Abstract
A meta-analysis was conducted to evaluate the association between non-occupational physical activity (PA) during pregnancy and the risk of preterm birth (PTB). By searching PubMed and EMBASE from inception to August 20, 2016, 25 observational studies (18 cohorts and 7 case-controls) and 12 interventional studies were identified. Comparing the highest to the lowest category of leisure-time PA during pregnancy, the pooled relative risk (RR) of PTB was 0.83 [95% confidence interval (CI) = 0.74–0.93] for cohort studies and 0.60 (95% CI = 0.43–0.84) for case-control studies. No overall significant association was found between domestic or commuting PA and the risk of PTB. In addition, PA intervention did not indicate significant beneficial effect on the risk of PTB. Evidence from the observational studies suggested that leisure-time, but not domestic or commuting, PA during pregnancy was inversely associated with the risk of PTB. The findings were not supported by small-scale and short-term interventional studies. Further research with objective measurement on leisure-time PA is warranted.
Similar content being viewed by others
Introduction
Preterm birth (PTB) is the second most common cause of death after pneumonia1 among children under 5 years old, accounting for approximately 35% of annual neonatal deaths worldwide1,2. Active participation in physical activity (PA) during pregnancy is not only beneficial in maintaining pregnant women’s general health condition3,4, but also reduces their risk of developing chronic diseases such as gestational diabetes mellitus and preeclampsia5,6. However, the relation between PA during pregnancy and the risk of PTB still remains unclear.
Many previous studies focused on total PA. For example, two studies7,8 evaluated PA as a single score of energy expenditure including occupational, leisure-time, and domestic PA and found it was not significantly associated with the risk of PTB. It may not be appropriate to combine all domains of PA into a single score and link it to health endpoints including PTB because different domains of PA may have different health impacts. For instance, one study9 found that leisure-time PA was inversely associated with the risk of PTB, but commuting PA appeared to be positively related to the risk.
In addition, systematic reviews have suggested that occupational PA such as trunk bend (>1 hour/day), prolonged standing (>4 hours/day), shifting work and heavy physical work, particularly in late pregnancy, may increase the risk of PTB10,11,12,13,14. However, the associations of non-occupational PA, including leisure-time, domestic, and commuting PA, with the risk of PTB have not been systematically evaluated.
Therefore, we aimed to quantitatively examine the overall association between maternal non-occupational PA during pregnancy and the risk of PTB.
Methods
Search strategy
This meta-analysis was performed based on the criteria of Preferred Reporting Items for Systematic reviews and Meta-analyses predefined protocol (PRISMA)15. Literature review was systematically conducted in PubMed and EMBASE through August 20, 2016. We used a set of terms related to the exposure (“recreation”, “exercise”, “activity”, “commuting”, “transportation”, “domestic”, “housework”, “household”, or “caregiving”) and another set of terms related to the outcome (“preterm birth”, “preterm delivery”, “preterm labo(u)r”, “early labo(u)r”, “premature birth” or “prematurity”). In addition, some other terms including “job”, “occupational”, and “work” were used because the associations of interest may be reported as secondary results in those studies focusing on the associations between occupational PA and birth outcomes. Google Scholar and the reference lists of the relevant narrative and systematic reviews were searched for additional citations.
Eligibility criteria
Studies were considered to be eligible for the meta-analysis of observational studies if they were a cohort (prospective or retrospective) or case-control study, and reported relative risk (RR), hazards ratio (HR) or odds ratio (OR) with the corresponding 95% confidence intervals (CIs) of the risk of PTB in relation to any non-occupational PA during pregnancy, or such information could be derived from the presented results. Non-occupational PA included three domains: the leisure time (recreational and sport activities) domain, the domestic (house and gardening work) domain and the commuting (active transportation) domain. For multiple publications with identical exposure and identical outcome using data from the same study, we selected the one with the larger sample size. Studies were considered to be eligible for the meta-analysis of interventional studies if they reported the preterm birth for both the physical activity intervention group and the control group in apparently healthy pregnant women. Two reviewers (J. W. and P. X.) independently reviewed all the relevant articles. Disagreements were resolved by consensus and discussion with a third reviewer (K. H.).
Quality assessment
All identified observational studies received quality assessment based on the Newcastle-Ottawa quality assessment scale16, which evaluates observational studies from three aspects: the selection of study population (4 criteria with 4 stars), the comparability of study population (1 criterion with 2 stars), and the assessment of exposure (3 criteria with 3 stars) for a cohort study or the ascertainment of outcome (3 criteria with 3 stars) for a case-control study. Each star was assigned 1 point with a total of 9 points. The quality of study was considered high if the sum score was ≥8 points, and moderate if the sum score ranged from 5 to 7 points.
