Abstract
Stress-induced sensitization represents a process whereby prior exposure to severe stressors leaves animals or humans in a hyper-responsive state to further stressors. Indeed, this phenomenon is assumed to be the basis of certain stress-associated pathologies, including post-traumatic stress disorder and psychosis. One biological system particularly prone to sensitization is the hypothalamic-pituitary-adrenal (HPA) axis, the prototypic stress system. It is well established that under certain conditions, prior exposure of animals to acute and chronic (triggering) stressors enhances HPA responses to novel (heterotypic) stressors on subsequent days (e.g. raised plasma ACTH and corticosterone levels). However, such changes remain somewhat controversial and thus, the present study aimed to identify the critical characteristics of the triggering and challenging stressors that affect acute stress-induced HPA cross-sensitization in adult rats. We found that HPA cross-sensitization is markedly influenced by the intensity of the triggering stressor, whereas the length of exposure mainly affects its persistence. Importantly, HPA sensitization is more evident with mild than strong challenging stressors, and it may remain unnoticed if exposure to the challenging stressor is prolonged beyond 15 min. We speculate that heterotypic HPA sensitization might have developed to optimize biologically adaptive responses to further brief stressors.
Similar content being viewed by others
Introduction
Animals exposed to acute stress immediately respond by altering their behavior and physiology. Typical behavioral changes involve reduced activity and enhanced anxiety, particularly in response to relatively severe stressors1,2. Prototypic physiological changes include the activation of components of the autonomous nervous system and the hypothalamic-pituitary-adrenal (HPA) axis. The activation of the former mainly affects its sympathetic branch, provoking important cardiovascular changes, and the release of noradrenaline and adrenaline into the bloodstream. Stress-induced activation of the HPA axis results from a convergence of the inputs to the medial parvocellular subdivision of the hypothalamic paraventricular nucleus (mpdPVN), which contains the neurons that synthesize the corticotropin-releasing hormone (CRH) and other ACTH secretagogues (e.g. vasopressin)3. CRH is considered the main hypothalamic stimulatory factor that controls both the synthesis and release of ACTH by corticotropin cells of the anterior pituitary. In turn, ACTH controls the synthesis and secretion of glucocorticoids from the adrenal cortex: cortisol in humans and most mammals; corticosterone in rats and mice. Stress-induced glucocorticoid release has a wide range of central and peripheral effects, including the mobilization of resources, the modulation of immune function and negative feedback4,5.
Although acute stress-induced changes are likely to be transient after exposure to mild stressors, exposure to severe and uncontrollable stressors can leave a trace that lasts for several days or weeks, and that may be reflected in neurochemical, neuroendocrine and behavioral changes2,6,7. Indeed, the long-lasting behavioral effects of severe stressors are reminiscent of behavioral alterations observed in humans exposed to traumatic experiences. As such, prominent behavioral perturbations are initially observed that vanish over days or weeks in most subjects, while in particularly susceptible individuals these perturbations persist for longer and they can lead to post-traumatic stress disorders and/or depression8.
Sensitization is an important consequence of exposure to certain stressors, a state of hyper-responsiveness to other stressful situations. Sensitization is particularly relevant to physiological pathologies like irritable bowel disease or chronic pain, as well as for psychiatric conditions such as post-traumatic stress disorder (PTSD), schizophrenia and drug addiction9,10,11,12,13. In animals, it is well-known that a prior history of acute stress can sensitize certain behavioral and physiological responses to novel, subsequent (heterotypic) stressors2,6,14, and this has been particularly well-characterized with regard to the HPA axis7. However, there are many cases in which prior exposure to an acute stressor does not alter the response to further stressors when typically tested in the following 24 h15. It is note that in terms of the HPA axis, cross-sensitization is typically referred to as facilitation16, yet here we prefer to use the more general concept of cross-sensitization or heterotypic sensitization.
Unfortunately, to date the characteristics that determine the magnitude and duration of stress-induced heterotypic HPA sensitization have not been studied, and several other critical questions remain unanswered. The severity of the triggering stressor is likely to have a major contribution, since HPA sensitization has mainly been reported after exposure to immobilization on boards (IMO) or electric tail-shock protocols that are typical of the classical learned helplessness procedure17,18,19,20,21,. Stressors may differ greatly and they are often difficult to compare22. Indeed, they appear to differ in terms of intensity, a property that can be objectively evaluated using classical biological stress markers like plasma catecholamines, HPA hormones and prolactin23. We are unaware of any studies that have used these biological markers to directly compare the tail-shock procedure with other stressors, yet IMO appears to be consistently stronger in our hands than other stressors like restraint, forced swim or foot-shock24,25,26,27,28. It seems reasonable to assume that the intensity of stressors will be an important influence on HPA cross-sensitization, even though this hypothesis remains to be tested. In addition, there is considerable evidence that acute stress-induced HPA sensitization is not only dependent on the intensity and/or length of the triggering stressor but also, on the duration of the heterotypic stressor. Accordingly, such effects are better observed after short-exposure to the challenging stressor17,18. This is important as heterotypic HPA sensitization could go unnoticed in sub-optimal conditions.
Therefore, this study aimed to specifically assess whether acute stress-induced HPA cross-sensitization is only observed with severe triggering stressors, and if more prolonged exposure to the acute stress could potentiate HPA sensitization. Moreover, we tested whether the length of exposure to the stressor might be critical to detect HPA sensitization (see Table 1 for a summary of the experimental design).
Results
The intensity of the triggering stress influences cross-sensitization (Experiment 1)
To demonstrate whether the intensity of the initial TRIGGERING STRESSOR affects cross-sensitization of the HPA axis and hence, its response to novel subsequent stressors, animals were assigned to four groups on day 1 (10 rats per group): CONTROL, maintained in their home cages; SWIM, subjected to forced swim for 30 min; SHOCK, receiving 30 intermittent foot-shocks over 30 min; and IMO, subjected to IMO for 30 min. These stressors were chosen on the basis that they differ in terms of intensity, as evaluated using various stress markers27,28. On day 2, all the animals were introduced individually into an open field (OF) for 15 min and their blood was sampled immediately after the test.
