and expression allele seem to be associated with the response to pCRT more significantly in a higher dose subgroup (Supplementary Table S1). Another factor affecting the rate of tumor regression was the interval time between pCRT and surgery because that a longer time might result in a higher rate too. The interval time in seven studies was 6–8 weeks and other four studies did not provide such data. In these analyzed studies, the rate of tumor regression ranged from 0.36 to 0.82 (TRG 1–2 as responders) and 0.14 to 0.3 (TRG 1 as responder) that might have resulted from different patient ethnicities and therapy schedules. The variation of tumor regression rate might ultimately bring negative effects on analyzing the predictive value of TYMS polymorphisms to the response of pCRT in rectal cancer. In addition, the samples used for polymorphism genotyping were obtained from different sources. Eight studies got genomic DNA from blood and three studies from tumor tissues. All these differences might bring unreliability to the conclusions of the present meta-analysis. Thus, further well-designed studies with larger sample size using homogeneous pCRT regimens should be conducted to confirm the results and avoid potential biases.
Conclusion
In summary, we got a comprehensive result from the current meta-analysis that TYMS 2R/3R polymorphism was correlated with the response to pCRT in rectal cancer, whereas neither 1494del6 nor 5′UTR expression allele polymorphism was associated with the response.
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T et al. Cancer statistics, 2008. CA Cancer J Clin 2008; 58: 71–96.
Spolverato G, Pucciarelli S, Bertorelle R, De Rossi A, Nitti D . Predictive factors of the response of rectal cancer to neoadjuvant radiochemotherapy. Cancers (Basel) 2011; 3: 2176–2194.
Dunst J, Reese T, Sutter T, Zuhlke H, Hinke A, Kolling-Schlebusch K et al. Phase I trial evaluating the concurrent combination of radiotherapy and capecitabine in rectal cancer. J Clin Oncol 2002; 20: 3983–3991.
Gerard JP, Azria D, Gourgou-Bourgade S, Martel-Laffay I, Hennequin C, Etienne PL et al. Comparison of two neoadjuvant chemoradiotherapy regimens for locally advanced rectal cancer: results of the phase III trial ACCORD 12/0405-Prodige 2. J Clin Oncol 2010; 28: 1638–1644.
Mohiuddin M, Winter K, Mitchell E, Hanna N, Yuen A, Nichols C et al. Randomized phase II study of neoadjuvant combined-modality chemoradiation for distal rectal cancer: Radiation Therapy Oncology Group Trial 0012. J Clin Oncol 2006; 24: 650–655.
Bertolini F, Chiara S, Bengala C, Antognoni P, Dealis C, Zironi S et al. Neoadjuvant treatment with single-agent cetuximab followed by 5-FU, cetuximab, and pelvic radiotherapy: a phase II study in locally advanced rectal cancer. Int J Radiat Oncol Biol Phys 2009; 73: 466–472.
Hirsch BR, Zafar SY . Capecitabine in the management of colorectal cancer. Cancer Manag Res 2011; 3: 79–89.
Allegra CJ, Yothers G, O'Connell MJ, Beart RW, Wozniak TF, Pitot HC et al. Neoadjuvant 5-FU or capecitabine plus radiation with or without oxaliplatin in rectal cancer patients: a phase III randomized clinical trial. J Natl Cancer Inst 2015; 107.
Hofheinz RD, Wenz F, Post S, Matzdorff A, Laechelt S, Hartmann JT et al. Chemoradiotherapy with capecitabine versus fluorouracil for locally advanced rectal cancer: a randomised, multicentre, non-inferiority, phase 3 trial. Lancet Oncol 2012; 13: 579–588.
Bosset JF . Distal rectal cancer: sphincter-sparing is also a challenge for the radiation oncologist. Radiother Oncol 2006; 80: 1–3.
Sauer R, Becker H, Hohenberger W, Rodel C, Wittekind C, Fietkau R et al. Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med 2004; 351: 1731–1740.