Data extraction
Two co-authors (J. W. and P. X.) independently reviewed each included study and extracted the relevant information. Discrepancies were resolved by group discussion with a third co-author (K. H.). The following information was extracted: last name of the first author, the year when the paper was published, the country where the study was conducted, study design, study period, the number of participants/cases, participant age, exposure and its assessment method, outcome ascertainment, measures of the associations of interest [i.e., RR, HR, or OR with the corresponding 95% CIs], and the covariates adjusted in the final model.
Data synthesis and analysis
Since PTB is a relatively rare disease, we ignored the distinction among the association measures (i.e., RR, HR, and OR) and undertook a random-effects meta-analysis to estimate the pooled relative risk (RR) and 95% CIs comparing the highest to the lowest category of PA level. If a study did not present multivariable-adjusted models, the unadjusted data was used. When no effect estimate was given, a crude estimate was calculated directly from a 2 by 2 table based on available information. If the estimates were reported for different trimesters respectively, they were combined first with a random-effects meta-analysis model.
Heterogeneity among studies was examined by using Cochran’s Q test and quantified by using the I2 statistic. To reduce the likelihood of drawing a false negative conclusion (type II error), a P value of ≤0.10 is considered as statistically significant for Cochran’s Q test. Very low, low, moderate, and high degree of heterogeneity were defined as ≤25%, 26–50%, 51–75% and >75%, respectively. Publication bias was assessed by Egger’s regression asymmetry test. The Duval and Tweedie nonparametric “trim and fill” method was used to estimate the pooled association of interest if publication bias was suggested17. Sensitivity analyses were performed to evaluate the robustness of our findings: 1) to remove one study from the pooled analysis each time; and 2) to replace random-effects with fixed-effects models.
All analyses were performed with STATA software (Version 14, STATA Corporation LP, College Station, TX). A two-sided P value of ≤0.05 was considered statistically significant if not otherwise specified.
Results
Study selection process
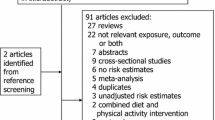
Figure 1 shows the detailed selection process. We retrieved 214 relevant studies from PubMed and EMBASE. Of them, 189 studies were excluded for one of the following reasons: (1) not an original study; (2) not in English; (3) occupational population, such as nurses, military personnel, physicians; (4) no information on the association of interest and such information cannot be derived from available data; (5) no regular measurement in PA; or (6) duplicated publication. In addition, we identified 12 studies through Google Scholar and the relevant reference lists. Therefore, 37 eligible studies (7 case control studies, 18 cohort studies, and 12 interventional studies) were included in the meta-analysis. All the included 25 observational studies (7 case-control studies and 18 cohort studies) were rated as either high18 or moderate9,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41 quality (see Supplemental Tables S1 and S2).
Study characteristics
Tables 1 and 2 summarize the characteristics of the included observational studies. Information on leisure-time PA and the risk of PTB were provided in 13 cohort studies9,20,25,26,27,28,29,30,32,33,34,35,36 (167,087 participants and 9,096 cases), and 4 case-control studies21,37,39,41 (966 cases and 1,685 controls). Of them, 9 studies were conducted in North America9,20,21,26,27,34,35,36,41, 3 in Europe29,30,33, 3 in the Asia-Pacific region25,37,39, and 2 in South America28,32. Data on domestic PA and the risk of PTB were presented by 7 cohort studies9,22,23,24,25,31,34 (11,009 participants and 747 cases), and 3 case-control studies18,21,40 (391 cases and 651 controls). Of these studies, 3 were conducted in North America9,21,34, 2 in Europe22,23, 2 in the Asia-Pacific region24,25, 2 in Africa18,31, and 1 in South America40. Information on commuting PA and the risk of PTB was available in 5 cohort studies9,19,23,24,35 (5,489 participants and 592 cases), and 1 case-control study38 (2,230 cases and 3,907 controls). Of these studies, 3 were conducted in Europe19,23,38, 2 in North America9,35, and 1 in the Asia-Pacific region24.
Table 3 summarizes the characteristics of 12 included interventional studies42,43,44,45,46,47,48,49,50,51,52,53 totaling 1,409 pregnant women with 69 PTBs in the PA intervention group, and 1,402 pregnant women with 60 PTBs in the control group. Of these studies, 2 were conducted in the US42,44, 4 in Sweden46,50,51,53, and the rest were in Australia43, Brazil45,47, Iran52, Norway48, and Spain49. The average age of pregnant women ranged from 24.4 to 31.8 years.