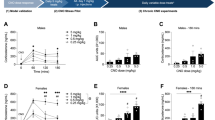
The changes in body weight from day 1 to day 2 (Fig. 1A) were analyzed using a Generalized Linear Model (GzLM) and this showed a significant effect of the TRIGGERING STRESSOR (χ2(3) = 33.6; p < 0.001), whereby there was a significant reduction of body weight in the SHOCK and IMO rats, with the strongest effect observed after IMO. To evaluate cross-sensitization of the HPA axis, we measured the plasma ACTH (Fig. 1B) and corticosterone (Fig. 1C) after a 15 min exposure to the OF, revealing a clear effect of the TRIGGERING STRESSOR on both ACTH (χ2(3) = 17.4; p = 0.001) and corticosterone (χ2(3) = 14.9; p = 0.002). Post-hoc comparisons of the ACTH levels revealed that prior foot-shock tended to enhance the response to the OF, yet such enhancement was only significant after prior IMO. By contrast, prior foot-shock and IMO increased corticosterone levels in response to the OF to the same extent.
The means and SEM (n = 10 per group) are shown and the groups labelled with different letters differ statistically (p < 0.05). The intensity of the triggering stressors (forced swim, foot-shock, IMO) was evaluated by the changes in body weight (A) from day 1 (D1) to day 2 (D2). The magnitude of ACTH and corticosterone cross-sensitization (B,C) roughly paralleled the body weight changes. Inhibition of OF activity (D–F) followed a different pattern such that prior foot-shock caused a stronger decrease than IMO.
An analysis of the behavior in the OF highlighted a significant effect of the TRIGGERING STRESSOR on peripheral ambulation (Fig. 1D; χ2(3) = 63.1; p < 0.001), central ambulation (Fig. 1E; χ2(3) = 27.0; p < 0.001) and rearing (Fig. 1F; χ2(3) = 57.0; p < 0.001). Whereas the rats in the prior SWIM group were not affected, and prior IMO reduced both peripheral and central ambulation but not rearing, the SHOCK groups displayed the strongest impact of the stressor, which markedly dampened all behaviors.
Cross-sensitization is also affected by the challenging stressor (Experiment 2)
We tested how the characteristics of the novel CHALLENGING STRESSOR affect HPA cross-sensitization, both intensity and duration. On day 1, animals were either left undisturbed (CONTROL, n = 26) or they were exposed to IMO for 2 h (IMO, n = 26). On day 2, the rats in each group were exposed to either an OF for 5 min (n = 9), or to forced swim for 5 (n = 9) or 30 min (n = 8). These two times were chosen to study the influence of the duration of the challenging stressor comparing the response to a 5 min exposure with that typically used in most experiments studying cross-sensitization (30 min)29,30. Blood samples were taken immediately after the period of stress (END) and again 30 min later (R30). This latter time point was added as sampling immediately after a 5 min stress may not accurately assess the corticosterone response but only reliably measure the ACTH changes. The statistical analysis of the effects of the different CHALLENGING STRESSORS on day 2 was performed separately as the differences in stress exposure time did not allow appropriate comparisons between the three conditions.
A Generalized Estimating Equations (GEE) analysis of the ACTH response to the 5 min OF (Fig. 2A) highlighted a significant effect of PRIOR IMO (χ2(1) = 87.6; p < 0.001), TIME (χ2(1) = 53.2; p < 0.001) and their interaction (χ2 (1) = 9.6; p = 0.002). The post-hoc comparisons showed that prior exposure to IMO enhanced the ACTH response at both time points (p < 0.001 in both cases). In terms of the corticosterone response (Fig. 2B), a significant effect of PRIOR IMO alone was evident in the GEE analysis (χ2(1) = 21.7; p < 0.001), with a higher overall response in rats previously exposed to IMO.
The means and SEM (n = 8–9 per group) of ACTH and corticosterone plasma levels are shown. The rats exposed to 2 h IMO on day 1 (D1) were exposed to a 5 min open-field (OF) or to 5 or 30 min forced swim on the following day (D2), and their blood was sampled just after the stressors (END) and again 30 min later (R30): *p < 0.05, ***p < 0.001 relative to the respective sampling time in the control group.
GEE Analysis of the ACTH response to 5 min forced swim (Fig. 2A) highlighted a significant effect of PRIOR IMO (χ2(1) = 15.5; p < 0.001), TIME (χ2(1) = 212.6; p < 0.001) and their interaction (χ2(1) = 14.2; p < 0.001). Prior exposure to IMO only enhanced the ACTH response immediately after swim (END; p < 0.001). By contrast, the GEE analysis showed only a significant effect of TIME on corticosterone (Wald χ2(1) = 21.7; p < 0.001: Fig. 2B), with no evidence of cross-sensitization. Finally, the GEE analysis of the ACTH response to 30 min swim (Fig. 2A) showed no effect of PRIOR IMO but a significant effect of TIME (χ2(1) = 250.6; p < 0.001). In terms of the corticosterone response (Fig. 2B), the GEE analysis showed a significant effect of PRIOR IMO (χ2(1) = 4.7; p = 0.031) and TIME (Wald χ2(1) = 7.5; p = 0.006), indicating mild cross-sensitization.
The influence of the duration of stress on cross-sensitization (Experiment 3)
In addition to the severity of the triggering stressors, the LENGTH of exposure to them might also influence the induction of HPA cross-sensitization. Thus, we studied the influence of the duration of the initial triggering stressor and we also compared the effect of prior prolonged exposure to IMO with that of an exogenous corticosterone administration schedule that mimicked the release of corticosterone during prolonged IMO. On day 1, the rats remained undisturbed (CONTROL, n = 10) or they were subjected to 5 min, 30 min or 4 h of IMO (n = 8 per group). An additional group of rats was administered corticosterone (5 mg/kg) twice (Sigma-Aldrich, Barcelona, Spain), at time 0 and 2 h later (B[5+5]; n = 8). Blood samples were collected from the CONTROL and IMO groups following the same schedule: just after IMO and again 45 min later (R45) with an additional sample taken from the 4 h IMO group at 1 h of IMO. Blood samples were collected from the B[5+5] animals at 1, 4 and 4:45 h after the first administration of corticosterone in order to compare the plasma ACTH and corticosterone levels with those in the 4 h IMO group. On day 2, all animals were exposed to 5 min OF (the optimum conditions to detect cross-sensitization) and blood was sampled immediately after the stress (END) and at R30. While OF behavior was videotaped, only the first 4 minutes of behavior were analyzed because for 4 rats the video camera inadvertently stopped within 5 min.