Gerard JP, Conroy T, Bonnetain F, Bouche O, Chapet O, Closon-Dejardin MT et al. Preoperative radiotherapy with or without concurrent fluorouracil and leucovorin in T3-4 rectal cancers: results of FFCD 9203. J Clin Oncol 2006; 24: 4620–4625.
Pucciarelli S, Del Bianco P, Efficace F, Serpentini S, Capirci C, De Paoli A et al. Patient-reported outcomes after neoadjuvant chemoradiotherapy for rectal cancer: a multicenter prospective observational study. Ann Surg 2011; 253: 71–77.
Minsky BD, Cohen AM, Kemeny N, Enker WE, Kelsen DP, Reichman B et al. Combined modality therapy of rectal cancer: decreased acute toxicity with the preoperative approach. J Clin Oncol 1992; 10: 1218–1224.
Mohiuddin M, Hayne M, Regine WF, Hanna N, Hagihara PF, McGrath P et al. Prognostic significance of postchemoradiation stage following preoperative chemotherapy and radiation for advanced/recurrent rectal cancers. Int J Radiat Oncol Biol Phys 2000; 48: 1075–1080.
Longley DB, Harkin DP, Johnston PG . 5-fluorouracil: mechanisms of action and clinical strategies. Nat Rev Cancer 2003; 3: 330–338.
Horie N, Aiba H, Oguro K, Hojo H, Takeishi K . Functional analysis and DNA polymorphism of the tandemly repeated sequences in the 5'-terminal regulatory region of the human gene for thymidylate synthase. Cell Struct Funct 1995; 20: 191–197.
Mandola MV, Stoehlmacher J, Muller-Weeks S, Cesarone G, Yu MC, Lenz HJ et al. A novel single nucleotide polymorphism within the 5' tandem repeat polymorphism of the thymidylate synthase gene abolishes USF-1 binding and alters transcriptional activity. Cancer Res 2003; 63: 2898–2904.
Ulrich CM, Bigler J, Velicer CM, Greene EA, Farin FM, Potter JD . Searching expressed sequence tag databases: discovery and confirmation of a common polymorphism in the thymidylate synthase gene. Cancer Epidemiol Biomarkers Prev 2000; 9: 1381–1385.
Park DJ, Stoehlmacher J, Zhang W, Tsao-Wei D, Groshen S, Lenz HJ . Thymidylate synthase gene polymorphism predicts response to capecitabine in advanced colorectal cancer. Int J Colorectal Dis 2002; 17: 46–49.
Mandard AM, Dalibard F, Mandard JC, Marnay J, Henry-Amar M, Petiot JF et al. Pathologic assessment of tumor regression after preoperative chemoradiotherapy of esophageal carcinoma. Clinicopathologic correlations. Cancer 1994; 73: 2680–2686.
Dworak O, Keilholz L, Hoffmann A . Pathological features of rectal cancer after preoperative radiochemotherapy. Int J Colorectal Dis 1997; 12: 19–23.
DerSimonian R, Laird N . Meta-analysis in clinical trials. Control Clin Trials 1986; 7: 177–188.
Begg CB, Mazumdar M . Operating characteristics of a rank correlation test for publication bias. Biometrics 1994; 50: 1088–1101.
Egger M, Davey Smith G, Schneider M, Minder C . Bias in meta-analysis detected by a simple, graphical test. BMJ 1997; 315: 629–634.
Terrazzino S, Agostini M, Pucciarelli S, Pasetto LM, Friso ML, Ambrosi A et al. A haplotype of the methylenetetrahydrofolate reductase gene predicts poor tumor response in rectal cancer patients receiving preoperative chemoradiation. Pharmacogenet Genomics 2006; 16: 817–824.
Spindler KL, Nielsen JN, Lindebjerg J, Jakobsen A . Germline polymorphisms may act as predictors of response to preoperative chemoradiation in locally advanced T3 rectal tumors. Dis Colon Rectum 2007; 50: 1363–1369.