Leisure-time physical activity and the risk of preterm birth
Thirteen cohort and 4 case-control studies have data on leisure-time PA and the risk of PTB. Comparing the highest to the lowest category of leisure-time PA, the pooled RR of PTB was 0.83 (95% CI = 0.74–0.93) for cohort studies and 0.60 (95% CI = 0.43–0.84) for case-control studies (Fig. 2). No significant heterogeneity (I2 = 18.5%, P = 0.26) was observed in cohort studies, but a moderate heterogeneity was found in case-control studies (I2 = 54.3%, P = 0.09). Since publication bias was detected among cohort studies (Egger’s test: P = 0.01), we adjusted for the pooled association using the Duval and Tweedie method and the pooled results became 0.78 (95% CI = 0.61–0.997). No evidence of publication bias was found in case-control studies (Egger’s test: P = 0.63).
The pooled estimates (diamond data markers) were obtained using a random-effects model. The dots indicate the RRs for the risk of PTB comparing the highest with the lowest category of leisure-time PA during pregnancy. The size of the shaded square is proportional to the percentage weight of each study. The horizontal lines represent 95% CIs. CI: confidence interval; RR: relative risk.
Sensitivity analysis indicated that no single study appreciably changed the results, and the pooled associations persisted when a fixed-effects model was used instead of a random-effects one. Notable, the pregnancy period (first, second, third trimester, or mixed) and format (intensity, duration or frequency) of PA assessed were different among these studies. However, the pooled RR was similar [0.83 (95% CI = 0.78–0.88)] when combining data from 11 cohort studies in which leisure-time PA was assessed using frequency (i.e., yes or no, minutes per week, hours per week, times per week and times per month). In addition, the pooled estimate was essentially unchanged [0.80 (0.69–0.94)] when we combined data from 4 cohort studies in which leisure-time PA was measured in the first two trimesters.
Domestic physical activity and the risk of preterm birth
Seven cohort and 3 case-control studies reported results on domestic PA during pregnancy and the risk of PTB. No significant association was revealed. The pooled RR was 0.86 (95% CI = 0.65–1.14) for cohort studies and 0.64 (95% CI = 0.39–1.07) for case-control studies (Fig. 3). Neither significant heterogeneity (I2 = 29.1% and P = 0.21 for cohort; I2 = 17.4% and P = 0.30 for case-control) nor publication bias (Egger’s test: P = 0.56 for cohort and P = 0.74 for case-control) was evident.
The pooled estimates (diamond data markers) were obtained using a random-effects model. The dots indicate the RRs for the risk of PTB comparing the highest with the lowest domestic PA level during pregnancy. The size of the shaded square is proportional to the percentage weight of each study. The horizontal lines represent 95% CIs. CI: confidence interval; RR: relative risk.
The pooled results generally remained when using a fixed-effects model. However, the pooled association became statistically significant [0.78 (95% CI = 0.60–0.997)] after omitting Misra9 et al. among cohort studies. Of note, the domestic PA in that study was defined as lifting heavy objects at home. This inverse association was slightly strengthened when further excluding another study22 that also included lifting objects at home as the domestic PA [0.74 (95% CI = 0.55–0.998)].
Commuting physical activity and the risk of preterm birth
Five cohort studies and 1 case-control study presented data on commuting PA during pregnancy and the risk of PTB. No significant association was found among cohort studies comparing the highest to the lowest level of commuting PA (the pooled RR = 1.08; 95% CI = 0.67–1.75). Also, publication bias was not evident (Egger’s test: P = 0.79). The observed null association was not appreciably altered by any single study and the pooled results persisted when the random-effects model was replaced with a fixed-effects model in the sensitivity analyses.
Physical activity intervention and the risk of preterm birth
Twelve interventional studies presented data on PA intervention and preterm birth, and found no significant association (the pooled RR = 1.15; 95% CI = 0.82–1.61). Neither significant heterogeneity (I2 = 0.0% and P = 0.95) nor publication bias (Egger’s test: P = 0.78) was documented. Sensitivity analysis indicated that no single study appreciably changed the results, and the pooled associations persisted when a fixed-effects model was used. When two studies specifically on sedentary women and one study on overweight women were excluded, the results were materially unchanged (the pooled RR = 1.14; 95% CI = 0.81–1.62) (Fig. 4).
The pooled estimates (diamond data markers) were obtained using a random-effects model. The dots indicate the RRs for the risk of PTB comparing the intervention group to the control group. The size of the shaded square is proportional to the percentage weight of each study. The horizontal lines represent 95% CIs. CI: confidence interval; RR: relative risk.
Discussion
In the meta-analysis of observational studies, we found a significant inverse association of leisure-time PA during pregnancy with the risk of PTB. Domestic PA was inversely associated with the risk of PTB only if studies defining domestic PA as lifting heavy objects at home were excluded. No significant association was observed between commuting PA and the risk of PTB. However, findings from the observational studies were not supported by interventional studies, which indicate null association.