We compared the ACTH response to 4 h IMO and B[5+5] on day 1 (Fig. 3A) and the GEE analysis showed a significant effect of TREATMENT (χ2(1) = 140.7; p < 0.001), TIME (χ2(2) = 136.8; p < 0.001) and their interaction (χ2(2) = 131.4; p < 0.001). Post-hoc comparisons showed that 4 h IMO provoked higher levels of ACTH at all the time points studied (p < 0.001 in all cases). Consistent with these data, the Student’s t test revealed that the area under the curve (AUC) of the ACTH response was also higher in the 4 h IMO rats (p < 0.001: Fig. 3B). The GEE analysis of the corticosterone response (Fig. 3C) showed no effect of TREATMENT (χ2(1) = 0.02; ns) but a significant effect of TIME (χ2(2) = 46.2; p < 0.001) and the TREATMENT by TIME interaction (χ2(2) = 52.2; p < 0.001). Decomposition of the interaction showed that 4 h IMO group had more corticosterone at 1 h of IMO (p = 0.001), similar levels at 4 h of IMO and less at R45 (p < 0.001). When the AUCs of the corticosterone responses were compared (Fig. 3D) no TREATMENT effect was found (hormone data of the rest of the groups on day 1 can be seen in the Supplementary Table 1).
The means and SEM are shown. Panels A and C show the resting levels in the CONTROL group (BAS; n = 10) and the dynamics of the hormonal responses to the treatments (n = 8 per group). Blood samples were taken from the two groups following the same schedule: at 1 h IMO, just after IMO (4 h IMO) and again 45 min later (R45) in the 4 h IMO group; and at 1, 4 and 4:45 h after the first administration of corticosterone in the B [5+5] group. The areas under the curve (AUC) of the hormonal responses are shown in panels B and D: ***p < 0.001 versus 4 h IMO group.
An additional cohort of rats was studied (excluding the 30 min IMO group) to determine how the treatments on day 1 affect the resting levels of ACTH and corticosterone on day 2 (n = 6 per group). The LENGTH of prior IMO exposure affected both the plasma ACTH (χ2(3) = 43.6; p < 0.001: Fig. 4A) and corticosterone (χ2(3) = 30.5; p < 0.001: Fig. 4B) levels and accordingly, the 4 h IMO group had higher resting levels of both hormones than the other groups. By contrast, exposure to 5 min IMO increased resting levels of corticosterone but not ACTH. Administration of exogenous corticosterone had no effect on the resting hormonal levels.
The means and SEM are shown and the groups labelled with different letters differ statistically (p < 0.05). Panels A and B show resting ACTH and corticosterone levels (n = 6 per group) from a different cohort of rats, and Panels C and D show the hormonal response to the OF (n = 8–10 per group). In the latter case, blood samples were taken just after the stressor (END) and again 30 min later (R30). Only 4 h IMO resulted in reduced OF activity (Panels E–G).
The GEE analysis of the ACTH response to 5 min OF on day 2 (Fig. 4C) highlighted a significant effect of LENGTH of prior IMO (χ2(4) = 298.5; p < 0.001), TIME (χ2(1) = 45.3; p < 0.001) and their interaction (χ2(4) = 54.1; p < 0.001). The ACTH response to the OF was enhanced in all the prior IMO groups, both at END and R30, and these effects were quite independent of the length of exposure to IMO. By contrast, prior corticosterone administration strongly inhibited the ACTH response. There was a significant effect of LENGTH of prior IMO (χ2(4) = 316.7; p < 0.001), TIME (χ2(1) = 15.1; p < 0.001) and their interaction on the corticosterone response ( χ2(4) = 38.0; p < 0.001: Fig. 4D). All prior IMO groups had higher levels of corticosterone at END, an effect that was more pronounced in the 4 h IMO group. However, while prior exposure to 30 min and 4 h IMO significantly increased the corticosterone response at R30, such an increase was not produced by 5 min IMO. Prior corticosterone administration did not affect the corticosterone response to the OF at END but it was profoundly inhibited at R30.
The statistical analysis of the impact of LENGTH of IMO on OF behavior showed a marked decrease on peripheral ambulation only after prior exposure to 4 h of IMO (χ2(3) = 15.9; p = 0.003: Fig. 4E). No significant effect was found on central ambulation or rearing activity (Fig. 4F,G, respectively).
The effect of the duration of the triggering stress on the persistence of cross-sensitization (Experiment 4)
Although previous data suggest that the length of the triggering stressor influences the magnitude of HPA cross-sensitization, we wanted to assess if it also influences the PERSISTENCE of this phenomenon. Rats were subjected to IMO for 15 min or 2 h (n = 24), either 2 or 7 days (n = 6 per group) before being subjected to 10 min swim together with an additional control group (n = 6). One blood sample was obtained immediately after swim. Note that sub-optimal conditions of the heterotypic stressor were chosen to better detect group differences (swim instead of OF, and 10 min instead of 5 min). In this way we aimed to reduce the probability of finding maximum cross-sensitization two days after a 15 min IMO exposure (this is possible considering the marked cross-sensitization observed after a 5 min IMO exposure in the previous experiment) and a residual cross-sensitization of 15 min IMO seven days later.
To evaluate whether prior IMO exposure actually caused HPA cross-sensitization, we analyzed the data by GzLM including only the factor prior IMO, which had a significant effect on both ACTH (χ2(4) = 27.7; p = 0.001) and corticosterone (χ2(4) = 22.0; p < 0.001). A post-hoc analysis showed that prior IMO exposure always induced cross-sensitization of the response of both hormones, with the exception of 15 min IMO exposure that did not provoke sensitization when tested 7 days later (Fig. 5A,B). These results indicate that longer prior IMO exposure translates into more persistent cross-sensitization. This conclusion was further supported by the analysis focusing on the effects of the factors IMO LENGTH and DAYS elapsed between stressors. The GzLM analysis of ACTH and corticosterone showed a significant effect of IMO LENGTH (χ2(1) = 9.4; p = 0.002; χ2(1) = 6.8; p = 0.009; respectively) and DAYS (χ2(1) = 7.8; p = 0.005; χ2(1) = 5.0; p = 0.026; respectively) but not of their interaction, indicating that the duration of prior IMO affected both the magnitude and the persistence of cross-sensitization.
Discussion
The studies presented here define the main parameters involved in acute sensitization of the HPA response to heterotypic stressors in adult male rats. The major findings are that a brief exposure to severe stressors is enough to induce cross-sensitization, although longer exposures produce more persistent cross-sensitization. In addition, optimal cross-sensitization is observed with short exposure to the challenging stressor (i.e. 5 min) and it is masked by much longer exposure (i.e. 30 min). Finally, a lower intensity of challenging stressors favors cross-sensitization.