Stoehlmacher J, Goekkurt E, Mogck U, Aust DE, Kramer M, Baretton GB et al. Thymidylate synthase genotypes and tumour regression in stage II/III rectal cancer patients after neoadjuvant fluorouracil-based chemoradiation. Cancer Lett 2008; 272: 221–225.
Balboa E, Duran G, Lamas MJ, Gomez-Caamano A, Celeiro-Munoz C, Lopez R et al. Pharmacogenetic analysis in neoadjuvant chemoradiation for rectal cancer: high incidence of somatic mutations and their relation with response. Pharmacogenomics 2010; 11: 747–761.
Paez D, Pare L, Altes A, Josep Sancho-Poch F, Petriz L, Garriga J et al. Thymidylate synthase germline polymorphisms in rectal cancer patients treated with neoadjuvant chemoradiotherapy based on 5-fluorouracil. J Cancer Res Clin Oncol 2010; 136: 1681–1689.
Hur H, Kang J, Kim NK, Min BS, Lee KY, Shin SJ et al. Thymidylate synthase gene polymorphism affects the response to preoperative 5-fluorouracil chemoradiation therapy in patients with rectal cancer. Int J Radiat Oncol Biol Phys 2011; 81: 669–676.
Paez D, Salazar J, Pare L, Pertriz L, Targarona E, del Rio E et al. Pharmacogenetic study in rectal cancer patients treated with preoperative chemoradiotherapy: polymorphisms in thymidylate synthase, epidermal growth factor receptor, GSTP1, and DNA repair genes. Int J Radiat Oncol Biol Phys 2011; 81: 1319–1327.
Cecchin E, Agostini M, Pucciarelli S, De Paoli A, Canzonieri V, Sigon R et al. Tumor response is predicted by patient genetic profile in rectal cancer patients treated with neo-adjuvant chemo-radiotherapy. Pharmacogenomics J 2011; 11: 214–226.
Hu-Lieskovan S, Vallbohmer D, Zhang W, Yang D, Pohl A, Labonte MJ et al. EGF61 polymorphism predicts complete pathologic response to cetuximab-based chemoradiation independent of KRAS status in locally advanced rectal cancer patients. Clin Cancer Res 2011; 17: 5161–5169.
Lamas MJ, Duran G, Gomez A, Balboa E, Anido U, Bernardez B et al. X-ray cross-complementing group 1 and thymidylate synthase polymorphisms might predict response to chemoradiotherapy in rectal cancer patients. Int J Radiat Oncol Biol Phys 2012; 82: 138–144.
Sebio A, Salazar J, Paez D, Berenguer-Llergo A, Del Rio E, Tobena M et al. EGFR ligands and DNA repair genes: genomic predictors of complete response after capecitabine-based chemoradiotherapy in locally advanced rectal cancer. Pharmacogenomics J 2015; 15: 77–83.
Acknowledgements
This study was supported by the Beijing Municipal Administration of Hospitals Clinical Medicine Development of Special Funding Support (ZYLX201504) and the Capital Foundation of Medical Developments (Z20150405090022).
Author contributions
Designed the study: JW and ZTZ. Search databases and collected full-text papers: YCY, GCW, and LJ. Extracted and analyzed the data: KLW, ZGB and YCY. Statistical analyses: GCW and LJ. Wrote the main manuscript text: JW, ZTZ and YCY.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the The Pharmacogenomics Journal website
Supplementary information
Rights and permissions
About this article
Cite this article
Yang, Y., Wu, G., Jin, L. et al. Association of thymidylate synthase polymorphisms with the tumor response to preoperative chemoradiotherapy in rectal cancer: a systematic review and meta-analysis. Pharmacogenomics J 17, 265–273 (2017). https://doi.org/10.1038/tpj.2016.11
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/tpj.2016.11
- Springer Nature Limited