Strengths and limitations
To date, this was the largest synthesis of observational studies and interventional studies that quantitatively assessed the association of non-occupational PA during pregnancy with the risk of PTB, which significantly increased the statistical power to detect potential associations. Specifically, we assessed the association separately for each domain of non-occupational PA. Also, all included observational studies were assessed as moderate or high quality using a standardized protocol, so that the likelihood was reduced that the pooled results were substantially biased. Nevertheless, findings from the observational studies should be interpreted in caution because of the following considerations: first, misclassification of PA levels is a concern since PA was assessed with an interview-based questionnaire during pregnancy in the primary studies, which might be subject to recall bias. However, the misclassification is likely to be non-differential and may attenuate the observed associations. To provide more accurate information on PA, objective measurements such as an accelerometer should be used. Second, although the meta-analysis was mainly based on fully adjusted models in the primary studies, the possibility that results were biased by residual confounding or unknown factors could not be completely excluded given the nature of observational study. For example, only a few primary studies considered occupational activity and socioeconomic status in the analysis. This might be an inherent limitation that might affect our findings in the meta-analysis. Third, moderate heterogeneity was observed in a couple of pooled analyses. The sources of heterogeneity include variations in study population, study region, sample size, exposure assessed at different stage of pregnancy, and adjustment for different covariates. We used a random-effects model in concordance with the heterogeneity. Fourth, publication bias due to unpublished data or publications in other languages could not be ruled out. Nevertheless, we used the Duval and Tweedie’s “trim and fill” method to adjust for publication bias. Thus, our findings should not be substantially biased. Fifth, the primary studies did not provide sufficient information to enable us to investigate some important effect modifications such as the age of the women at pregnancy.
By design, intervention studies or clinical trials have certain advantages over the observational studies. However, a few limitations should be acknowledged when interpreting the pooled results from the interventional studies in this meta-analysis. First, the sample size and the number of cases of PTB are relatively small, which indicates the statistical power may not be sufficient. Second, most of the included studies were not designed specifically for studying PTB, i.e., the primary outcome was not PTB (a rare disease) but other outcomes, such as the newborn’s body size43,44,46,48, maternal aerobic capacity change42,45 or weight gain49,51 during pregnancy, and pregnancy-induced hypertension53, which from the other angle explained the low power for the analysis. Third, the most important limitation of the interventional studies was the practical difficulty of maintaining a high compliance in the exercise group due to logistical and family constraints; similarly, the control group may be aware the benefit of exercise and consequently continue or increase their PA, which may explain the null association.
Comparison with other reviews
Several reviews54,55,56,57,58,59,60,61,62 of observational studies investigated the associations of non-occupational PA during pregnancy with the risk of PTB. While most of them concentrated on leisure-time PA, only three discussed different domains of non-occupational PA during pregnancy in relation to the risk of PTB55,56,57. Of these studies, one systematic review62 of literature up to 2014 qualitatively assessed the association of leisure-time PA during pregnancy with the risk of PTB and supported the assertion that healthy pregnant women can engage in low, moderate, and even some vigorous levels of leisure-time PA without risk for preterm birth. Another review60 quantitatively combined data from only 4 cohort studies, but found null association between leisure-time PA during pregnancy and the risk of PTB, which may be due to insufficient statistical power.
Several other reviews of interventional studies have discussed the effect of PA during pregnancy on the risk of PTB. For example, a Cochrane review published in 201063, which combined data from 3 studies with a total of 6 PTB cases concluded that the data are insufficient to draw any conclusion. In addition, a meta-analysis of interventional studies64, which used maternal weight as the primary outcome, found that PA had a trend of reducing the risk of PTB, though the pooled result from 5 trials (450 participants with 20 cases) was statistically non-significant. One recent systematic review and meta-analysis of 9 interventional studies65, including one abstract and one published in other language, concluded that aerobic exercise was not associated with an increased risk of PTB. Similarly, a meta-analysis62 of 17 trials found no significant difference in gestational age at delivery between the PA group and the control group.
Although our results are generally consistent with the previous findings, we think the present meta-analysis provides more robust results and additional information to the literature by combining evidence from both observational and interventional studies and focusing on the different domains of non-occupational PA.