Although severe stressors like tail-shock (typical of the learned-helplessness paradigm) and IMO appear to consistently induce HPA cross-sensitization17,18,19,20,21,29,30, the specific contribution of the severity of the stressors has not previously been studied. We first studied whether HPA sensitization was dependent on the intensity of the triggering (inductive) stressor, exposing rats for 30 min to three stressors with putatively different intensities27,28: forced swim, foot-shock and IMO. Changes in body weight gain confirmed that IMO was the strongest stressor, followed by foot-shock and and then swim. We are aware that stressors can differ both in terms of intensity and also qualitatively, and that the overall physiological response to stressors that have a strong systemic component differs greatly (e.g. formalin-induced pain, insulin-induced hypoglycemia, cold, hemorrhage)22. However, we have systematically compared different types of stressors in which the emotional component is predominant and there are several biological markers that consistently classify them in terms of putative intensity: plasma ACTH levels, prolactin or glucose (as a surrogate of adrenaline release), and food intake or body weight gain for the most severe stressors (see ref. 23).
After exposure to the OF for 15 min on the next day, there was no sensitization of the ACTH or corticosterone response in the prior SWIM animals, whereas sensitization was evident in those exposed to prior SHOCK or IMO. A gradual effect was apparent as in SHOCK rats, sensitization was evident for the corticosterone but not the ACTH response relative to the control animals, whereas sensitization of the response of both hormones was evident in IMO rats. The lack of significant ACTH sensitization after SHOCK despite a clear corticosterone sensitization can be explained in two ways. First, sensitization might not only affect ACTH release but it may also enhance adrenal responsiveness to circulating ACTH. Second, sensitization of ACTH release could be transient (see below), mainly affecting the initial response to the stressor (i.e. first 5 min). Indeed, after a 15 min exposure ACTH levels were already declining, although sensitization is clearly observed in plasma corticosterone due to the lag time of about 15 min between the peaks of ACTH and corticosterone.
Behavior in the OF reveals a marked dissociation from the HPA response. Rats in the SWIM group did not display altered ambulatory activity in the central and peripheral part of the OF, or rearing. IMO reduced horizontal activity but not rearing, suggesting that vertical activity is more resistant to the negative impact of stress. The changes after IMO are typical of exposure to severe stressors21,31,32,33. In contrast to the relatively modest effect of IMO, foot-shock strongly inhibited both horizontal activity and rearing. The observed dissociation between HPA indexes and behavior regarding IMO and foot-shock are likely to be due to the particular properties of foot-shock in the development of cognitive generalization of fear to environments completely different from that where they experienced the foot-shock34,35,36,37. In turn, this phenomenon is associated to the acquisition of context fear conditioning35,36, a property not shared with IMO38. These results highlight the importance of the particular characteristics of stressors other than their intensity (defined by classical biological markers of stress) in determining long-lasting behavioral consequences. Indeed, while all severe emotional stressors might be able to induce protracted but relatively transient behavioral sensitization, only some of them might induce additional associative, more persistent behavioral effects (particularly foot-shock)39,40,41.
The aforementioned possibility that HPA cross-sensitization is dependent on the length of exposure to the challenging stressor is tentatively supported by the results obtained with tail-shock, whereby sensitization was clearly evident during the initial period of exposure to the novel stressor but not later17. However, whether the intensity of the heterotypic stressor is also important to produce sensitization has not been specifically addressed. Rats here were immobilized for 2 h the day before they were exposed for 5 min to an OF or to swim to study the contribution of the intensity of the heterotypic stressor, or to 30 min swim to determine the contribution of the duration of the heterotypic stressor. Marked IMO-induced cross-sensitization of the ACTH response was observed immediately after the 5 min exposure to the OF or to swim, yet only after the OF during the post-stress period, with no significant ACTH sensitization after 30 min swim. The pattern of the corticosterone response partially differed from that of ACTH, with clear sensitization after the 5 min OF, no sensitization after the 5 min swim and only modest sensitization after the 30 min swim. The lack of change in corticosterone in the post-swim period in the 5 min swim group is puzzling as ACTH sensitization was observed immediately after swim. However, it is possible that the effect on ACTH was so transient that it was not later reflected by a corticosterone response.
The results presented here strongly indicate that HPA cross-sensitization is dependent on the intensity of a challenging stressor, although it is much more strongly affected by the period of exposure to such stress. The critical contribution of the duration of exposure to heterotypic stressors might help explain the inconsistent results regarding the influence of prior chronic stress on the heterotypic sensitization of the HPA axis to emotional stress. A consistent response to the heterotypic stressor is not evident when assessed 30–60 min after exposure42,43,44,45,46,47, yet cross-sensitization is consistently observed when the exposure to the challenging stressor was more immediate (about 5 min)30,48,49. For instance, daily repeated exposure to IMO causes marked cross-sensitization of the ACTH response to a 5 min OF30, whereas the results differ when the same chronic stressor is coupled to a longer exposure of the challenging stressor50.
It is noteworthy that sensitization is particularly evident after a short exposure to the heterotypic stressors, which can be explained in different ways. First, and with the exception of social stressors, in nature emotional stressors are usually of short duration and therefore the adaptive biological consequences of HPA sensitization might have evolved to respond to such stimuli. After this initial period, sensitization could be counteracted by other events, probably related to the absence of signs of true danger. Alternatively, sensitization could actually represent an acceleration rather than an increase in the HPA response to novel stressors, offering advantages when confronted with acute stressors. For instance, faster glucocorticoid release could help improve memory consolidation related to short-lasting stressful situations.
While acute severe stressors consistently induce HPA cross-sensitization, the temporal threshold of these stressors to induce sensitization is unknown. We immobilized rats for 5 min, 30 min or 4 h and studied sensitization of the HPA response to a short OF exposure on the following day. Surprisingly, HPA sensitization was of similar magnitude after the 5 and 30 min IMO, with only a small increase after 4 h IMO. These results not only demonstrate that HPA sensitization can be produced by a brief exposure to traumatic stressors but also, that negative glucocorticoid feedback is probably not operative under stress as the prolonged activation of the HPA axis caused by 4 h IMO did not impair the HPA response to additional stress on the next day. The lack of effect of stress-induced glucocorticoid release on negative feedback is consistent with the hypothesis that exposure to stress facilitates HPA responsiveness, thereby overcoming glucocorticoid negative feedback16. This facilitation may also explain the normal response to stress in the 4 h IMO group, despite the elevated resting levels of corticosterone prior to exposure to the challenging stressor. To directly demonstrate that exogenous administration of glucocorticoids is far from mimicking the complex effects of stress on the activity of the HPA axis, we introduced a group of rats given exogenous corticosterone to simulate the release caused by 4 h IMO. Although plasma corticosterone levels were somewhat higher after IMO than after corticosterone administration at 1 h, these levels were similar at 4 h and somewhat lower at R45. Consequently, the AUCs were similar, even though exposure to 4 h IMO caused sensitization whereas corticosterone administration completely blocked the ACTH response to the OF, indicating that a strong negative feedback was still present at this time. Although the impact of glucocorticoid negative feedback on acute stress-induced HPA responsiveness is not usually tested in animals on the day after a single administration of glucocorticoids, there is some evidence for similar long-lasting effects of cortisol administration51. As such, it appears that IMO-induced facilitation of the HPA axis could fully overcome glucocorticoid negative feedback. Alternatively, glucocorticoid released during a stressful situation may not trigger negative feedback because such a mechanism is not operative under stress. This second possibility is compatible with our earlier findings that administration of the glucocorticoid receptor (GR) antagonist mifepristone prior to IMO did not further enhance IMO-induced HPA cross-sensitization21.