Potential mechanisms
It is generally recognized that pregnant women can get tremendous benefit from regular PA. First, maintaining PA during pregnancy will help pregnant women maintain a general condition of health via improving their lipid profiles and lowering their blood pressures3,4. Second, regular PA during pregnancy will help women relieve symptoms during pregnancy (e.g., nausea and vomiting)66,67,68 via hormonal and metabolic adaptations associated with improved cardiovascular functioning and alterations in catecholamine release and response69. Third, it can help pregnant women reduce the risk of developing chronic diseases such as gestational diabetes mellitus and preeclampsia5,6 via improved insulin sensitivity, decreased concentrations of proinflammatory cytokines in peripheral circulation, reduced oxidative stress, and improved plasma lipid and lipoprotein concentrations.
There are several explanations for the potential beneficial effect of leisure-time PA on the risk of PTB. First, leisure-time PA may be less strenuous than the other two domains of non-occupational PA. Second, compared with domestic and commuting PA, women who engage in leisure-time PA may represent a select group who are more relaxed, with less stress, since gestational depression is an established risk factor of PTB70.
In addition, two cohort studies9,22 reported results on domestic PA during pregnancy and risk of PTB, in which the domestic PA was defined as lifting objects at home. Non-heavy domestic PA may provide a similar benefit as leisure-time PA after omitting these two studies. Weight lifting may raise the blood pressure and does little or nothing to benefit the heart and cardiovascular system in general, which may explain the change in the result. However, a potential effect of lifting objects at home on risk of PTB cannot be firmly established because the available data were derived from a limited number of studies.
Implications for clinical practice and future research directions
Based on the best currently available evidence, the results of this meta-analysis show a beneficial effect of leisure-time PA during pregnancy in reducing the risk of PTB. The optimal dose of PA is still unknown, but the present results recommend that appropriate leisure-time PA during pregnancy has the potential to reduce the risk of PTB. This study also indicates that non-heavy domestic PA (e.g., care giving) might benefit the pregnant women with respect to PTB. Future studies, especially well-controlled experimental/interventional studies with sufficient power, are encouraged to better understand the dose-response relationship of leisure-time PA during pregnancy and the risk of PTB.
Our systematic review lends support to the hypothesis that leisure-time physical activity during pregnancy may protect against the incidence of preterm birth. Further studies are needed to identify the most appropriate levels of intensity, duration and frequency of leisure-time PA during pregnancy. Future studies should consider the four domains of PA and potential moderators (e.g., age, race), as well as utilize tools that reliably measure exposure variables. Such studies would provide useful guidelines for pregnant women and clinicians.
In conclusion, evidence from the observational studies suggests that leisure-time PA but not commuting PA during pregnancy was inversely associated with the risk of PTB. Domestic PA may provide a similar benefit, with the exception of lifting heavy objects. Results from the observational studies are not supported by the interventional studies that indicate null associations. Future studies are needed to determine the optimal intensity and frequency of leisure-time PA during pregnancy with respect to the risk of PTB and to elucidate the potential mechanisms.
Additional Information
How to cite this article: Wen, J. et al. Non-occupational physical activity during pregnancy and the risk of preterm birth: a meta-analysis of observational and interventional studies. Sci. Rep. 7, 44842; doi: 10.1038/srep44842 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Chang, H. H. et al. Preventing preterm births: analysis of trends and potential reductions with interventions in 39 countries with very high human development index. Lancet 381, 223–234 (2013).
Blencowe, H. et al. National, regional, and worldwide estimates of preterm birth rates in the year 2010 with time trends since 1990 for selected countries: a systematic analysis and implications. Lancet 379, 2162–2172 (2012).
Brown, W. The benefits of physical activity during pregnancy. Journal of science and medicine in sport/Sports Medicine Australia 5, 37–45 (2002).
Butler, C. L., Williams, M. A., Sorensen, T. K., Frederick, I. O. & Leisenring, W. M. Relation between maternal recreational physical activity and plasma lipids in early pregnancy. American journal of epidemiology 160, 350–359 (2004).
Dempsey, J. C. et al. A case-control study of maternal recreational physical activity and risk of gestational diabetes mellitus. Diabetes research and clinical practice 66, 203–215 (2004).
Sorensen, T. K. et al. Recreational physical activity during pregnancy and risk of preeclampsia. Hypertension 41, 1273–1280 (2003).
Alderman, B. W., Zhao, H., Holt, V. L., Watts, D. H. & Beresford, S. A. Maternal physical activity in pregnancy and infant size for gestational age. Annals of epidemiology 8, 513–519 (1998).
Barnes, D. L., Adair, L. S. & Popkin, B. M. Women’s physical activity and pregnancy outcome: a longitudinal analysis from the Philippines. international Journal of Epidemiology 20, 162–172 (1991).
Misra, D. P., Strobino, D. M., Stashinko, E. E., Nagey, D. A. & Nanda, J. Effects of physical activity on preterm birth. American journal of epidemiology 147, 628–635 (1998).