Although a brief exposure to IMO is enough to induce HPA cross-sensitization, the persistence of the sensitized state could be dependent on the length of initial exposure to IMO. This possibility was tested in suboptimal conditions of the challenging stressor to better discriminate between the effects of two IMO periods, both of which elicited ACTH and corticosterone cross-sensitization, although the effect was greater after longer IMO. Seven days after IMO, significant sensitization was restricted to this latter IMO group, albeit of lower magnitude than that observed on the following day. Hence, HPA cross-sensitization is longer lasting after a more prolonged exposure to the inductive stressor.
The precise mechanisms involved in acute stress-induced cross-sensitization of the HPA axis remain largely unknown7, although this phenomenon appears to be located at the level of the PVN, or above rather than at the level of the anterior pituitary19. Interestingly, pharmacological activation of the HPA axis with metyrapone does not induce HPA cross-sensitization21, despite a marked activation of CRH gene expression and ACTH release52,53. Moreover, cross-sensitization does not require activation of CRH-1 receptors21. Hence, HPA cross-sensitization might be specifically associated with strong emotional activation caused by severe stressors. Since cross-sensitization was observed after a short exposure to IMO, severity rather than duration would appear to determine sensitization, which is important when considering the possible consequences of trauma in humans. Unfortunately, to the best of our knowledge there are no studies into HPA responsiveness to stress on the days following exposure to traumatic situations in humans.
It is unclear to what extent the data on chronic stress-induced cross-sensitization, and particularly that related to the facilitation of the HPA axis, can be extrapolated to acute stress. Moreover, the inconsistency of chronic stress-induced facilitation of the response to heterotypic stressors54,55,56 represents an additional problem. Chronic stress is likely to be affected by the same factors as those described for acute stress-induced HPA facilitation, for instance the duration of the challenging stressor or the type and/or intensity of the challenging stressor45. Moreover, models of chronic stress that include severe systemic stressors are more likely to detect cross-sensitization, as is the case of hypertonic saline57, chronic cold exposure58,59 (although see ref. 60) and some models of chronic unpredictable stress61,62. Importantly, the systemic nature of the stressor appears to be critical for sensitization of the central noradrenergic system (see ref. 7). Localizing brain areas where a prior history of chronic stress could induce enhanced neuronal activation (as evaluated by c-fos and other immediate early genes) has given inconsistent results that are difficult to explain43,46,58,61,63,64.
There is evidence that the posterior region of the paraventricular thalamic nucleus (pPVTh) plays a role in chronic stress-induced cross-sensitization, while not influencing the HPA response to acute stressors in stress-naïve rats58. Some neuropeptides that act at the pPVTh appear to play important roles in cross-sensitization. Orexin inputs to the nucleus are important for the induction but not for the expression of facilitation65, whereas the expression of the phenomenon is restrained by cholecystokinin acting through CCK-B receptors59. Unfortunately, the inputs to the pPVth in this process and the pathways linking the pPVTh to the PVN remain unclear. Nevertheless, HPA cross-sensitization probably does not involve impaired fast or delayed negative glucocorticoid feedback44,62. Although the non-hypothalamic brain CRH pathways appear to influence the induction or expression of cross-sensitization between chronic stress and addictive drugs, as well as some effects of chronic stress on brain noradrenergic activity66,67,68,69,70, there is no evidence of a critical role of CRH in chronic-stress induced cross-sensitization of the HPA axis.
In conclusion, the results presented here demonstrate that HPA sensitization is critically dependent on the characteristics of both the triggering (inductive) and the challenging stressor. Specifically, the intensity of the inductive stressor is more critical than its duration, although longer exposures to stressors can induce more persistent sensitization. Rather than its intensity, the length of exposure to the triggering stressor is most important to provoke sensitization, which may explain the controversial results in the literature and particularly those regarding chronic stress-induced HPA sensitization.
Materials and Methods
Animals and general procedures
Male Sprague-Dawley rats from the Animal Facility of the Universitat Autònoma de Barcelona (UAB) were used in these studies. The rats were 2–3 months old at the beginning of the experiments and they were housed in pairs under standard conditions: temperature (21 ± 1 °C), 12 h light cycle (lights on 07:00–19:00 h), and food and water ad libitum. All animals were habituated to handling (at least three times) and blood-sampling by the tail-nick procedure21 (once) prior to the experiments, which were always carried out in the morning. Cage-mates were always tested simultaneously. The experimental protocol was approved by the Ethics Committee at the Universitat Autònoma de Barcelona and the Generalitat de Catalunya, and it was carried out in accordance to the European Council Directive (2010/63/UE) and Spanish legislation (RD 53/2013).
Stressors
Forced swim: rats were placed individually in transparent cylindrical plastic tanks (height = 40 cm, internal diameter = 19 cm) containing water (36 °C) to a level of 24 cm71.
Foot-shock: rats were placed in shock chambers made of clear Plexiglas boxes (57 × 41 × 70 cm) with a removable metal grid on the floor composed of 44 stainless steel rods (0.4 cm diameter) that were spaced 1.5 cm apart, center-to-center38, and they received one 3 s, 1.0 mA, foot-shock each min.
Immobilization (IMO): the four limbs of the rats were taped to metal mounts attached to a board, as described previously21.