Bonzini, M. et al. Occupational physical activities, working hours and outcome of pregnancy: findings from the Southampton Women’s Survey. Occupational and environmental medicine 66, 685–690 (2009).
Bonzini, M. et al. Shift work and pregnancy outcomes: a systematic review with meta-analysis of currently available epidemiological studies. BJOG: an international journal of obstetrics and gynaecology 118, 1429–1437 (2011).
Palmer, K. T., Bonzini, M., Harris, E. C., Linaker, C. & Bonde, J. P. Work activities and risk of prematurity, low birth weight and pre-eclampsia: an updated review with meta-analysis. Occupational and environmental medicine 70, 213–222 (2013).
van Melick, M. J., van Beukering, M. D., Mol, B. W., Frings-Dresen, M. H. & Hulshof, C. T. Shift work, long working hours and preterm birth: a systematic review and meta-analysis. International archives of occupational and environmental health 87, 835–849 (2014).
Bonzini, M., Coggon, D. & Palmer, K. T. Risk of prematurity, low birthweight and pre-eclampsia in relation to working hours and physical activities: a systematic review. Occupational and environmental medicine 64, 228–243 (2007).
Moher, D., Liberati, A., Tetzlaff, J., Altman, D. G. & Group, P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Journal of clinical epidemiology 62, 1006–1012 (2009).
Wells, G. et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses (2000).
Duval, S. & Tweedie, R. A nonparametric “trim and fill” method of accounting for publication bias in meta-analysis. J Am Stat Assoc 95, 89–98 (2000).
Agbla, F., Ergin, A. & Boris, N. W. Occupational working conditions as risk factors for preterm birth in Benin, West Africa. Revue d'Épidémiologie et de Santé Publique 54, 157–165 (2006).
Mamelle, N., Laumon, B. & Lazar, P. Prematurity and occupational activity during pregnancy. American journal of epidemiology 119, 309–322 (1984).
Hatch, M. C. et al. Maternal exercise during pregnancy, physical fitness, and fetal growth. American journal of epidemiology 137, 1105–1114 (1993).
Berkowitz, G. S., Kelsey, J. L., Holford, T. R. & Berkowitz, R. L. Physical activity and the risk of spontaneous preterm delivery. The Journal of reproductive medicine 28, 581–588 (1983).
Henriksen, T. B., Hedegaard, M., Secher, N. J. & Wilcox, A. J. Standing at Work and Preterm Delivery. Brit J Obstet Gynaec 102, 198–206 (1995).
Hickey, C. A. et al. Employment-related stress and preterm delivery: a contextual examination. Public health reports 110, 410–418 (1995).
Tuntiseranee, P., Geater, A., Chongsuvivatwong, V. & Kor-anantakul, O. The effect of heavy maternal workload on fetal growth retardation and preterm delivery. A study among southern Thai women. Journal of occupational and environmental medicine/American College of Occupational and Environmental Medicine 40, 1013–1021 (1998).
Cavalli, A. S. & Tanaka, T. Relationship between maternal physical activities and preterm birth. Environmental health and preventive medicine 6, 74–81 (2001).
Evenson, K. R., Siega-Riz, A. M., Savitz, D. A., Leiferman, J. A. & Thorp, J. M. Jr. Vigorous leisure activity and pregnancy outcome. Epidemiology 13, 653–659 (2002).
Orr, S. T., James, S. A., Garry, J., Prince, C. B. & Newton, E. R. Exercise and pregnancy outcome among urban, low-income, black women. Ethnicity & disease 16, 933–937 (2006).
Domingues, M. R., Barros, A. J. & Matijasevich, A. Leisure time physical activity during pregnancy and preterm birth in Brazil. International journal of gynaecology and obstetrics: the official organ of the International Federation of Gynaecology and Obstetrics 103, 9–15 (2008).
Hegaard, H. K. et al. Leisure time physical activity is associated with a reduced risk of preterm delivery. American journal of obstetrics and gynecology 198, 180.e1–180.e5 (2008).
Juhl, M. et al. Physical exercise during pregnancy and the risk of preterm birth: a study within the Danish National Birth Cohort. American journal of epidemiology 167, 859–866 (2008).
Omokhodion, F. O. et al. Paid work, domestic work, and other determinants of pregnancy outcome in Ibadan, southwest Nigeria. International journal of gynaecology and obstetrics: the official organ of the International Federation of Gynaecology and Obstetrics 111, 165–170 (2010).
Dumith, S. C., Domingues, M. R., Mendoza-Sassi, R. A. & Cesar, J. A. Physical activity during pregnancy and its association with maternal and child health indicators. Revista de saude publica 46, 327–333 (2012).