Open-field test (OF): The OF was a plastic gray rectangular box (56 × 36.5 × 31 cm) open at the top, where each animal was initially placed facing a corner. Exposure to novel environments is a mild stress that allows behavioral assessment. Between animals, the apparatus was cleaned carefully with tap water containing ethanol (5% v/v). OF behavior was recorded with a video camera (Sony SSC-M388 CE, BW) situated 150 cm above the center of the cage. The box was divided into 12 areas (10 of them in contact with the walls and 2 in the center of the box) and the number of areas crossed (peripheral and central) was calculated manually21.
Radioimmunoassays
Plasma ACTH and corticosterone levels were analyzed using radioimmunoassays as described previously72. All samples to be statistically compared were processed in the same assay to avoid inter-assay variability.
Statistical analysis
The statistical package for social science (SPSS, version 23 for Windows) software was used for the analysis. The AUCs (calculated using Graph-Pad Prism 5.0) of the hormonal data were analyzed using a 2-tailed Student’s t-test. For the rest of the comparisons, statistical analyses were performed using the GzLM when only between-subjects factors were included73 and the GEE model when within-subjects factors were present in the analysis74. The between-subjects factor was either the treatment on day 1 or day 2, and the sampling time was the within-subjects factor (when more than one blood sample was obtained). These models are a more flexible statistical tool than the standard general linear model (GLM) because several types of distribution and different covariance structures of the repeated measures data could be chosen. In addition, the GzLM does not require homogeneity of variances and it admits missing values without removing all the subject data. The significance of the effects was determined with the Wald chi-square statistic (χ2). After the main analysis, appropriate pair-wise comparisons were carried out using the sequential Bonferroni corrections. The criterion for significance was set at p < 0.05 for all tests.
Additional Information
How to cite this article: Belda, X. et al. Critical features of acute stress-induced cross-sensitization identified through the hypothalamic-pituitary-adrenal axis output. Sci. Rep. 6, 31244; doi: 10.1038/srep31244 (2016).
References
Wiedenmayer, C. P. Adaptations or pathologies? Long-term changes in brain and behavior after a single exposure to severe threat. Neurosci Biobehav Rev. 28, 1–12 (2004).
Armario, A., Escorihuela, R. M. & Nadal, R. Long-term neuroendocrine and behavioural effects of a single exposure to stress in adult animals. Neurosci Biobehav Rev. 32, 1121–1135 (2008).
Armario, A. The hypothalamic-pituitary-adrenal axis: what can it tell us about stressors? CNS Neurol Disord Drug Targets. 5, 485–501 (2006).
Sapolsky, R. M., Romero, L. M. & Munck, A. U. How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative actions. Endocr Rev. 21, 55–89 (2000).
Frank, M. G., Watkins, L. R. & Maier, S. F. Stress-induced glucocorticoids as a neuroendocrine alarm signal of danger. Brain Behav Immun. 33, 1–6 (2013).
Maier, S. F. & Watkins, L. R. Stressor controllability and learned helplessness: the roles of the dorsal raphe nucleus, serotonin, and corticotropin-releasing factor. Neurosci Biobehav Rev. 29, 829–841 (2005).
Belda, X., Fuentes, S., Daviu, N., Nadal, R. & Armario, A. Stress-induced sensitization: the hypothalamic-pituitary-adrenal axis and beyond. Stress. 18, 269–279 (2015).
Davidson, J. R., Stein, D. J., Shalev, A. Y. & Yehuda, R. Posttraumatic stress disorder: acquisition, recognition, course, and treatment. J Neuropsychiatry Clin Neurosci. 16, 135–147 (2004).
Overmier, J. B., Murison, R. & Milde, A. M. Sensitization and conditioning as contributors to gastrointestinal vulnerability. Auton Neurosci. 125, 22–27 (2006).
Stam, R. PTSD and stress sensitisation: a tale of brain and body Part 1: human studies. Neurosci Biobehav Rev. 31, 530–557 (2007).
Collip, D., Myin-Germeys, I. & Van Os, J. Does the concept of “sensitization” provide a plausible mechanism for the putative link between the environment and schizophrenia? Schizophr Bull. 34, 220–225 (2008).
Robinson, T. E. & Berridge, K. C. Review. The incentive sensitization theory of addiction: some current issues. Philos Trans R Soc Lond B Biol Sci. 363, 3137–3146 (2008).
Ursin, H. Brain sensitization to external and internal stimuli. Psychoneuroendocrinology. 42, 134–145 (2014).
Rau, V., DeCola, J. P. & Fanselow, M. S. Stress-induced enhancement of fear learning: an animal model of posttraumatic stress disorder. Neurosci Biobehav Rev. 29, 1207–1223 (2005).
Martí, O., Andrés, R. & Armario, A. Defective ACTH response to stress in previously stressed rats: dependence on glucocorticoid status. Am J Physiol. 277, R869–R877 (1999).
Dallman, M. F. & Jones, M. T. Corticosteroid feedback control of ACTH secretion: effect of stress-induced corticosterone secretion on subsequent stress responses in the rat. Endocrinology. 92, 1367–1375 (1973).
Johnson, J. D. et al. Prior stressor exposure primes the HPA axis. Psychoneuroendocrinology. 27, 353–365 (2002).
O’Connor, K. A. et al. Inescapable shock induces resistance to the effects of dexamethasone. Psychoneuroendocrinology. 28, 481–500 (2003).
O’Connor, K. A. et al. Stress-induced sensitization of the hypothalamic-pituitary adrenal axis is associated with alterations of hypothalamic and pituitary gene expression. Neuroendocrinology. 80, 252–263 (2004).
Belda, X., Fuentes, S., Nadal, R. & Armario, A. A single exposure to immobilization causes long-lasting pituitary-adrenal and behavioral sensitization to mild stressors. Horm Behav. 54, 654–661 (2008).
Belda, X., Daviu, N., Nadal, R. & Armario, A. Acute stress-induced sensitization of the pituitary-adrenal response to heterotypic stressors: independence of glucocorticoid release and activation of CRH1 receptors. Horm Behav. 62, 515–524 (2012).
Pacak, K. et al. Heterogeneous neurochemical responses to different stressors: a test of Selye’s doctrine of nonspecificity. Am J Physiol. 275, R1247–R1255 (1998).
Armario, A. et al. What can we know from pituitary-adrenal hormones about the nature and consequences of exposure to emotional stressors? Cell Mol Neurobiol. 32, 749–758 (2012).
Armario, A., Martí, J. & Gil, M. The serum glucose response to acute stress is sensitive to the intensity of the stressor and to habituation. Psychoneuroendocrinology. 15, 341–347 (1990).
Campmany, L., Pol, O. & Armario, A. The effects of two chronic intermittent stressors on brain monoamines. Pharmacol Biochem Behav. 53, 517–523 (1996).
García, A., Martí, O., Vallés, A., Dal-Zotto, S. & Armario, A. Recovery of the hypothalamic-pituitary-adrenal response to stress. Effect of stress intensity, stress duration and previous stress exposure. Neuroendocrinology. 72, 114–125 (2000).
Márquez, C., Belda, X. & Armario, A. Post-stress recovery of pituitary-adrenal hormones and glucose, but not the response during exposure to the stressor, is a marker of stress intensity in highly stressful situations. Brain Res. 926, 181–185 (2002).
Rabasa, C. et al. Adaptation of the hypothalamus-pituitary-adrenal axis to daily repeated stress does not follow the rules of habituation: A new perspective. Neurosci Biobehav Rev. 56, 35–49 (2015).
Gagliano, H., Fuentes, S., Nadal, R. & Armario, A. Previous exposure to immobilisation and repeated exposure to a novel environment demonstrate a marked dissociation between behavioral and pituitary-adrenal responses. Behav Brain Res. 187, 239–245 (2008).
Daviu, N., Rabasa, C., Nadal, R. & Armario, A. Comparison of the effects of single and daily repeated immobilization stress on resting activity and heterotypic sensitization of the hypothalamic-pituitary-adrenal axis. Stress. 17, 176–185 (2014).
Lehnert, H., Reinstein, D. K., Strowbridge, B. W. & Wurtman, R. J. Neurochemical and behavioral consequences of acute, uncontrollable stress: effects of dietary tyrosine. Brain Res. 303, 215–223 (1984).
Kennett, G. A., Dickinson, S. L. & Curzon, G. Central serotonergic responses and behavioural adaptation to repeated immobilisation: the effect of the corticosterone synthesis inhibitor metyrapone. Eur J Pharmacol. 119, 143–152 (1985).
Berridge, C. W. & Dunn, A. J. Restraint-stress-induced changes in exploratory behavior appear to be mediated by norepinephrine-stimulated release of CRF. J Neurosci. 9, 3513–3521 (1989).
Van Dijken, H. H., Van der Heyden, J. A., Mos, J. & Tilders, F. J. Inescapable footshocks induce progressive and long-lasting behavioural changes in male rats. Physiol Behav. 51, 787–794 (1992).
Radulovic, J., Kammermeier, J. & Spiess, J. Generalization of fear responses in C57BL/6N mice subjected to one-trial foreground contextual fear conditioning. Behav Brain Res. 95, 179–189 (1998).
Daviu, N., Fuentes, S., Nadal, R. & Armario, A. A single footshock causes long-lasting hypoactivity in unknown environments that is dependent on the development of contextual fear conditioning. Neurobiol Learn Mem. 94, 183–190 (2010).
Daviu, N., Andero, R., Armario, A. & Nadal, R. Sex differences in the behavioural and hypothalamic-pituitary-adrenal response to contextual fear conditioning in rats. Horm Behav. 66, 713–723 (2014).
Daviu, N., Delgado-Morales, R., Nadal, R. & Armario, A. Not all stressors are equal: behavioral and endocrine evidence for development of contextual fear conditioning after a single session of footshocks but not of immobilization. Front Behav Neurosci. 6, 69 (2012).
Kamprath, K. & Wotjak, C. T. Nonassociative learning processes determine expression and extinction of conditioned fear in mice. Learn Mem. 11, 770–786 (2004).
Siegmund, A. & Wotjak, C. T. A mouse model of posttraumatic stress disorder that distinguishes between conditioned and sensitised fear. J Psychiatr Res. 41, 848–860 (2007).
Sauerhofer, E. et al. Generalization of contextual fear depends on associative rather than non-associative memory components. Behav Brain Res. 233, 483–493 (2012).
Mansi, J. A. & Drolet, G. Chronic stress induces sensitization in sympathoadrenal responses to stress in borderline hypertensive rats. Am J Physiol. 272, R813–R820 (1997).
Chung, K. K., Martinez, M. & Herbert, J. C-fos expression, behavioural, endocrine and autonomic responses to acute social stress in male rats after chronic restraint: modulation by serotonin. Neuroscience. 95, 453–463 (2000).
Bhatnagar, S. & Vining, C. Facilitation of hypothalamic-pituitary-adrenal responses to novel stress following repeated social stress using the resident/intruder paradigm. Horm Behav. 43, 158–165 (2003).
Spiga, F. et al. Effect of vasopressin 1b receptor blockade on the hypothalamic-pituitary-adrenal response of chronically stressed rats to a heterotypic stressor. J Endocrinol. 200, 285–291 (2009).
Weinberg, M. S. et al. Repeated ferret odor exposure induces different temporal patterns of same-stressor habituation and novel-stressor sensitization in both hypothalamic-pituitary-adrenal axis activity and forebrain c-fos expression in the rat. Endocrinology. 150, 749–761 (2009).
Babb, J. A., Masini, C. V., Day, H. E. & Campeau, S. Habituation of hypothalamic-pituitary-adrenocortical axis hormones to repeated homotypic stress and subsequent heterotypic stressor exposure in male and female rats. Stress. 17, 224–234 (2014).
Hauger, R. L., Lorang, M., Irwin, M. & Aguilera, G. CRF receptor regulation and sensitization of ACTH responses to acute ether stress during chronic intermittent immobilization stress. Brain Res. 532, 34–40 (1990).
Lachuer, J., Delton, I., Buda, M. & Tappaz, M. The habituation of brainstem catecholaminergic groups to chronic daily restraint stress is stress specific like that of the hypothalamo-pituitary-adrenal axis. Brain Res. 638, 196–202 (1994).
Armario, A., Hidalgo, J. & Giralt, M. Evidence that the pituitary-adrenal axis does not cross-adapt to stressors: comparison to other physiological variables. Neuroendocrinology. 47, 263–267 (1988).
Filaretov, A. A. & Balashov Yu, G. Corticosteroid feedback: I. General characteristics, role of adrenal cortex. Exp Clin Endocrinol. 91, 143–148 (1988).
Herman, J. P., Schafer, M. K., Thompson, R. C. & Watson, S. J. Rapid regulation of corticotropin-releasing hormone gene transcription in vivo . Mol Endocrinol. 6, 1061–1069 (1992).
Rotllant, D., Ons, S., Carrasco, J. & Armario, A. Evidence that metyrapone can act as a stressor: effect on pituitary-adrenal hormones, plasma glucose and brain c-fos induction. Eur J Neurosci. 16, 693–700 (2002).