Owe, K. M., Nystad, W., Skjaerven, R., Stigum, H. & Bo, K. Exercise during pregnancy and the gestational age distribution: a cohort study. Medicine and science in sports and exercise 44, 1067–1074 (2012).
Jukic, A. M. et al. A prospective study of the association between vigorous physical activity during pregnancy and length of gestation and birthweight. Maternal and child health journal 16, 1031–1044 (2012).
Sealy-Jefferson, S., Hegner, K. & Misra, D. P. Linking nontraditional physical activity and preterm delivery in urban African-American women. Women’s health issues: official publication of the Jacobs Institute of Women’s Health 24, e389–e395 (2014).
Tinloy, J. et al. Exercise during pregnancy and risk of late preterm birth, cesarean delivery, and hospitalizations. Women’s health issues: official publication of the Jacobs Institute of Women’s Health 24, e99–e104 (2014).
Ritsmitchai, S., Geater, A. F. & Chongsuviwatvong, V. Prolonged standing and physical exertion at work during pregnancy increases the risk of preterm birth for Thai mothers. Journal of Occupational Health 39, 217–222 (1997).
Saurel-Cubizolles, M. J. Employment, working conditions, and preterm birth: results from the Europop case-control survey. Journal of Epidemiology & Community Health 58, 395–401 (2004).
Nelson, K., Lohsoonthorn, V. & Williams, M. A. Preterm Delivery Risk in Relation to Maternal Occupational and Leisure Time Physical Activity Among Thai Women. Asian biomedicine: research, reviews and news 3, 267–277 (2009).
Takito, M. Y. & Benicio, M. H. Physical activity during pregnancy and fetal outcomes: a case-control study. Revista de saude publica 44, 90–101 (2010).
Guendelman, S. et al. Association between preterm delivery and pre-pregnancy body mass index (BMI), exercise and sleep during pregnancy among working women in Southern California. Maternal and child health journal 17, 723–731 (2013).
Collings, C. A., Curet, L. B. & Mullin, J. P. Maternal and fetal responses to a maternal aerobic exercise program. American journal of obstetrics and gynecology 145, 702–707 (1983).
Bell, R. & Palma, S. Antenatal exercise and birthweight. Aust N Z J Obstet Gynaecol 40, 70–73 (2000).
Clapp, J. F. 3rd, Kim, H., Burciu, B. & Lopez, B. Beginning regular exercise in early pregnancy: effect on fetoplacental growth. American journal of obstetrics and gynecology 183, 1484–1488 (2000).
Santos, I. A. et al. Aerobic exercise and submaximal functional capacity in overweight pregnant women: a randomized trial. Obstet Gynecol 106, 243–249 (2005).
Barakat, R., Lucia, A. & Ruiz, J. R. Resistance exercise training during pregnancy and newborn’s birth size: a randomised controlled trial. Int J Obes (Lond) 33, 1048–1057 (2009).
Cavalcante, S. R. et al. Water aerobics II: maternal body composition and perinatal outcomes after a program for low risk pregnant women. Reprod Health 6, 1, doi: 10.1186/1742-4755-6-1 (2009).
Haakstad, L. A. & Bo, K. Exercise in pregnant women and birth weight: a randomized controlled trial. BMC Pregnancy Childbirth 11, 66, doi: 10.1186/1471-2393-11-66 (2011).
Ruiz, J. R. et al. Supervised exercise-based intervention to prevent excessive gestational weight gain: a randomized controlled trial. Mayo Clin Proc 88, 1388–1397 (2013).
Barakat, R., Pelaez, M., Montejo, R., Refoyo, I. & Coteron, J. Exercise throughout pregnancy does not cause preterm delivery: a randomized, controlled trial. J Phys Act Health 11, 1012–1017 (2014).
Barakat, R., Perales, M., Bacchi, M., Coteron, J. & Refoyo, I. A program of exercise throughout pregnancy. Is it safe to mother and newborn? Am J Health Promot 29, 2–8 (2014).
Ghodsi, Z. & Asltoghiri, M. Effects of aerobic exercise training on maternal and neonatal outcome: a randomized controlled trial on pregnant women in Iran. J Pak Med Assoc 64, 1053–1056 (2014).
Barakat, R. et al. Exercise during pregnancy protects against hypertension and macrosomia: randomized clinical trial. American journal of obstetrics and gynecology 214(649), e641–e648 (2016).
Schlussel, M. M., Souza, E. B., Reichenheim, M. E. & Kac, G. Physical activity during pregnancy and maternal-child health outcomes: a systematic literature review. Cadernos de saude publica 24 Suppl 4, s531–s544 (2008).
Dye, De Ver, Fernandez, T., Rains, I. D. A. & Fershteyn, Z. Recent studies in the epidemiologic assessment of physical activity, fetal growth, and preterm delivery: a narrative review. Clinical obstetrics and gynecology 46, 415–422 (2003).
Takito, M. Y., Benicio, M. H. & Neri Lde, C. Physical activity by pregnant women and outcomes for newborns: a systematic review. Revista de saude publica 43, 1059–1069 (2009).
Domingues, M. R., Matijasevich, A. & Barros, A. J. Physical activity and preterm birth: a literature review. Sports medicine 39, 961–975 (2009).
Hegaard, H. K., Pedersen, B. K., Nielsen, B. B. & Damm, P. Leisure time physical activity during pregnancy and impact on gestational diabetes mellitus, pre-eclampsia, preterm delivery and birth weight: a review. Acta obstetricia et gynecologica Scandinavica 86, 1290–1296 (2007).
Dye, T. D. & Oldenettel, D. Physical activity and the risk of preterm labor: An epidemiological review and synthesis of recent literature. Seminars in Perinatology 20, 334–339 (1996).
Savitz, D. A. & Murnane, P. Behavioral influences on preterm birth: a review. Epidemiology 21, 291–299 (2010).
Kader, M. & Naim-Shuchana, S. Physical activity and exercise during pregnancy. The European Journal of Physiotherapy 16, 2–9 (2014).
Kahn, M., Robien, K. & DiPietro, L. Maternal Leisure-time Physical Activity and Risk of Preterm Birth: A Systematic Review of the Literature. J Phys Act Health 13, 796–807 (2016).
Kramer, M. S. & McDonald, S. W. Aerobic exercise for women during pregnancy. The Cochrane database of systematic reviews CD000180, doi: 10.1002/14651858.CD000180.pub2 (2006).
Thangaratinam, S. et al. Effects of interventions in pregnancy on maternal weight and obstetric outcomes: meta-analysis of randomised evidence. Bmj 344, 377–378 (2012).
Di Mascio, D., Magro-Malosso, E. R., Saccone, G., Marhefka, G. D. & Berghella, V. Exercise during pregnancy in normal-weight women and risk of preterm birth: a systematic review and meta-analysis of randomized controlled trials. American journal of obstetrics and gynecology, doi: 10.1016/j.ajog.2016.06.014 (2016).
Hall, D. C. & Kaufmann, D. A. Effects of aerobic and strength conditioning on pregnancy outcomes. American journal of obstetrics and gynecology 157, 1199–1203 (1987).
Wallace, A. M., Boyer, D. B., Dan, A. & Holm, K. Aerobic exercise, maternal self-esteem, and physical discomforts during pregnancy. Journal of nurse-midwifery 31, 255–262 (1986).
Sternfeld, B., Quesenberry, C. P. Jr., Eskenazi, B. & Newman, L. A. Exercise during pregnancy and pregnancy outcome. Medicine and science in sports and exercise 27, 634–640 (1995).
Sternfeld, B. Physical activity and pregnancy outcome. Review and recommendations. Sports medicine 23, 33–47 (1997).
Grote, N. K. et al. A meta-analysis of depression during pregnancy and the risk of preterm birth, low birth weight, and intrauterine growth restriction. Archives of general psychiatry 67, 1012–1024 (2010).
Acknowledgements
This study was supported by two grants from the National Natural Science Foundation of China (81572213, to Y. L.; 81573140, to K. H.). Ms. J. W. was supported by the graduate abroad visiting program from Shanghai University of Sport, China (STFX20150103). The funding agencies had no role in study design, conduct, analysis, reporting or the decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Contributions
Y.L. gave funding and administrative supports. Y.L. and K.H. were responsible for conception and study design; J.W. and P.X. searched databases and reviewed literature; J.W., P.X. and C.C. extracted and analyzed data; J.W., P.X. and K.H. drafted the manuscript; J.W., P.X., C.C., M.Q., R.W., Y.L. and K.H. were responsible for critical revision of the article for important intellectual content, and final approval of the manuscript. J.W. and P.X. contributed equally to this work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Wen, J., Xun, P., Chen, C. et al. Non-occupational physical activity during pregnancy and the risk of preterm birth: a meta-analysis of observational and interventional studies. Sci Rep 7, 44842 (2017). https://doi.org/10.1038/srep44842
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep44842
- Springer Nature Limited
This article is cited by
-
Are pre- and early pregnancy lifestyle factors associated with the risk of preterm birth? A secondary cohort analysis of the cluster-randomised GeliS trial
BMC Pregnancy and Childbirth (2022)
-
Effects of Maternal Exercise During Pregnancy on Perinatal Growth and Childhood Obesity Outcomes: A Meta-analysis and Meta-regression
Sports Medicine (2021)