Ostrander, M. M., Ulrich-Lai, Y. M., Choi, D. C., Richtand, N. M. & Herman, J. P. Hypoactivity of the hypothalamo-pituitary-adrenocortical axis during recovery from chronic variable stress. Endocrinology. 147, 2008–2017 (2006).
Choi, D. C. et al. The anteroventral bed nucleus of the stria terminalis differentially regulates hypothalamic-pituitary-adrenocortical axis responses to acute and chronic stress. Endocrinology. 149, 818–826 (2008).
Choi, D. C. et al. The role of the posterior medial bed nucleus of the stria terminalis in modulating hypothalamic-pituitary-adrenocortical axis responsiveness to acute and chronic stress. Psychoneuroendocrinology. 33, 659–669 (2008).
Kiss, A. & Aguilera, G. Regulation of the hypothalamic pituitary adrenal axis during chronic stress: responses to repeated intraperitoneal hypertonic saline injection. Brain Res. 630, 262–270 (1993).
Bhatnagar, S. & Dallman, M. Neuroanatomical basis for facilitation of hypothalamic-pituitary-adrenal responses to a novel stressor after chronic stress. Neuroscience. 84, 1025–1039 (1998).
Bhatnagar, S. et al. A cholecystokinin-mediated pathway to the paraventricular thalamus is recruited in chronically stressed rats and regulates hypothalamic-pituitary-adrenal function. J Neurosci. 20, 5564–5573 (2000).
Vining, C., Iyer, V. & Bhatnagar, S. Intracerebroventricular administration of corticotrophin-releasing hormone receptor antagonists produces different effects on hypothalamic pituitary adrenal responses to novel restraint depending on the stress history of the animal. J Neuroendocrinol. 19, 198–207 (2007).
Radley, J. J. & Sawchenko, P. E. Evidence for involvement of a limbic paraventricular hypothalamic inhibitory network in hypothalamic-pituitary-adrenal axis adaptations to repeated stress. J Comp Neurol. 523, 2769–2787 (2015).
Franco, A. J. et al. Sensitization of the Hypothalamic-Pituitary-Adrenal Axis in a Male Rat Chronic Stress Model. Endocrinology. 157, 2346–2355 (2016).
Ostrander, M. M. et al. Chronic stress produces enduring decreases in novel stress-evoked c-fos mRNA expression in discrete brain regions of the rat. Stress. 12, 469–477 (2009).
Singewald, G. M., Nguyen, N. K., Neumann, I. D., Singewald, N. & Reber, S. O. Effect of chronic psychosocial stress-induced by subordinate colony (CSC) housing on brain neuronal activity patterns in mice. Stress. 12, 58–69 (2009).
Heydendael, W. et al. Orexins/hypocretins act in the posterior paraventricular thalamic nucleus during repeated stress to regulate facilitation to novel stress. Endocrinology. 152, 4738–4752 (2011).
Cole, B. J. et al. Central administration of a CRF antagonist blocks the development of stress-induced behavioral sensitization. Brain Res. 512, 343–346 (1990).
Finlay, J. M. et al. Impact of corticotropin-releasing hormone on extracellular norepinephrine in prefrontal cortex after chronic cold stress. J Neurochem. 69, 144–150 (1997).
Jedema, H. P., Finlay, J. M., Sved, A. F. & Grace, A. A. Chronic cold exposure potentiates CRH-evoked increases in electrophysiologic activity of locus coeruleus neurons. Biol Psychiatry. 49, 351–359 (2001).
Breese, G. R., Knapp, D. J. & Overstreet, D. H. Stress sensitization of ethanol withdrawal-induced reduction in social interaction: inhibition by CRF-1 and benzodiazepine receptor antagonists and a 5-HT1A-receptor agonist. Neuropsychopharmacology. 29, 470–482 (2004).
Boyson, C. O. et al. Social stress and CRF-dopamine interactions in the VTA: role in long-term escalation of cocaine self-administration. J Neurosci. 34, 6659–6667 (2014).
Dal-Zotto, S., Martí, O. & Armario, A. Influence of single or repeated experience of rats with forced swimming on behavioural and physiological responses to the stressor. Behav Brain Res. 114, 175–181 (2000).
Rabasa, C., Muñoz-Abellán, C., Daviu, N., Nadal, R. & Armario, A. Repeated exposure to immobilization or two different footshock intensities reveals differential adaptation of the hypothalamic-pituitary-adrenal axis. Physiol Behav. 103, 125–133 (2011).
McCulloch, C. & Searle, S. Generalized, linear and mixed models (John Wiley & Sons, 2001).
Hardin, J. W. & Hilbe, J. M. Generalized estimating equations (Chapman & Hall/CRC, 2003).
Acknowledgements
The work in the laboratory was supported by Spanish grants to AA and/or RN from the “Plan Nacional sobre Drogas” (2011/021), “Ministerio de Economía y Competitividad” (SAF2014-53876R), “Instituto de Salud Carlos III” (RD12/0028/0014, “Redes Temáticas de Investigación Cooperativa en Salud”, “Ministerio de Sanidad y Consumo” and “Generalitat de Catalunya” (SGR2014-1020). RN is the recipient of an ICREA-ACADEMIA award (Generalitat de Catalunya). The funding sources played no role either in the design, collection, analysis and interpretation of the data, or in the decision to submit the article for publication.
Author information
Authors and Affiliations
Contributions
A.A. and X.B. were responsible for the concept and study design; R.N. participated in the design of the study, and in the analysis and discussion of the results; X.B. was the main person responsible for all the experiments, data and statistical analyses; A.A. wrote the manuscript. All the authors have reviewed the manuscript and approved the final version submitted for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Belda, X., Nadal, R. & Armario, A. Critical features of acute stress-induced cross-sensitization identified through the hypothalamic-pituitary-adrenal axis output. Sci Rep 6, 31244 (2016). https://doi.org/10.1038/srep31244
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep31244
- Springer Nature Limited
This article is cited by
-
Embryonic exposure to native and alien predator cues tunes tadpole defensive behaviour
Aquatic Ecology (2023)
-
How Past Work Stressors Influence Psychological Well-Being in the Face of Current Adversity: Affective Reactivity to Adversity as an Explanatory Mechanism
Journal of Business and Psychology (2023)
-
Impact of impaired glucose metabolism on responses to a psychophysical stressor: modulation by ketamine
Psychopharmacology (2021)
-
Psychostimulants and forced swim stress interaction: how activation of the hypothalamic-pituitary-adrenal axis and stress-induced hyperglycemia are affected
Psychopharmacology (2017)